
| High-technology and environmental applications of the rare earth elements (REE) have grown dramatically in diversity and importance over the past four decades. As many of these applications are highly specific, in that substitutes for the REE are inferior or unknown, the REE have acquired a level of technological significance much greater than expected from their relative obscurity. Although actually more abundant than many familiar industrial metals, the REE have much less tendency to become concentrated in exploitable ore deposits. Consequently, most of the world’s supply comes from only a few sources. The United States once was largely self-sufficient in REE, but in the past decade has become dependent upon imports from China. |
The rare earth elements (REE) form the largest chemically coherent group in the periodic table. Though generally unfamiliar, the REE are essential for many hundreds of applications. The versatility and specificity of the REE has given them a level of technological, environmental, and economic importance considerably greater than might be expected from their relative obscurity. The United States once was largely self-sufficient in these critical materials, but over the past decade has become dependent upon imports (fig. 1). In 1999 and 2000, more than 90% of REE required by U.S. industry came from deposits in China.
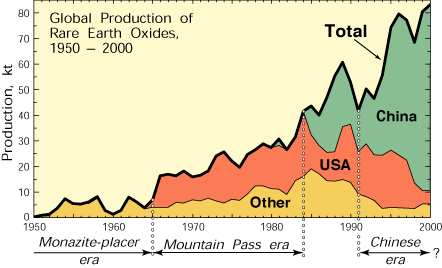 |
| Figure 1. Global rare earth element production (1 kt=106 kg) from 1950 through 2000, in four categories: United States, almost entirely from Mountain Pass, California; China, from several deposits; all other countries combined, largely from monazite-bearing placers; and global total. Four periods of production are evident: the monazite-placer era, starting in the late 1800s and ending abruptly in 1964; the Mountain Pass era, starting in 1965 and ending about 1984; a transitional period from about 1984 to 1991; and the Chinese era, beginning about 1991. |
Although the
15 naturally occurring REE (table 1; fig. 2) are generally similar in their
geochemical properties, their individual abundances in the Earth are by no means
equal. In the continental crust and its REE ore deposits, concentrations of
the most and least abundant REE typically differ by two to five orders of magnitude
(fig. 3). As technological applications of REE have multiplied over the past
several decades, demand for several of the less abundant (and formerly quite
obscure) REE has increased dramatically.
The diverse
nuclear, metallurgical, chemical, catalytic, electrical, magnetic, and optical
properties of the REE have led to an ever increasing variety of applications.
These uses range from mundane (lighter flints, glass polishing) to high-tech
(phosphors, lasers, magnets, batteries, magnetic refrigeration) to futuristic
(high-temperature superconductivity, safe storage and transport of hydrogen
for a post-hydrocarbon economy).
Some Applications
of the Rare Earth Elements
Many applications
of REE are characterized by high specificity and high unit value. For example,
color cathode-ray tubes and liquid-crystal displays used in computer monitors
and televisions employ europium as the red phosphor; no substitute is known.
Owing to relatively low abundance and high demand, Eu is quite valuable—$250
to $1,700/kg (for Eu2O3) over the past decade.
Fiber-optic
telecommunication cables provide much greater bandwidth than the copper wires
and cables they have largely replaced. Fiber-optic cables can transmit signals
over long distances because they incorporate periodically spaced lengths of
erbium-doped fiber that function as laser amplifiers. Er is used in these laser
repeaters, despite its high cost (~$700/kg), because it alone possesses the
required optical properties.
Specificity is not limited to the more exotic REE, such as Eu or Er. Cerium, the most abundant and least expensive REE, has dozens of applications, some highly specific. For example, Ce oxide is uniquely suited as a polishing agent for glass. The polishing action of CeO2 depends on both its physical and chemical properties, including the two accessible oxidation states of cerium, Ce,3+ and Ce4+, in aqueous solution. Virtually all polished glass products, from ordinary mirrors and eyeglasses to precision lenses, are finished with CeO2.
| Table 1. Names and symbols of the REE | ||||
| La | lanthanum | Tb | terbium | |
| Ce | cerium | Dy | dysprosium | |
| Pr | praseodymium | Ho | holmium | |
| Nd | neodymium | Er | erbium | |
| Pm | promethium | Tm | thulium | |
| Sm | samarium | Yb | ytterbium | |
| Eu | europium | Lu | lutetium | |
| Gd | gadolinium | Y | yttrium | |
Permanent magnet
technology has been revolutionized by alloys containing Nd, Sm, Gd, Dy, or Pr.
Small, lightweight, high-strength REE magnets have allowed miniaturization of
numerous electrical and electronic components used in appliances, audio and
video equipment, computers, automobiles, communications systems, and military
gear. Many recent technological innovations already taken for granted (for example,
miniaturized multi-gigabyte portable disk drives and DVD drives) would not be
possible without REE magnets.
