Fact Sheet 2011–3070
 |
| Figure 1. Block diagram showing karstic system of aquifers, generalized components of the hydrologic cycle, and potential opportunities for contaminated recharge. - click to enlarge |
The hydrogeology of Florida, especially in the northern part of the state, and southwestern Georgia is characterized by a predominance of limestone aquifers overlain by varying amounts of sands, silts, and clays. This karstic system of aquifers and their associated springs is particularly vulnerable to contamination from various anthropogenic activities at the land surface. Numerous sinkholes, disappearing streams, and conduit systems or dissolution pathways, often associated with large spring systems, allow rapid movement of contaminants from the land surface to the groundwater system with little or no attenuation or degradation (fig. 1). The fate of contaminants in the groundwater system is not fully understood, but traveltimes from sources are greatly reduced when conduits are intercepted by pumping wells and springs. Contaminant introduction to groundwater systems in Florida and Georgia is not limited to seepage from land surface, but can be associated with passive (drainage wells) and forced subsurface injection (aquifer storage and recovery, waste-water disposal).
USGS research and collaborative science investigations of contaminant transport in groundwater systems have provided water resource managers and regulators in Florida and Georgia mechanisms to support their management and regulatory roles. Hydrologic research often seeks to answer questions relative to traveltime and fate of contaminants. Information about the age of groundwater can be used to define recharge rates, refine hydrologic models of groundwater systems, predict contamination potential, and estimate the time needed to flush contaminants from groundwater systems (Plummer and Friedman, 1999). A more thorough knowledge of these processes is needed to accurately assess the effects of land-use or regulatory and mitigation efforts on water quality.
Current (2011) and past research in Florida and Georgia have focused on the presence and transport of several existing and potential anthropogenic compounds in shallow and deep groundwater systems: solvents, disinfection by-products, other volatile organic compounds (VOCs), nutrients (such as nitrate), pharmaceutical compounds, and pesticides. Also, high levels of naturally occurring contaminants such as radon and arsenic, have been detected in some groundwater drinking-water sources (Katz and others, 2009b). Some recently investigated sources of nutrients in groundwater are fertilizer, septic-tank effluent in rural areas, animal wastes, and treated municipal sewage effluent used for irrigation. Although undesirable in drinking water, the aforementioned compounds can be used in conjunction with modeling activities to determine the timing and location of recharge, and to assess transport through groundwater systems.
Chemical indicators are used in an effort to determine when groundwater contamination occurs and where the affected water flows. Plummer (2004) explained that it is important to measure these environmental tracers, as these data will enable scientists, water resource managers, and managers to:
When tracking contaminants in groundwater, the chemical composition of the water and the length of time from source input to sampling event must be known. Typically, several indicators are used in concert with physical parameters and common inorganic ions when determining time of travel to aid in the age determination. Anthropogenic activities since the mid-1940s, including atmospheric testing of thermonuclear devices and variety of industrial processes, have released tritium (3H), chlorofluorocarbons (CFCs -- trichlorofluoromethane, CCl3F, CFC-11; dichlorodifluoromethane, CCl2F2, CFC-12; and trichlorotrifluoroethane, C2Cl3F3, CFC-113), and sulfur hexafluoride (SF6) into the atmosphere at low but measurable concentrations (Katz and others, 2001). Precipitation that reaches the land surface conveys these chemicals into the groundwater system and provides a "time stamp" for the atmospheric conditions present at the time of recharge. As a result, water samples collected from springs and wells can be analyzed for these compounds, and an apparent age of the water sample computed. A brief discussion follows regarding the usefulness of analyses; thorough discussions of theory and analytical methods have been published by Schlosser and others (1988; 1989) and Busenberg and Plummer (1992; 2000)
An increase of 3H in rainfall occurred with atmospheric weapons testing beginning in the early 1950s. Concentrations of 3H in Florida increased to several hundred tritium units (TU; 1 TU is equal to 1 3H atom in 1018 hydrogen atoms) during the mid-1960s, then decreased logarithmically to the present (2010) levels (fig 2). Combined measurements of 3H and 3He (radioactive decay product of tritium decay) define a relatively stable tracer of the initial 3H input to groundwater that can be used to calculate an apparent 3H/3He age (Katz and others, 2005). Age dating using this method is advantageous over CFCs and SF6 because 3H/3He ages are not affected by contamination, sorption, or microbial degradation processes that can alter the others. However, a disadvantage of using 3H/3He analyses is concentrations and ratios can be affected by hydrodynamic dispersion and mixing of waters with various ages (Solomon and Sudicky, 1991; Reilly and others, 1994).
