This document is also available in pdf format: ![]() fs-134-99.pdf
fs-134-99.pdf
Data on concentrations of environmental tracers, such as chlorofluorocarbons (CFCs), tritium (3H), and other chemical and isotopic substances in ground water, can be used to trace the flow of young water (water recharged within the past 50 years) and to determine the time elapsed since recharge. Information about the age of ground water can be used to define recharge rates, refine hydrologic models of ground-water systems, predict contamination potential, and estimate the time needed to flush contaminants from ground-water systems. CFCs also can be used to trace seepage from rivers into ground-water systems, provide diagnostic tools for detection and early warning of leakage from landfills and septic tanks, and to assess susceptibility of water-supply wells to contamination from near-surface sources.
During the past 50 years, human activities have released an array of chemical and isotopic substances to the atmosphere. In the atmosphere, these substances have mixed and spread worldwide. These atmospheric substances, such as tritium (3H) in water vapor from detonation of nuclear bombs in the 1950s and early 1960s,and chlorofluorocarbons (CFCs) from refrigeration and other uses from the 1950s through the 1980s, dissolve in precipitation, become incorporated in the Earth’s hydrologic cycle, and can be found in ground water that has been recharged within the past 50 years. The detection of chlorofluorocarbons and tritium in ground water provides valuable information that can be used for dating and tracing young ground water—techniques that help water-resources managers develop management strategies for shallow ground-water systems.
Young ground water in shallow ground-water systems
Young ground water is typically found at depths from 0 to 100 feet in unconsolidated sediments and at depths up to 1000 feet in fractured-rock systems. Shallow ground-water systems are commonly used for drinking water sources and they make up a large part of the baseflow in rivers and lakes. However, shallow ground-water supplies are generally young (recently recharged) and, because there has been a wide variety of man-made pollutants produced in the 20th century, are more susceptible to contamination than deeper ground water. Information about ground-water age can be used to determine recharge rates and refine hydrologic models of ground-water systems (Reilly and others, 1994; Szabo and others, 1996) and thus to predict the contamination potential and estimate the time needed to flush contaminants through a ground-water system.
The 0- to 50-year time scale is particularly relevant to environmentally sensitive shallow ground-water systems. Prior to the late 1980s, however, there were no reliable means of dating ground water recharged during this time scale and, until recently, none of those methods were considered practical for use in establishing regional patterns. In the early 1990s, USGS scientists (Busenberg and Plummer, 1992) developed a method to date ground water on the basis of chlorofluorocarbon (CFC) content of the water that is practical, cost-effective, and applicable to most shallow ground-water systems.
Sampling for tracers
The feasibility of using CFCs as tracers of recent recharge and indicators of ground-water age was first recognized in the 1970s (see Plummer and Busenberg, 1997 and references therein). CFCs have been increasingly used in oceanic studies since the late 1970s as tracers of oceanic circulation, ventilation, and mixing processes. USGS scientists (Busenberg and Plummer, 1992) adapted analytical procedures developed by the oceanographic scientific community for ground-water studies and designed sampling equipment and procedures for collection and preservation of water samples in the field. Water samples for CFC analysis are now routinely collected from domestic, irrigation, monitoring, and municipal wells, and from springs. A closed path is established between the well or pump to a valve system that is used to fill glass ampoules with water, creating a headspace with CFC-free, ultra-pure nitrogen gas. The samples are then transported to the U.S. Nuclear Regulatory Commission-licensed USGS laboratory for analysis of CFC content by gas chromatography to a detection limit of about 0.3 picograms per kilogram (0.3 pg/kg) of water, which is equivalent to 0.3x10-12 grams per kilogram, or 0.3 parts per quadrillion.