Environmental
applications of REE have increased markedly over the past three decades. This
trend will undoubtedly continue, given growing concerns about global warming
and energy efficiency. Several REE are essential constituents of both petroleum
fluid cracking catalysts and automotive pollution-control catalytic converters.
Use of REE magnets reduces the weight of automobiles. Widespread adoption of
new energy-efficient fluorescent lamps (using Y, La, Ce, Eu, Gd, and Tb) for
institutional lighting could potentially achieve reductions in U.S. carbon dioxide
emissions equivalent to removing one-third of the automobiles currently on the
road. Large-scale application of magnetic-refrigeration technology (described
below) also could significantly reduce energy consumption and CO2 emissions.
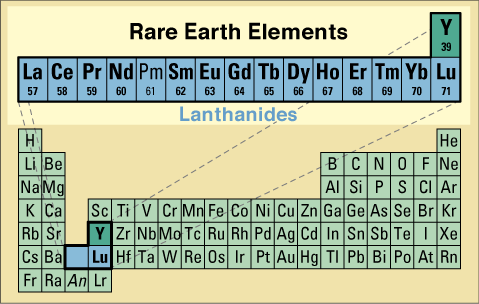 |
| Figure 2. Chemical periodic table delineating the 16 rare earth elements (REE): the lanthanides, La through Lu, plus Y, whose geochemical behavior is virtually identical to that of the heavier lanthanides. Promethium has no long-lived isotopes and occurs naturally on Earth only in vanishingly small quantities. An represents the first 14 actinide elements; Lr is the last actinide. |
In many applications,
REE are advantageous because of their relatively low toxicity. For example,
the most common types of rechargeable batteries contain either cadmium (Cd)
or lead. Rechargeable lanthanum-nickel-hydride (La-Ni-H) batteries are gradually
replacing Ni-Cd batteries in computer and communications applications and could
eventually replace lead-acid batteries in automobiles. Although more expensive,
La-Ni-H batteries offer greater energy density, better charge-discharge characteristics,
and fewer environmental problems upon disposal or recycling. As another example,
red and red-orange pigments made with La or Ce are superseding traditional commercial
pigments containing Cd or other toxic heavy metals.
The next high-technology
application of the REE to achieve maturity may be magnetic refrigeration. The
six REE ions Gd3+ through Tm3+ have unusually large magnetic moments, owing
to their several unpaired electrons. A newly developed alloy, Gd5(Si2Ge2), with
a “giant magne-tocaloric effect” near room temperature reportedly
will allow magnetic refrigeration to become competitive with conventional gas-compression
refrigeration. This new technology could be employed in refrigerators, freezers,
and residential, commercial, and automotive air conditioners. Magnetic refrigeration
is considerably more efficient than gas-compression refrigeration and does not
require refrigerants that are flammable or toxic, deplete the Earth’s ozone
layer, or contribute to global warming.
“Rare”
earth elements is a historical misnomer; persistence of the term reflects unfamiliarity
rather than true rarity. The more abundant REE are each similar in crustal concentration
to commonplace industrial metals such as chromium, nickel, copper, zinc, molybdenum,
tin, tungsten, or lead (fig. 4). Even the two least abundant REE (Tm, Lu) are
nearly 200 times more common than gold. However, in contrast to ordinary base
and precious metals, REE have very little tendency to become concentrated in
exploitable ore deposits. Consequently, most of the world’s supply of REE
comes from only a handful of sources.
Differences in abundances of individual REE in the upper continental crust of the Earth (figs. 3, 4) represent the super-position of two effects, one nuclear and one geochemical. First, REE with even atomic numbers (58Ce, 60Nd, …) have greater cosmic and terrestrial abundances than adjacent REE with odd atomic numbers (57La, 59Pr, …). Second, the lighter REE are more incompatible (because they have larger ionic radii) and therefore more strongly concentrated in the continental crust than the heavier REE. In most rare earth deposits, the first four REE—La, Ce, Pr, and Nd—constitute 80 to 99% of the total. Therefore, deposits containing relatively high grades of the scarcer and more valuable heavy REE (HREE: Gd to Lu, Y) and Eu are particularly desirable.
