

Prepared as part of the NATIONAL WATER-QUALITY ASSESSMENT PROGRAM
Implications for Ground-Water Management
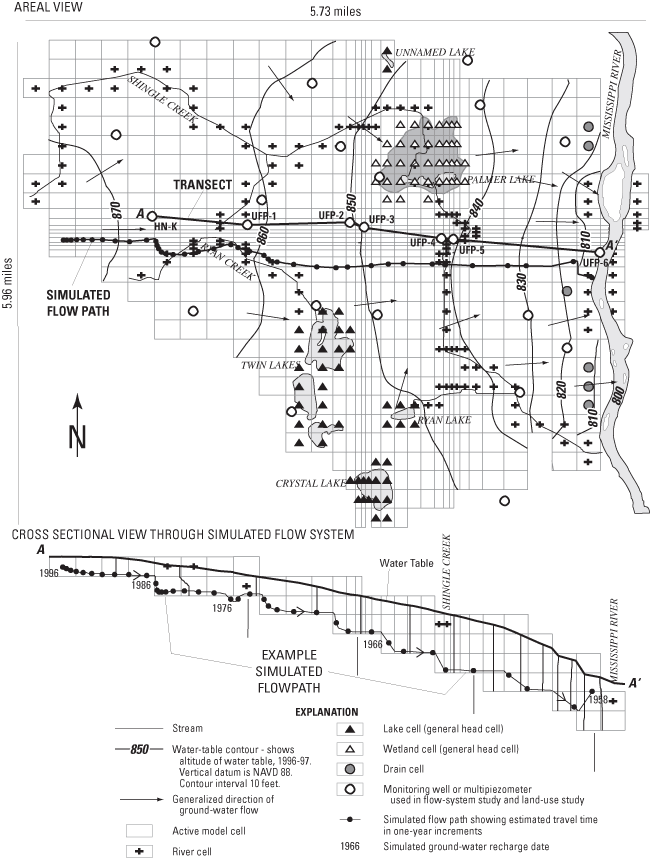
Figure 1. Location of the urban flow-system study area, surficial geology, ...
Figure 2. Dissolved-oxygen concentrations in water samples from the flow-sy...
Figure 3. Physical and chemical properties of water samples from the flow-s...
Figure 4. Specific conductance of water samples from the flow-system well a...
Figure 5. Major ion concentrations in water samples from the flow-system we...
Figure 6. Major ion concentrations in water samples from the flow-system we...
Figure 7. Chloride concentrations in water samples from the flow-system wel...
Figure 8. Nutrient concentrations in water samples from the flow-system wel...
Figure 9. Dissolved nitrite plus nitrate as nitrogen concentrations in wate...
Figure 10. Dissolved ammonia plus organic nitrogen concentrations in water ...
Figure 11. Nitrogen and argon gas concentrations in water samples from the ...
Figure 12. Correlation between d2H and d180 in water samples from the flow-...
Figure 13. Total detected dissolved pesticide concentrations in water sampl...
Figure 14. Frequencies of detection (in percent) of pesticides in water sam...
Figure 15. Total detected volatile organic compound concentrations in water...
Figure 16. Frequencies of detection (in percent) of volatile organic compou...
Figure 17. Schematic of model configuration, simulated water-table altitude...
Figure 18. Recharge-age dates of water samples from the flow-system well an...
Figure 19. Relations of selected dates of recharge, as determined by chloro...
Table 1. Laboratory analytical methods for measured water-quality constitue...
Table 2. Pesticide compounds analyzed in water samples for flow-system stud...
Table 3. Volatile organic compounds analyzed in water samples for flow-syst...
Table 4. Estimated horizontal hydraulic conductivity (K) values calculated ...
Table 5. Sampling depths, depths to water table, sediment pH, and organic a...
Table 6. Hydraulic properties of the modeled study area, Twin Cities metrop...