Determining ground-water ages
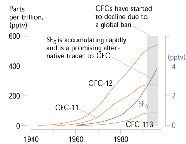 |
|
CFCs and sulfur hexafluoride (SF6) concentrations (mixing ratios) for air over North America during the last 50 years. |
Ground-water dating with CFC-11, CFC-12 and CFC-113 is possible because (1) their amounts in the atmosphere over the past 50 years have been reconstructed, (2) their solubilities in water are known, and (3) concentrations in air and young water are high enough that they can be measured. Age is determined from CFCs by relating their measured concentrations in ground water back to known historical atmospheric concentrations and/or to calculated concentrations expected in water in equilibrium with air. As with any environmental tracer, age applies to the date of introduction of the chemical substance into the water, and not to the water itself. The accuracy of the determined age depends in part on how perfectly the CFCs are transported with the water. Chemical processes, such as microbial degradation and sorption during transit, can also affect the concentration of CFCs and other compounds used in dating. For this reason, the term “age” is normally qualified with the word “model” or “apparent,” that is, “model age” or “apparent age.”
USGS scientists have investigated some of the most important factors that can affect CFC concentrations in ground water and the resulting age interpretations (Busenberg and Plummer, 1992; Plummer and others, 1993; Dunkle and others, 1993; Ekwurzel and others, 1994; Cook and others, 1995; Plummer and others 1998a, 1998b; Plummer and Busenberg, 1999). These include (1) uncertainty of the temperature at the water table during recharge, (2) entrapment of excess air during recharge, (3) uncertainty in recharge elevation, (4) thickness of the unsaturated zone, (5) effect of urban air where CFC values may exceed regional values, (6) contamination from a specific local source, (7) microbial degradation (in anaerobic environments), (8) sorption onto organic and mineral surfaces, and (9) mixing of younger and older water in a well. Because of the effect of these factors on CFC concentration, collection of additional data is often needed to determine the apparent age of ground water. For example, measurements of concentrations of dissolved gases, such as dissolved oxygen, help to define the potential for microbial degradation. Measurements of dissolved methane are useful in recognizing environments where all three CFCs can be degraded. Measurements of dissolved nitrogen and argon can be used to help determine recharge temperature and excess air and to recognize environments undergoing denitrification.
Other chemical dating tools
Tritium (half-life 12.4 years) provides another useful tracer of young ground water. Although it is difficult to evaluate age information from tritium data alone, age commonly can be reliably determined from data on tritium (3H) and its decay product, helium-3 (3He). The 3H/3He age is based on a calculation that determines the amount of 3He derived from radioactive decay of 3H in the water. Several conditions are necessary to solve the calculation and interpret the age: (1) The sample must contain detectable 3H (greater than approximately 0.5 tritium unit, or TU, which is defined as one 3H atom in 1018 hydrogen atoms) and (2) if the sample contains terrigenic helium from the Earth’s crust and mantle sources, the relative abundances of helium-3 and helium-4 isotopes in the terrigenic helium must be known, and data on dissolved neon concentrations in the sample are needed to help determine how much helium-3 is derived from tritium decay. Water samples for 3H/3He age determination can be collected more easily than those for CFC determination, but are more difficult and costly to analyze than CFC samples.
Krypton-85 (85Kr) has also been used to date ground water. The source for atmospheric input of 85Kr is reprocessing of fuel rods from nuclear reactors. Because of difficulties in collection and analysis, 85Kr is not yet a practical dating tool for ground-water studies. In CFC-contaminated environments, or environments where methane is forming from microbial degradation of organic matter, noble-gas dating techniques such as those based on 3H/3He and 85Kr measurements may be the only techniques for dating. However, because of their low detection limit and low sensitivity to hydrodynamic dispersion, CFCs can be more reliable than 3H/3He and 85Kr dating for waters recharged in the late 1940s and 1950s.