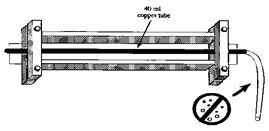 |
| Figure 3. Diagram of apparatus used to collect water samples for 3H/3He analyses. - click to enlarge |
Samples for 3H/3He analyses are collected using the apparatus illustrated below (fig. 3). Water running through copper tubes (3/8-inch diameter, 30-inch length, containing about 40 milliliters of water, and fitted with stainless steel pinch-off clamps at each end) is captured after flushing the copper tubes and hoses connecting the sampling apparatus to the pump for about 5 minutes, thus assuring that no air is in the hoses or tubes.
Various industrial processes have released CFCs into the atmosphere in low but measurable concentrations since the late 1940s (fig. 2). Independent apparent ages for CFCs are estimated based on the gas-air equilibrium of CFC-11, CFC-12, and CFC-113 at the base of the unsaturated zone at the time of recharge (Katz, 2004). The CFC-dating technique relies on the stability of these gases in the hydrosphere, which has led to their effective use as tracers to age-date groundwater recharged since 1940 (Plummer and Busenberg, 1999). Apparent ages can be estimated if CFC concentrations in the aquifer have not been altered by biological or geochemical processes and the isotopes are from a atmospheric source.
Sampling for CFC analyses consists of pumping from a purged well or directly from a spring vent and overfilling / overflowing a sample bottle set inside a beaker that is taller than the bottle (fig. 4). When at least 2 liters of water has overflowed from the beaker, the fill tube is removed from the bottle, and the cap (also submerged in the beaker) is screwed tightly onto the bottle while remaining submerged. Extreme care is taken not to allow any air into the bottle.
Sulfur hexafluoride (SF6) is a trace atmospheric gas that is primarily of anthropogenic origin (industrial processes) and is relatively stable in the hydrosphere, making it effective as a tracer of water recharged in the last 30 years. Groundwater can be dated with SF6 if it is in equilibrium with atmospheric SF6 at the time of recharge and does not contain significant SF6 from other sources. It is a suitable dating tool of post-1990s groundwater because, unlike the CFCs with steady or declining relative concentrations, atmospheric concentrations of SF6 are expected to continue increasing (Busenberg and Plummer, 2000).
Water sample collection for SF6 analyses is a simple technique achieved by filling from the bottom of a 1-liter plastic safety coated, amber glass bottle through a fill tube inserted into the bottle (fig. 5). After 2 liters of water have flowed into the bottle, the filling tube is slowly removed while water is still flowing and a cap is tightly screwed and taped onto the bottle, being careful not to allow any headspace to remain in the bottle.
Although analytical methods are capable of determining extremely low concentrations of 3H, 3He, CFCs, and SF6 in groundwater, the subsequent interpretation of age or mean residence time of water in complex karstic aquifer systems contains considerable uncertainty. In many studies, flow systems are relatively well characterized and water samples are collected from discrete depth intervals. Extending tracer age-dating techniques to springs or wells with large open intervals in complex karstic systems can be accomplished by applying lumped-parameter models whose output are matched with field-measured tracer concentrations (Katz and others, 2009a).
Lumped-parameter models used in Florida research typically are applied (1) assuming a steady-state flow system, and (2) that selected tracers behave like a water molecule. Although this assumption is valid for 3H, which is part of the water molecule, CFCs and SF6 may or may not be transported in exactly the same way as the water due to sorption and biogeochemical processes (Plummer and Busenberg, 1999) that can cause model results to vary from observed data. Piston-flow, exponential, and binary mixing models have all been used successfully when computing apparent ages of Florida groundwater (Katz and others, 2005, 2007; Katz and Griffin, 2008). Piston-flow (time-constant flow lines) and exponential mixing (time-varying flow lines) models (Mook, 2000), or a combination of the two, are used when groundwater flow and discharge are associated with a single hydrogeologic unit. The binary mixing model is used when the contribution of flow from the well or spring vent is probably from units with vastly different times of recharge. To reiterate, these models are used to help determine the residence time of the water and to minimize uncertainty of the derived age.