Table 7. Recharge-age dates and model estimates for ground water sampled fr...
Table 8. Laboratory reporting levels and method detection limits and concen...
Table 9. Laboratory reporting levels, method detection limits, and concentr...
Table 10. Recovery percents from Schedule 2010 pesticide spikes for multipi...
Table 11. Recovery percents from Schedule 2020 volatile organic compound sp...
Table 12. Laboratory reporting levels and concentrations of compounds with ...
1U.S Geological Survey
2City of Minneapolis, Environmental Management
The U.S. Geological Survey (USGS) is committed to serve the Nation with accurate and timely scientific information that helps enhance and protect the overall quality of life, and facilitates effective management of water, biological, energy, and mineral resources. (http://www.usgs.gov/). Information on the quality of the Nation's water resources is of critical interest to the USGS because it is so integrally linked to the long-term availability of water that is clean and safe for drinking and recreation and that is suitable for industry, irrigation, and habitat for fish and wildlife. Escalating population growth and increasing demands for the multiple water uses make water availability, now measured in terms of quantity and quality, even more critical to the long-term sustainability of our communities and ecosystems.
The USGS implemented the National Water-Quality Assessment (NAWQA) Program to support national, regional, and local information needs and decisions related to water-quality management and policy. (http://water.usgs.gov/nawqa/). Shaped by and coordinated with ongoing efforts of other Federal, State, and local agencies, the NAWQA Program is designed to answer: What is the condition of our Nation's streams and ground water? How are the conditions changing over time? How do natural features and human activities affect the quality of streams and ground water, and where are those effects most pronounced? By combining information on water chemistry, physical characteristics, stream habitat, and aquatic life, the NAWQA Program aims to provide science-based insights for current and emerging water issues and priorities. NAWQA results can contribute to informed decisions that result in practical and effective water-resource management and strategies that protect and restore water quality.
Since 1991, the NAWQA Program implemented interdisciplinary assessments in more than 50 of the Nation's most important river basins and aquifers, referred to as Study Units. (http://water.usgs.gov/nawqa/nawqamap.html). Collectively, these Study Units account for more than 60 percent of the overall water use and population served by public water supply, and are representative of the Nation's major hydrologic landscapes, priority ecological resources, and agricultural, urban, and natural sources of contamination.
Each assessment is guided by a nationally consistent study design and methods of sampling and analysis. The assessments thereby build local knowledge about water-quality issues and trends in a particular stream or aquifer while providing an understanding of how and why water quality varies regionally and nationally. The consistent, multi-scale approach helps to determine if certain types of water-quality issues are isolated or pervasive, and allows direct comparisons of how human activities and natural processes affect water quality and ecological health in the Nation's diverse geographic and environmental settings. Comprehensive assessments on pesticides, nutrients, volatile organic compounds, trace metals, and aquatic ecology are developed at the national scale through comparative analysis of the Study-Unit findings. (http://water.usgs.gov/nawqa/natsyn.html).
The USGS places high value on the communication and dissemination of credible, timely, and relevant science so that the most recent and available knowledge about water resources can be applied in management and policy decisions. We hope this NAWQA publication will provide you the needed insights and information to meet your needs, and thereby foster increased awareness and involvement in the protection and restoration of our Nation's waters.
The NAWQA Program recognizes that a national assessment by a single program cannot address all water-resource issues of interest. External coordination at all levels is critical for a fully integrated understanding of watersheds and for cost-effective management, regulation, and conservation of our Nation's water resources. The Program, therefore, depends extensively on the advice, cooperation, and information from other Federal, State, interstate, Tribal, and local agencies, non-government organizations, industry, academia, and other stakeholder groups. The assistance and suggestions of all are greatly appreciated.