Seeking alternatives to CFC dating
Alternatives to using CFCs in dating ground water will clearly be needed as atmospheric CFC concentrations continue to fall. The use of sulfur hexafluoride (SF6) appears to be a promising technique and is under investigation by USGS scientists (Busenberg and Plummer, 1997; Plummer and Busenberg, 1999). Industrial production of SF6 began in 1953 with the introduction of gas-filled high-voltage electrical switches. SF6 is extremely stable and is accumulating rapidly in the atmosphere. The historical atmospheric mixing ratio of SF6 is being reconstructed from production records, archived air samples, and atmospheric measurements, and retrieved from concentrations measured in seawater and in previously-dated ground water. A preliminary reconstruction has been made of northern-hemisphere SF6 mixing ratios (figure far left). As atmospheric CFC concentrations fall, an even more sensitive dating tool will be the ratio of SF6 to, for example, CFC-12. Although SF6 is almost entirely of human origin, there is likely a natural, igneous source of SF6 that will complicate dating in some environments. USGS scientists have successfully used SF6 to date shallow ground water on the Delmarva Peninsula, Maryland, and water from springs in the Blue Ridge Mountains of Virginia with SF6.
Additional comments and case studies
For best results, the apparent age should be determined using multiple dating techniques because each dating technique has limitations. CFC dating is best suited for ground water in relatively rural environments where localized contamination, such as that from septic systems, sewage effluent, landfills, or urban runoff, can contain CFC concentrations in excess of that of atmospheric sources. The dating method appears to work well in shallow, aerobic, sand aquifers that are low in particulate organic matter and the results can be accurate within 2–3 years in this type of environment. Even where there are problems with CFC dating of ground water, the presence of CFCs indicates that the water sample contains at least some post-1940s water, making CFCs useful as tracers of recent recharge. Where CFC and 3H/3He ages agree, or where all three CFCs indicate similar ages, considerable confidence can be placed in the apparent age.
Delmarva Peninsula Results from the determination of CFCs in some agricultural areas on the Delmarva Peninsula of Maryland and Virginia indicate that water recharged since the early 1970s exceeds the U.S. Environmental Protection Agency drinking water Maximum Contaminant Level (MCL) for nitrate of 10 mg/L (as N), while water recharged prior to the early 1970s, before the heavy use of nitrogen fertilizers, does not exceed the MCL (Dunkle and others, 1992; Ekwurzel and others, 1994; Böhlke and Denver, 1995). Nitrate concentrations in ground water under woodlands were low, whereas ground water recharged under agricultural fields had nitrate concentrations that exceeded the MCL. CFC concentrations indicate that ground water now discharging to streams that drain agricultural areas of the Delmarva and then flow into the Chesapeake Bay or the Atlantic Ocean, was recharged in nearby fields in the 1960s and 1970s (Böhlke and Denver, 1995; Focazio and others, 1998). Thus, even if the application of nitrogen fertilizers to the fields stopped today, streams, rivers, and estuaries can be expected to receive increasing amounts of nitrate from ground-water discharge until the contaminated water is flushed through the system (Modica and others, 1998); up to 30 years may be needed to flush the high-nitrate water present in several small agricultural watersheds.
Central Oklahoma Water discharged from deep (400 to 800 feet) municipal supply wells in the central Oklahoma aquifer contains CFCs (Busenberg and Plummer, 1992). This unexpected finding indicated that shallow ground water was being mixed with deep, older water during pumping. In this case, CFCs were a valuable tracer for testing hydraulic seals of municipal water wells and their susceptibility to contamination. River water downstream from outfalls of sewage treatment plants in central Oklahoma contained very high concentrations of CFCs, which were also found in shallow ground water in the alluvium and terrace system near rivers.
Idaho At the Idaho National Engineering and Environmental Laboratory (INEEL), the ground-water CFC concentrations were found to be in equilibrium with the shallow soil air, rather than air from the deeper unsaturated zone (Busenberg and others, 1993). This indicates that these waters passed rapidly through the unsaturated zone to the water table, rather than recharging by slow percolation. Temperatures derived from gas solubilities in ground water suggest that the aquifer is recharged locally. Ground-water velocities on the order of 6 to 24 feet per day were found in the Snake River Plain aquifer beneath the INEEL. USGS scientists (Busenberg and others, 1998) have also used CFCs in water and unsaturated zone air to trace the movement of organic waste plumes at INEEL.