Much can be learned from the inorganic and organic constituents occurring naturally and anthropogenically in groundwater as it flows from its source of recharge. Concentrations of major ions and dissolved gases in the groundwater, as well as other characteristics such as temperature and pH, provide information about the processes that affect concentrations of other chemical constituents (Katz and others, 2007). Differences in groundwater type between hydrogeologic units are used to determine groundwater flow patterns and pathways, and to generate hypotheses for processes controlling water quality in water samples from wells and springs.
Water samples analyzed for anthropogenic contaminants, when evaluated with aquifer hydraulic characteristics, provide critical data for water resource managers, regulators, and scientists that can be used to address contamination problems. Commonly used analytes for understanding specific hydrologic issues are isotopes of nitrogen, oxygen, and carbon, as well as pharmaceuticals and organic waste compounds (OWCs). The presence of pesticides and VOCs certainly indicates anthropogenic influences on recharge to an aquifer.
Nitrogen isotope fractionation (partial distribution in equilibrium) provides researchers with a powerful tool to differentiate between human or animal waste and fertilizer sources in groundwater or surface water. Low ratios of the nitrogen-15 isotope to nitrogen-14 as nitrate (δ15N-NO3; values 0 to 3 per mil) generally indicate an inorganic nitrate source (artificial or synthetic fertilizer); whereas higher values (10 to 20 per mil) generally indicate an organic source of nitrate (for example, animal wastes from manure spreading, human wastes from septic tank effluent). The evaluation of sources and cycling of nitrate in the environment can be aided by analysis of the oxygen (17O and 18O) isotopic composition of nitrate (Kendall and Doctor, 2007). Of the nearly 80 per mil range in δ18O for major nitrate reservoirs, the approximately 20 to 70 per mil spread is from precipitation while the -5 to 15 per mil spread is from nitrification of ammonium and organic matter. δ18O values of nitrate present in soils, streams, and groundwaters typically contain mixtures of atmospheric and microbial nitrate.
Carbon isotopes of dissolved inorganic carbon (DIC) can be valuable tools for understanding the biogeochemical reactions controlling carbonate alkalinity in groundwater and watersheds under favorable conditions (Kendall and Doctor, 2007). The δ13C of DIC can be a useful tracer of the seasonal and discharge-related contributions of different hydrologic flow paths to surface streamflow and to shallow groundwater discharge from springs in carbonate and fractured rock terrains. In carbonate terrains, the extent of chemical evolution of the δ13C of carbon species in groundwater can be used to distinguish between groundwaters traveling along different flow paths.
The presence of various pharmaceutical compounds or OWCs in groundwater samples indicates contamination from humans introduced typically as treated sewage effluent or septic tank leachate (Katz and Griffin, 2008). Pharmaceutical compounds commonly found in groundwater have included widely used prescription and over-the-counter drugs and certain degradates/metabolites of these compounds. A typical analysis of OWCs includes fragrances and flavorants, flame retardants, antioxidants, fuel-related compounds, detergent metabolites, plasticizers, disinfectants, solvents and preservatives, polynuclear aromatic hydrocarbons (PAHs), pesticides, plant and animal steroids, caffeine, and others. Sampling for pharmaceutical compounds and OWCs includes filtering water through 0.7-µm nominal pore size glass fiber filters and storing in 1-L glass bottles maintained at 4 °C during shipment for analyses.
Human enteroviruses and Hepatitis A viruses have been used in recent years to successfully determine the source of fecal contamination (Griffin and others, 1999; 2003). Two other groups of viruses, noroviruses, and adenoviruses have been utilized in conjunction with enteroviruses and HAV for source tracking in marine ecosystems. These three groups of viruses have been utilized to determine whether human wastewater exists as a constant presence and whether it responds to surge and precipitation events. Additionally, these viruses can adapt and live in shallow aquatic sediments and terrestrial soils resulting in consistently elevated concentrations in nearshore waters. The benefit of using these viruses is that they are only shed in human feces and, if detected, directly identify human wastewater as a source of contaminant, unlike the standard bacterial indicators (fecal coliforms and enterococci) that are shed in the feces of humans and other animals. Viral source tracking is library independent and, thus, is more accurate than bacterial source tracking assays.