Robert M. Hirsch Associate Director for Water
| Multiply | By | To obtain |
|---|---|---|
| Length | ||
| inch (in.) | 2.54 | centimeter (cm) |
| foot (ft) | 0.3048 | meter (m) |
| Area | ||
| square mile (mi2) | 2.590 | square kilometer (km2) |
| Volume | ||
| million gallons (Mgal) | 3,785 | cubic meter (m3) |
| Flow rate | ||
| foot per day (ft/d) | 0.3048 | meter per day (m/d) |
| cubic foot per second (ft3/s) | 0.02832 | cubic meter per second (m3/s) |
| gallon per day (gal/d) | 0.003785 | cubic meter per day (m3/d) |
| million gallons per day (Mgal/d) | 0.04381 | cubic meter per second (m3/s) |
| Mass | ||
| pound, avoirdupois (lb) | 0.4536 | kilogram (kg) |
Temperature in degrees Celsius (°C) may be converted to degrees Fahrenheit (°F) as follows:
°F=(1.8x°C)+32
Temperature in degrees Fahrenheit (°F) may be converted to degrees Celsius (°C) as follows:
°C=(°F-32)/1.8
Chemical concentrations: Chemical concentrations of substances in water are given in metric units of milligrams per liter (mg/L) and micrograms per liter (µg/L). Milligrams per liter is a unit expressing the concentration of chemical constituents in solution as mass (milligrams) of solute per unit volume (liter) of water. Micrograms per liter is a unit expressing the concentration of chemical constituents in solution as mass (micrograms) of solute per unit volume (liter) of water. One milligram is equivalent to 1,000 micrograms.
Vertical coordinate information is referenced to the North American Vertical Datum of 1988 (NAVD 88).
Altitude, as used in this report, refers to distance above the vertical datum.
Specific conductance is given in microsiemens per centimeter at 25 degrees Celsius (µS/cm at 25°C).
As part of a national analysis of the effects of land use on ground-water quality for the National Water-Quality Assessment Program, the U.S. Geological Survey sampled wells along a flow system in surficial glacial aquifers in the northwestern part of the Twin Cities metropolitan area during 1997 and 1998. In addition, a reconnaissance steady-state ground-water model was developed to estimate flowpaths and dates of ground-water recharge using a particle-tracking routine.
Sediment samples collected during drilling had high horizontal hydraulic conductivities (ranging from about 3.1 to about 190 feet per day, based on sediment-size analysis of well cuttings), small organic carbon concentrations (ranging from less than 0.2 to 160 grams per kilogram), and pH values that were mostly alkaline (ranging from 4.9 to 8.2).
Water samples were analyzed for physical properties, major ions, iron, manganese, nutrients, organic carbon, radon, pesticides, volatile organic compounds (VOCs), chlorofluorocarbons, tritium, and isotopes of nitrogen, hydrogen, and oxygen. Most of the water samples had small dissolved-oxygen concentrations (less than 1 milligram per liter). Calcium, magnesium, sodium, bicarbonate, sulfate, and chloride were the primary dissolved constituents in water samples. Nitrite plus nitrate as nitrogen (nitrate) concentrations were less than the U.S. Environmental Protection Agency (USEPA) Maximum Contaminant Level of 10 mg/L. Nitrogen isotope ratios indicated that the sources of nitrate primarily were soils, animal waste, or denitrification that increased nitrogen isotope ratios in nitrate.
Small concentrations of pesticides were detected in the shallow parts of the aquifer. The herbicide prometon was the most frequently detected pesticide. Herbicides applied to control grasses and weeds in corn (atrazine, simazine, and metolachlor) also were frequently detected in water samples. All pesticide and VOCs detected were below USEPA Maximum Contaminant Levels or Health Advisory Limits. Chlorofluorocarbon compounds and tritium concentrations were used to estimate dates of recharge of ground-water samples. In general, shallower ground-water samples were more recently recharged although most water sampled from the aquifer was recharged after 1955.