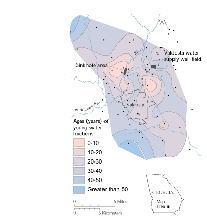 |
|
CFCs were used to trace and date water in the karst aquifer near Valdosta, Georgia. |
Nevada Measurements of CFCs in unsaturated-zone air along the crest of Yucca Mountain, Nevada, a potential site for a high-level radioactive waste repository, show that the residence time of shallow advecting gas is less than 5 years (Thorstenson and others, 1998). Previous estimates based on carbon-14 measurements of unsaturated-zone carbon dioxide indicated a residence time of less than 40 years. In addition to decreasing the estimated travel time by an order of magnitude, the CFC data allowed gas circulation patterns in the mountain to be identified and quantified. These results are highly relevant to issues pertaining to isolation of possible repository-generated radioactive 14CO2.
Georgia CFCs were used to trace and date leakage of river water through sinkholes into the Upper Floridan aquifer near Valdosta, Georgia (Plummer and others, 1998a, 1998b). The ages of the young-water fractions in the Upper Floridan aquifer were mapped throughout the Valdosta area. The dating indicates that few domestic and municipal supply wells produce river-water fractions that are younger than 5 years, and most river water sampled was likely recharged through the sinkholes during the past 20 to 30 years. Ground-water velocities ranged from 1 to 27 feet per day. Ground-water dating at Valdosta helps define the susceptibility to contamination of the ground-water resources.
REFERENCES
Böhlke, J.K., and Denver, J.M., 1995, Combined use of groundwater dating, chemical, and isotopic analyses to resolve the history and fate of nitrate contamination in two agricultural watersheds, Atlantic coastal plain, Maryland: Water Resources Research, v. 31, p. 2319–2339.
Busenberg, Eurybiades, and Plummer, L.N., 1992, Use of chlorofluorocarbons (CCl3F and CCl2F2) as hydrologic tracers and age—dating tools: Example–The alluvium and terrace system of central Oklahoma: Water Resources Research, v. 28, 2257–2284.
Busenberg, Eurybiades, and Plummer, L.N., 1997, Use of sulfur hexafluoride as a dating tool and as a tracer of igneous and volcanic fluids in ground water (abs.): Geological Society of America, Salt Lake City, 1997, Abstracts and Programs, v. 29(6), p. A-78.
Busenberg, Eurybiades, Plummer, L.N., Bartholomay, R.C., and Wayland, J.E., 1999, Chlorofluorocarbons, sulfur hexafluoride, and dissolved permanent gases in ground water from selected sites in and near the Idaho National Engineering and Environmental Laboratory, Idaho, 1994–97: U.S. Geological Survey Open-File Report 98-274, 72 p.
Busenberg, Eurybiades, Weeks, E.P., Plummer, L.N., and Bartholomay, R.C., 1993, Age dating ground water by use of chlorofluorocarbons (CCl3F and CCl2F2), and distribution of chlorofluorocarbons in the unsaturated zone, Snake River Plain aquifer, Idaho National Engineering Laboratory, Idaho: U.S. Geological Survey Water-Resources Investigations Report 93-4054, 47 p.
Cook, P.G., Solomon, D.K., Plummer, L.N., Busenberg, Eurybiades, and Schiff, S.L.,1995, Chlorofluorocarbons as tracers of groundwater transport processes in a shallow, silty sand aquifer: Water Resources Research, v. 31, p. 425–434.