Elevated bacterial indicator concentrations are frequently reported in coastal areas and streams, but it is unknown whether their presence is due to a regional source of fecal matter or if certain species have adapted to sediments and soils. Salt gradient culturing (to imitate river to coast transport survival) coupled with restriction fragment length polymorphism (RFLP, fragmenting DNA to create a DNA fingerprint) of Escherichia coli (a bacteria within the fecal coliform group) is a useful tool to determine whether certain subsets or types of indicator organisms are more suited for survival in marine environments than the general like-members which have been freshly shed.
Combined assays (viral and bacterial) enable water resources managers, regulators, and scientists to determine (Dale Griffin, U.S. Geological Survey, written commun., 2010):
Recent (2010) U.S. Geological Survey research in Florida and Georgia have utilized the aforementioned tools for better understanding some of the commonly occurring hydrologic processes. Higher than normal background levels of nutrients in groundwater and streams in southwest Georgia have drawn attention to current agricultural practices in the Flint River basin. Isotope analyses have indicated a fertilizer signature in streamwater sampled adjacent to the farming areas, whereas analyses of Apalachicola and Chattahoochee River, as well as Lake Seminole samples, revealed a combination of fertilizer and animal waste signatures (B.G. Katz, U.S. Geological Survey, oral commun., 2010).
The effects of traditional septic system installation in areas of thin, well-drained sediments overlying karstic limestone has caused an approximately 25 to 40 percent N loss as septic tank effluent moves through the unsaturated zone to the water table. This loss is due to denitrification, ammonium sorption, and ammonia volatilization (Katz and others, 2010). Because the drainfield is below the soil horizon, indicator bacteria and human enteric viruses were detected only intermittently in soil water and groundwater. Most OWCs and pharmaceutical compounds were highly attenuated beneath the drainfields; however, five OWCs (caffeine, 1,7-dimethylxanthine, phenol, galaxolide, and tris(dichloroisotopropyl)phosphate) and two pharmaceutical compounds (acetaminophen and sulfameth-oxazole) were detected in groundwater samples.
Geochemical and microbiological techniques were used to assess water-quality impacts from the land application of treated municipal wastewater in northern Florida. Nitrate-N concentrations have increased from about 0.2 to as high as 1.1 milligrams per liter since 1980 in Wakulla Springs where water discharges from the Upper Floridan aquifer (Katz and others, 2009a). Mixing of sprayfield effluent water was indicated by a systematic decrease in concentrations of sample analytes (chemical indicators) with distance downgradient from the sprayfield, with about a 10-fold dilution at Wakulla Springs. The research found the sprayfield operation was highly effective in removing most studied OWCs, pharmaceutical compounds, and microbial indicators.
Recent bacterial indicator data for the St. Marks and Wakulla Rivers indicates elevated concentrations in the upper freshwater areas of each river. These concentrations sharply decrease at the more saline regions of the St. Marks River south of where the two rivers merge. This finding indicates that the elevated bacterial indicators consistently observed at Shell Point’s beach are coastal in nature and that either bacterial populations have adapted to the sediments and soils or they are emitted to the coastal realm by humans or animals (D.W. Griffin, U.S. Geological Survey, written commun., 2010).
Busenberg, E., and Plummer, L.N., 1992, Use of chlorofluoromethanes (CCl3F and CCl2F2) as hydrologic tracers and age dating tools: Example – the alluvium and terrace system of central Oklahoma: Water Resources Research, v. 28, p. 2257–2283.
Busenberg, E., and Plummer, L.N., 2000, Dating young ground water with sulfur hexafluoride – natural and anthropogenic sources of SF6: Water Resources Research, v. 36, p. 3011–3030.
Griffin, D.W., Gibson, C.J., III, Lipp, E.K., Riley, K., Paul, J.H., and Rose, J.B., 1999, Detection of viral pathogens by RT-PCR and of microbial indicators using standard methods in the canals of the Florida Keys: Applied and Environmental Microbiology, v. 65, no. 9, p. 4118-4125.
Griffin, D.W., Donaldson, K.A., Paul, J.H., and Rose, J.B., 2003, Pathogenic human viruses in coastal waters: Clinical Microbiology Reviews, v. 16, no. 1, p. 129-143.
Katz, 2004, Sources of nitrate contamination and age of water in large karstic springs of Florida: Environmental Geology, v. 46, p 689-706.
Katz, B.G., Böhlke, J.K., and Hornsby, H.D., 2001, Timescales for nitrate contamination of spring waters, northern Florida, USA: Chemical Geology, v. 179, p. 167-186.