Although land use had substantial effects on ground-water quality, the distribution of contaminants in the aquifer also is affected by complex combinations of factors and processes that include sources of natural and anthropogenic contaminants, three-dimensional advective flow, physical and hydrologic settings, age and evolution of ground water, and transformation of chemical compounds along the flow system. Compounds such as nitrate and dissolved oxygen were greatest in water samples from the upgradient end of the flow system and near the water table. Specific conductance and dissolved solids increased along the flow system and with depth due to increase in residence time in the flow system and dissolution of aquifer materials.
In 1991, the U.S. Geological Survey (USGS) began implementation of the National Water-Quality Assessment (NAWQA) Program. Long-term goals of NAWQA include describing the status and trends in the quality of large representative parts of the Nation's surface- and ground-water resources, and identifying the major natural and anthropogenic factors that affect the quality of the Nation's water resources. To meet these goals, nationally consistent data useful to policy makers, scientists, and managers are being collected and analyzed. Because assessment of the water quality in the entire Nation is impractical, major activities of the NAWQA Program take place within a set of hydrologic systems called Study Units, which contain diverse hydrologic systems of river basins or aquifers.
The Upper Mississippi River Basin Study (UMIS) Unit (fig. 1) encompasses about 47,000 mi2 in Minnesota, Wisconsin, South Dakota, Iowa, and North Dakota, and includes the Twin Cities (Minneapolis and St. Paul, Minn.) metropolitan area (TCMA). The three major rivers in the UMIS are the Mississippi, Minnesota, and St. Croix. In 1990, about 3.7 million people resided in the UMIS, mostly in the TCMA (U.S. Bureau of Census, 1991). Most ground-water withdrawals in the TCMA are from surficial glacial aquifers or from the Prairie du Chien-Jordan bedrock aquifer.
Study-unit activities involving ground water included assessment of ground water and land use combinations using three sampling strategies: (1) regional studies of selected major aquifers (subunit surveys), (2) targeted-area studies of selected land-use areas (land-use studies), and (3) this localized study of processes occurring along a shallow ground-water-flow system (flow-system study). This study emphasizes shallow ground water, the quality of which is likely to be influenced by land use and land cover.
The flow-system study described in this report is within the area of an urban land-use study conducted in 1996 (Andrews and others, 1998). The urban land-use study sampled shallow wells completed in surficial (glacial) aquifers. This flow-system study was designed to obtain information about ground-water flow and chemical processes occurring throughout the thickness of the surficial aquifers. This study had four objectives: (1) determine the presence, spatial distribution, and temporal variability of selected constituents in ground water along a general flow system from areas of recharge to discharge; (2) relate the presence and distribution of selected constituents in ground water to land use and physical factors; (3) develop an understanding of the transport of the selected constituents from land surface through the flow system; and (4) provide an estimate of the time needed for dissolved constituents to move through the flow system.
This report summarizes hydrologic and water-quality data collected in 1997 and 1998 from one existing monitoring well and six multiport samplers (multipiezometers) installed along a transect about parallel to ground-water flow in Brooklyn Park and Brooklyn Center, Minnesota. Collected data characterize spatial changes in ground-water quality along the flow system within the urban land-use study area described in Andrews and others (1998). Water-quality data are presented for physical properties, major ions, iron, manganese, nutrients, organic carbon, radon, pesticides, volatile organic compounds, and isotopes of nitrogen, hydrogen and oxygen. Data also are presented for chlorofluorocarbons and tritium, which were used to estimate the age of recharge water. Results of a reconnaissance steady-state, ground-water-flow model used to estimate flowpaths and dates of ground-water recharge, using a particle-tracking routine, also are presented.