Dunkle, S.A., Plummer, L.N., Busenberg, Eurybiades, Phillips, P.J., Denver, J.M., Hamilton, P.A., Michel, R.L., and Coplen, T.B., 1993, Chlorofluorocarbons (CCl3F and CCl2F2) as dating tools and hydrologic tracers in shallow ground water of the Delmarva Peninsula, Atlantic Coastal Plain, United States: Water Resources Research, v. 29, p. 3837–3860.
Ekwurzel, Brenda, Schlosser, Peter, Smethie, W., Jr., Plummer, L.N., Busenberg, Eurybiades, Michel, R.L., Weppernig, Ralf, and, Stute, M., 1994, Dating of shallow groundwater: Comparison of the transient tracers 3H/3He, chlorofluorocarbons and 85Kr: Water Resources Research, v. 30, p. 1693–1708.
Focazio, M.J., Plummer, L.N., Böhlke, J.K., Busenberg, Eurybiades, Bachman, L.J., and Powers, D.S., 1998, Preliminary estimates of residence times and apparent ages of ground water in the Chesapeake Bay watershed and water-quality data from a survey of springs: U.S. Geological Survey Water-Resources Investigations Report 97-4225, 75 p.
Modica, Edward, Buxton, H.T., and Plummer, L.N., 1998, Evaluating the source and residence times of ground-water seepage to headwaters streams, New Jersey Coastal Plain: Water Resources Res., v. 34, p. 2797–2810.
Plummer, L.N. and Busenberg Eurybiades, 1999, Chlorofluorocarbons in P. Cook and A. Herczeg, eds., Environmental Tracers in Subsurface Hydrology: Kluwer Academic Press, in press.
Plummer, L.N., McConnell, J.B., Busenberg, Eurybiades, Drenkard, Stefan, Schlosser, Peter, and Michel, R.L., 1998a, Flow of river water into a karstic limestone aquifer, 1. Tracing the young fraction in groundwater mixtures in the Upper Floridan aquifer near Valdosta, Georgia: Applied Geochemistry, v. 13, p. 995&150;1015.
Plummer, L.N., Busenberg, Eurybiades, Drenkard, Stefan, Schlosser, Peter, McConnell, J.B., Michel, R.L., Ekwurzel, Brenda, and Weppernig, Ralf, 1998b, Flow of river water into a karstic limestone aquifer, 2. Dating the young fraction in groundwater mixtures in the Upper Floridan aquifer near Valdosta, Georgia: Applied Geochemistry, v. 13, p. 1017–1043.
Plummer, L.N., Michel, R.L., Thurman, E.M., and Glynn, P.D., 1993, Environmental tracers for age-dating young ground water: Alley, W.M., ed., Regional Ground-water Quality: Van Nostrand Reinhold, p. 255–294.
Reilly, T.E., Plummer, L.N., Phillips, P.J., and Busenberg, Eurybiades 1994, Estimation and corroboration of shallow ground-water flow paths and travel times by environmental tracer and hydraulic analyses—A case study near Locust Grove, Maryland: Water Resources Research, v. 30, p. 421–433.
Szabo, Z., Rice, D.E., Plummer, L.N., Busenberg, Eurybiades, Drenkard, Stefan, and Schlosser, Peter, 1996, Age-dating of shallow groundwater with chlorofluorocarbons, tritium/helium 3, and flow path analysis, southern New Jersey coastal plain: Water Resources Research, v. 32, p. 1023–1038.
Thorstenson, D.C., Weeks, E.P., Haas, H., Busenberg, Eurybiades, Plummer, L.N., and Peters, C.A., 1998, Chemistry of unsaturated zone gases sampled in open boreholes at the crest of Yucca Mountain, Nevada: Data and basic concepts of chemical and physical processes in the mountain: Water Resources Research, v. 34, p.1507–1529.
For additional information,
see http://water.usgs.gov/lab/cfc/
or contact:
Niel Plummer (email: nplummer@usgs.gov)
U.S. Geological Survey 12201 Sunrise
Valley Drive,
MS 432
Reston, VA 20192