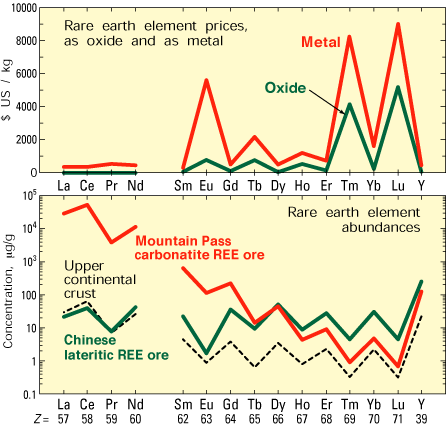 |
| Figure 3. Prices and abundances of the rare earth elements. Prices are for 1999 or 2000 in U.S. dollars per kilogram of REE metal, in two forms: (1) as oxides, in 2 to 25 kg packages, at 95 to 99.99% purity; (2) as 0.1 to 0.45 kg metal ingot, at 99.9% purity. Two representative REE ores—high-grade carbonatite ore from Mountain Pass, California, and lateritic ion-adsorption ore from southern China—are compared with Earth’s upper continental crust. Z, atomic number. |
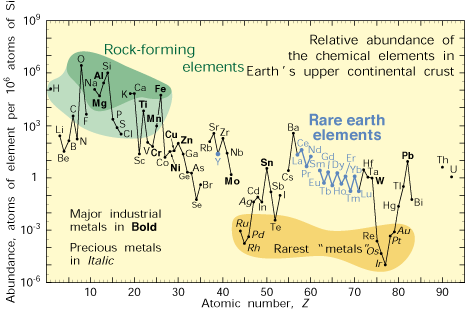 |
| Figure 4. Abundance (atom fraction) of the chemical elements in Earth’s upper continental crust as a function of atomic number. Many of the elements are classified into (partially overlapping) categories: (1) rock-forming elements (major elements in green field and minor elements in light green field); (2) rare earth elements (lanthanides, La–Lu, and Y; labeled in blue); (3) major industrial metals (global production >~3x107 kg/year; labeled in bold); (4) precious metals (italic); and (5) the nine rarest “metals”—the six platinum group elements plus Au, Re, and Te (a metalloid). |
From the discovery
of the REE (during the period 1794–1907) through the mid-1950s, a few of
the REE were produced in modest amounts from monazite-bearing placers and veins,
from pegmatites and carbonatites, and as minor byproducts of uranium and niobium
extraction. During this time, the middle and heavy REE generally were available
in pure form only in sub-kilogram quantities and were chiefly chemical curiosities.
In 1949, a
carbonatite intrusion with extraordinary contents of light REE (8 to 12% rare
earth oxides [REO]), was discovered at Mountain Pass, in the upper Mojave Desert,
California (fig. 5). The REE at Mountain Pass are hosted chiefly by bastnäsite,
(Ce,La,Nd,…)CO3F, and related minerals. By 1966, this single, world-class
deposit (owned and operated by Molycorp, Inc.) had become the paramount source
of REE. Early development was supported largely by the sudden demand for Eu
created by the commercialization of color television. Mountain Pass, with an
average grade of 9.3% and reserves of 20 million metric tons (Mt) REO (at 5%
cutoff), remains the only large ore deposit mined solely for its REE content.
Mountain Pass ore is very strongly dominated by the light REE (figs. 3, 6).
Nonetheless, the large quantities of ore processed and development of solvent-extraction
techniques for large-scale separation of individual REE from one another allowed
recovery of several of the middle REE. Increased availability led in turn to
applications for these formerly exotic elements.
From 1965 through
the mid-1980s, Mountain Pass was the dominant source of REE, and the United
States was largely self-sufficient in REE. Since 1985, production of REE in
China has increased dramatically (fig. 1). Chinese REE production comes chiefly
from two sources. The most important is the Bayan Obo iron-niobium-REE deposit,
Inner Mongolia. This deposit has geological affinities both to carbonatite REE
deposits and to hydrothermal iron-oxide (-Cu-Au-REE) deposits, such as Olympic
Dam, Australia, and Kiruna, Sweden. Grades at Bayan Obo are 3 to 6% REO; reserves
are at least 40 Mt, possibly considerably more. The second major source of Chinese
REE is ion-adsorption ores in lateritic weathering crusts developed on granitic
and syenitic rocks in tropical southern China. These oxide ores are advantageous
in their relatively high proportions of HREE (fig. 6) and, especially, in the
ease with which they can be mined and the REE extracted.
The number
of workable REE deposits, already severely limited by the geochemical properties
of the REE, has in recent years also been affected by environmental and regulatory
factors. Monazite, the single most common REE mineral, generally contains elevated
levels of thorium. Although Th itself is only weakly radioactive, it is accompanied
by highly radioactive intermediate daughter products, particularly radium, that
can accumulate during processing. Concern about radioactivity hazards has now
largely eliminated monazite as a significant source of REE and focused atten-tion
on those few deposits where the REE occur in other, low-Th minerals, particularly
bastnäsite.