The authors express appreciation to the following people and agencies for allowing the installation of monitoring wells on their properties and for their interest in the project: Mr. Steve Shelstead of the Anoka-Hennepin Independent School District #279 and Mr. Scott Brink of the City of Brooklyn Center Engineering Department. The authors also thank Dr. James Piegat and Ms. Leigh Harrod, formerly of the Hennepin County Conservation District, for their assistance in sampling and for evaluating land uses, ages of development, geology, and significant local features of the study area. Appreciation is extended to Larry Puckett of the U.S. Geological Survey who assisted with sampling. Dr. Mark Pearson and Emily Youcha, formerly of the University of Minnesota; Alan Burns, Timothy Cowdery, and Geoffrey Delin of the U.S. Geological Survey who assisted in the preparation of the ground-water-flow model. Paul Hansen, formerly of the U.S. Geological Survey, and Robert Borgstede of the U.S. Geological Survey for assistance with the preparation of illustrations. Ginger Amos, formerly of the U.S. Geological Survey, helped with editorial suggestions and direction.
The study area for this report (fig. 1) is located in the northwestern part of the TCMA. The area consists primarily of suburban residential and commercial development. Shingle Creek and several major roadways cross the flow system.
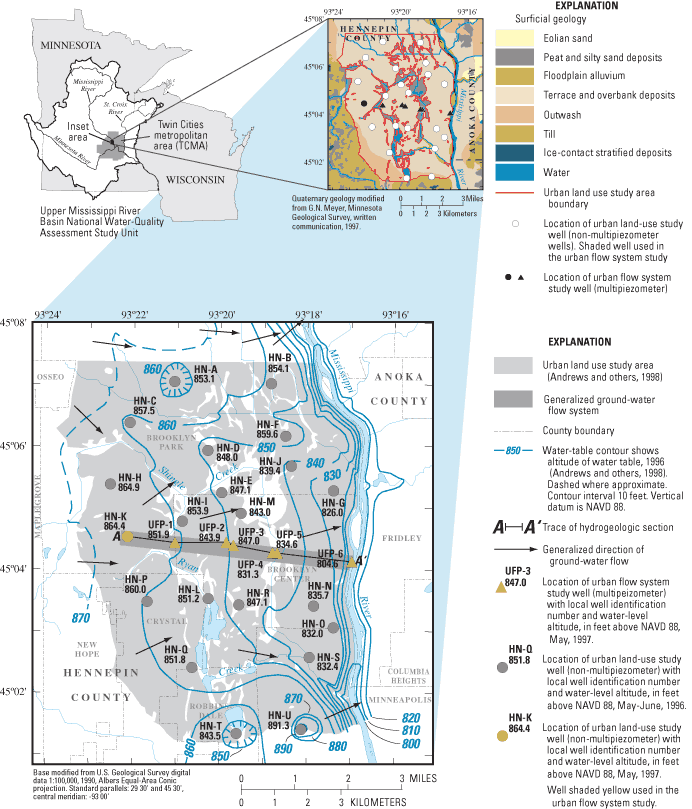
Population density in the study area during 1995 was about 1,530 people per mi2 (Metropolitan Council, 2003). Public water supplies included 12.6 Mgal/d with greater than 90 percent of withdrawals from ground water. The cities of Brooklyn Center and Brooklyn Park withdraw ground water from wells completed in the surficial aquifers composed of sand and gravel terrace deposits and in underlying bedrock aquifers. In addition to municipal wells, there are approximately 200 low-capacity domestic or lawn-irrigation wells completed in surficial aquifers in the study area that pump an estimated 15,000 gal/d during summer months (James Piegat, Hennepin Conservation District, written commun., 1997).
Land use and land cover can affect the presence of contaminants in ground water. Fertilizers and pesticides applied to lawns can leach to ground water. Roadways and railroads are sources of sodium and chloride from de-icing salt. Pesticides and volatile organic compounds (VOCs) emitted to the atmosphere and applied to the land surface also are potential contaminants. Commercial and industrial establishments may also discharge VOCs, metals, and other substances to the atmosphere or to the land surface in the course of their activities.