Katz, B.G., Copeland, Rick, Grenhalgh, Tom, Ceryak, Ron, and Zwanka, Warren, 2005, Using chemical indicators to assess sources of nitrate and age of groundwater in a karstic spring basin: Environmental & Engineering Geology,v. XI, no. 4, pp. 333-346.
Katz, B.G., Crandall, C.A., Metz, P.A., McBride, S., and Berndt, M.P., 2007, Chemical characteristics, water sources and pathways, and age distribution of ground water in the contributing recharge area of a public-supply well near Tampa, Florida, 2002-05: U.S. Geological Survey Scientific Investigations Report 2007-5139, 83 p.
Katz, B.G., and Griffin, D.W., 2008, Using chemical and microbiological indicators to track the impacts from the land application of treated municipal wastewater and other sources on groundwater quality in a karstic springs basin: Environmental Geology,v. 55, p. 801-821.
Katz, B.G., Griffin, D.W., and Davis, H.D., 2009a, Groundwater quality impacts from the land application of treated municipal wastewater in a large karstic spring basin: Chemical and microbiological indicators: Science of the Total Environment, v. 407, p. 2872-2886.
Katz, B.G., McBride, W.S., Hunt, A.G., Crandall, C.A., Metz, P.A., Eberts, S.M., and Berndt, M.P., 2009b, Assessing vulnerability of a public supply well in a karstic aquifer to contamination using ground-water age measurements and geochemical indicators: Journal of Ground Water, v. 47, no. 3, p. 438-452.
Katz, B.G., Griffin, D.W., McMahon, P.B., Harden, H.S., Wade, E., Hicks, R.W., and Chanton, J.P., 2010, Fate of effluent-borne contaminants beneath septic tank drainfields overlying a karst aquifer. Journal of Environmental Quality, v. 39, pp. 1181-1195.
Kendall, C., and Doctor, D.H., 2007, Stable isotope applications in hydrologic studies: Treatise on Geochemistry,v. 5, chap 5.11, p. 319-364.
Mook, W.G., ed., 2000, UNESCO/IAEA series on environmental isotopes in the hydrologic cycle, Principles and Applications: Modeling, v. VI, 125 p.
Plummer, L.N., 2004, Dating of young ground water with CFCs, SF6, 3H, and 3H/3He: Examples from the Blue Ridge and Valley and Ridge of parts of Virginia and Pennsylvania, accessed January 2010, at http://water.usgs.gov/lab/presentations/dating_young_groundwater/gwmeeting_6_2004.pdf.
Plummer L.N., and Busenberg, E., 1999, Chlorofluorocarbons, in Cook, P.G., and Herczeg, A.L., eds., Environmental tracers in subsurface hydrology: Kluwer Academic Press, chap. 15, p. 441-478.
Plummer, L.N., and Friedman, L.C., 1999, Tracing and dating young ground water: U.S. Geological Survey Fact Sheet FS-134-99, 4 p., accessed October 12, 2010, at https://pubs.usgs.gov/fs/FS-134-99/pdf/fs-134-99.pdf.
Reilly, T.E., Plummer, L.N., Phillips, P.J., and Busenberg, E., 1994, The use of simulation and multiple environmental tracers to quantify groundwater flow in a shallow aquifer: Water Resources Research,v. 30, p. 421–433.
Schlosser, P., Stute, M., Dorr, H., Sonntag, C., and Munnich, K.O., 1988, Tritium/3He dating of shallow groundwater: Earth Planetary Science Letters,v. 89, p. 353–362.
Schlosser P., Stute, M., Sonntag, C., and Munnich, K.O., 1989, Tritiogenic 3He in shallow ground water: Earth Planetary Science Letters,v. 94, p. 245–256.
Solomon, D. K., and Sudicky, E.A., 1991, Tritium and helium-3 isotope ratios for direct estimation of spatial variations in groundwater recharge: Water Resources Research,v. 27, p. 2309–2319.
Mahon, G.L., 2011, Assessment of Groundwater Pathways and Contaminant Transport in Florida and Georgia using Multiple Chemical and Microbiological Indicators: U.S. Geological Survey Fact Sheet 2011–3070, https://pubs.usgs.gov/fs/2011/3070/. (Available online only.)
For more information, contact:
Director
Florida Water Science Center
U.S. Geological Survey
4446 Pet Lane, Suite 108
Lutz, Florida 33559
Tel: 813-498-5000
Fax: 813-498-5002
http://fl.water.usgs.gov/