Over the past several years the only domestic source of REE, the mine at Mountain Pass, California, has operated below capacity and only intermittently. Following environmental and regulatory problems with the main wastewater pipeline, the REE separation (solvent extraction) plant was shut down. Mountain Pass currently produces only bastnäsite concentrates and sells separated REE only from stockpiles produced before the shutdown. Even after the regulatory situation has been resolved, however, the long-term viability of Mountain Pass as a supplier of separated REE for high-technology applications is threatened by market factors.
 |
| Figure 5. The Mountain Pass rare earth element mine, Mojave Desert, California, with Clark Mountain (part of the Mojave National Preserve) in the background and Interstate 15 just beyond the hills in the foreground. |
In 1999 and 2000, nearly all (more than 90%) of the separated REE used in the United States was imported either directly from China or from countries that imported their plant feed materials from China. The surprisingly rapid progression from self-sufficiency prior to about 1990 to nearly complete dependence on imports from a single country today involves a number of causative factors. These include much lower labor and regulatory costs in China than in the United States; continued expansion of electronics and other manufacturing in Asia; the favorable number, size, and HREE content of Chinese deposits; and the ongoing environmental and regulatory problems at Mountain Pass. China now dominates world REE markets (fig. 1), raising several important issues of REE supply for the United States:
(1) The United States is in danger of losing its longstanding leadership in many areas of REE technology. Transfer of expertise in REE processing technology and REE applications from the United States and Europe to Asia has allowed China to develop a major REE industry, eclipsing all other countries in production of both ore and refined products. The Chinese Ministry of Science and Technology recently announced a new national basic research program. Among the first group of 15 high-priority projects to be funded was “Basic research in rare earth materials” (Science, Dec. 18, 1998, p. 2171).
(2) United States dependence on imports from China comes at a time when REE have become increasingly important in defense applications, including jet fighter engines and other aircraft components, missile guidance systems, electronic countermeasures, underwater mine detection, antimissile defense, range finding, and space-based satellite power and communication systems.
(3) Availability of Chinese REE to U.S. markets depends on continued stability in China’s internal politics and economy, and its relations to other countries.
(4) Although the present low REE prices caused by abundant supply from China are stressing producers, particularly Mountain Pass, low prices will also stimulate development of new applications. For example, a recent chemistry text notes that “… for many years the main use of lutetium was the study of the behavior of lutetium …” Several promising applications for Lu are known, but most are precluded by high cost. If the price of Lu were to decrease from many thousand to a few thousand dollars per kilogram (fig. 3), additional high-technology applications of even this least abundant of the REE undoubtedly would follow. How large a role the United States will play in future expansion of REE technology and markets remains an important, but open, question.
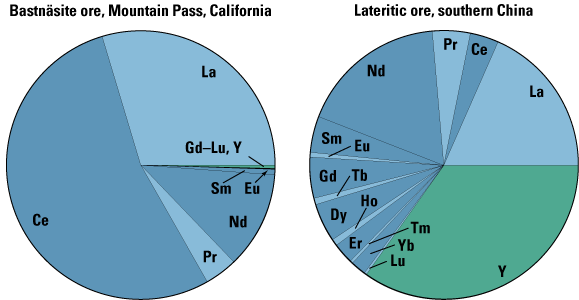 |
| Figure 6. Proportions of individual REE in two representative ores: bastnäsite, dominated by La, Ce, and Nd, with Eu through Lu plus Y totaling only 0.4%; and lateritic ion-adsorption ore, Y-dominated. Dark blue and light blue sectors represent lanthanides of even and odd atomic number, respectively (see figs. 2, 3). Yttrium is indicated by green. |
The rare earth elements are essential for a diverse and expanding array of high-technology applications, which constitute an important part of the industrial economy of the United States. Long-term shortage or unavailability of REE would force significant changes in many technological aspects of American society. Domestic REE sources, known and potential, may therefore become an increasingly important issue for scientists and policymakers in both the public and private sectors.
By Gordon B. Haxel, James B. Hedrick, and Greta J. Orris
Edited by
Peter H. Stauffer and James W. Hendley II
Graphic design by Gordon B. Haxel, Sara Boore, and Susan Mayfield; Web layout by Carolyn Donlin
For questions about the content of this report, contact Gordon Haxel
For more information see: http://minerals.usgs.gov/minerals/pubs/commodity/rare_earths/
PDF version of this fact sheet
Download a copy of Acrobat Reader for free
| Help | PDF help | Geopubs main page | Fact Sheets |
| Department
of the Interior | Privacy Statement
| Disclaimer | Accessibility
|
URL of this
page: https://pubs.usgs.gov/fs/2002/fs087-02
Maintained by: Michael Diggles
Created: November 20, 2002
Last modified: May 17, 2005 (mfd)