Average monthly temperatures range from 11 degrees Fahrenheit in January to 74 degrees Fahrenheit in July (Minnesota State Climatologist, electronic commun., 1995). Average annual precipitation is between 28 and 30 inches (Minnesota State Climatologist, 1995). About three-fourths of annual precipitation falls from May through September (Baker and others, 1979). Mean annual evaporation is approximately 36 inches (Farnsworth and others, 1982). Seasonal fluctuations in temperature and rainfall can affect solubilities of VOCs in rainfall, volatilization of VOCs to the atmosphere, and seasonal loadings of pesticides in rainfall.
Hydrologic and water-quality data for this report were from one existing monitoring well and six multiport samplers (multipiezometers). Collected data characterized ground-water hydrology and water quality along the flow system. A reconnaissance steady-state ground-water flow-model was constructed to estimate flowpaths and dates of ground-water recharge using a particle tracking routine.
A transect of sites along the ground-water-flow system including an existing well (HN-K) and multipiezometers samplers (UFP-1 to UFP-6) (fig. 1) was selected on the basis of estimated ground-water flow directions and the location of streams and wetlands. Multipiezometer samplers (Delin and Landon, 1996) were constructed of 0.167 ft inside-diameter (ID) by 10-ft long schedule 40 polyvinyl chloride (PVC) casings with 3.12 x 10-3 ft diameter stainless steel sampling tubes. The annular space between the casing and the borehole was packed with sand. In cases where natural sand collapse did not completely fill the borehole surrounding the open interval, commercially prepared washed sand was added to a level approximately 1 ft above the top of the ports. The remaining annular spaces were grouted with bentonite slurry to within 2 ft of the land surface. The top 2 ft of the boreholes were grouted with concrete. One-half foot ID, schedule 40 steel protective casings, with locking aluminium caps, were set into mortar to a depth of 4 ft, and extended 2 ft above land surface. All wells and multipiezometers were constructed in accordance with Minnesota Department of Health regulations (Minnesota Department of Health, 1994) and NAWQA specifications (Lapham and others, 1995). Multipiezometer ports were developed by surging with a peristaltic pump. Sampling ports were designated as follows: (1) all multipiezometer wells were denoted as "UFP-X" with "X" ranging from 1 downgradient from well HN-K to 6 at the downgradient end of the flow system, near the Mississippi River. At each multipiezometer, individual ports were designated with letters A-D. "A" ports were completed above the water table for possible sampling of gases in the unsaturated zone, "B" ports were completed just below the water table, "C" ports were approximately equidistant between the water table and the bottom of the aquifer, and "D" ports were near the bottom of the aquifer. Water levels were measured before water samples were collected using the wetted-tape method.
Sediment samples from the unsaturated zone, and from the first few feet of the saturated zone, were collected during well drilling with split-spoon samplers. Sediment was analyzed for pH, sediment-size distribution, and organic-carbon content. Sediment-size distributions were analyzed by sieving (Guy, 1969) for sandy samples and by pipet analysis (Guy, 1969) for fine-grained samples at the USGS Sediment Laboratory in Iowa City, Iowa. Organic and inorganic carbon contents of sediment were analyzed by induction furnace and a modification of the Van Slyke Method (Wershaw and others, 1987) at the USGS National Water-Quality Laboratory in Lakewood, Colorado. Sediment sample pH values were measured in the field by mixing samples with deionized water on a 1:1 weight basis and measuring the pH of the resulting solution.
Hydraulic conductivity values were estimated from sediment-size distributions using Hazen's equation (Freeze and Cherry, 1979):
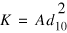 (1)
(1)
where K is hydraulic conductivity in centimeters per second (cm/s), A equals 1.0 centimeter per second per square millimeter (cm/s/mm2) and d10 is the sediment-size diameter (in millimeters) at which 10 percent of the particles by weight are finer and 90 percent are coarser.
Physical properties of specific conductance, pH, water temperature, turbidity, dissolved oxygen, and alkalinity, as well as air temperature and barometric pressure, were measured in the field with instruments calibrated with a mercury manometer and standard solutions (obtained commercially for pH and from the USGS Quality of Water Service Unit in Ocala, Florida for specific conductance and dissolved oxygen). An equivalent of five standing volumes of water in each access tube was withdrawn through the ports, and stability of water chemistry was verified through periodic measurements of properties made while purging the ports prior to sampling. Water samples were collected during July 1997, October 1997, and August 1998, using a sealed system of viton or nylon tubing according to protocols described in Koterba and others (1995). Water samples were collected from most of the 16 sampling ports once during each of the three sample periods. Water samples were shipped overnight to the USGS National Water-Quality Laboratory in Lakewood, Colorado where analyses were done according to USGS protocols for analysis, quality assurance, and quality control (table 1). Water-quality results are available in the USGS National Water Information System (NWIS) at http://water.usgs.gov/nwis. Quality-assurance sample data are listed and described in the appendix.
Samples were analyzed for a variety of constituents. These included major ions, nutrients, selected pesticides, and VOCs. The groups of constituents analyzed are listed in table 1. The water-soluble pesticides analyzed are shown in table 2. The VOCs analyzed are shown in table 3.
Samples analyzed for chlorofluorocarbons (CFCs) CFC-11, CFC-12, CFC-l3, and CFC-113 were collected using sampling methods and protocols described by Busenberg and Plummer (1992). Analyses were by electron-capture gas chromatography, with a detection limit of 0.3 picogram per kilogram of water (pg/kg) for CFC-11 and CFC-12, and 1.0 pg/kg for CFC-113. Sample ages were assigned based on comparison of CFC equilibrium partial pressures, corrected for recharge temperature, with a chronology of atmospheric partial pressures (Thompson and Hays, 1979). Dissolved-gas analyses, used to determine redox states of water samples, were made using gas chromatographic methods.
Minimum laboratory reporting limits used in this report are based on the Method Detection Limit (MDL) and the Laboratory Reporting Level (LRL). The MDL is the minimum concentration of a substance that can be measured and reported with 99-percent confidence that the analyte concentration is greater than zero. It is determined from the analysis of a sample in a given matrix containing the analyte. At the MDL concentration, the risk of a false positive is predicted to be less than or equal to 1 percent. The LRL is generally equal to twice the yearly determined Long-Term Method Detection Level (LT-MDL). The LT-MDL is derived by determining the standard deviation of a minimum of 24 MDL spike sample measurements over an extended period of time. LT-MDL data are collected on a continuous basis to assess year-to-year variations in the LT-MDL. The LT-MDL controls false positive error. The chance of falsely reporting a concentration at or greater than the LT-MDL for a sample that does not contain the analyte is predicted to be less than or equal to 1 percent. These definitions can be accessed at URL http://nwql.usgs.gov/Public/ltmdl/glossary.html.
Comparisons of water-quality data were made using the nonparametric Kruskal-Wallis test, a statistical test which compares groups of data based on data ranks, and which is well suited to data that are normally distributed (Helsel and Hirsch, 1992; Miller, 1985) and because sample sizes were small and variable. For comparisons between categories, multiple stage tests were performed on Kruskal-Wallis statistics (Helsel and Hirsch, 1992). Results were considered statistically significant between p <0.05 during the first step of the test, and p <0.025 for the final step between any two categories (Helsel and Hirsch, 1992).
[USGS, U.S. Geological Survey]
| Constituent or constituent group | Reference |
|---|---|
| Major ions (USGS schedule 2750) | Fishman and Friedman (1989) |
| Nutrients (USGS schedule 2752) | Fishman and Friedman (1989) |
| Dissolved organic carbon (USGS schedule 2085) | Brenton and Arnett (1993) |
| Nitrogen isotopes (USGS procedure 1718) | Bremner (1965), Bremner and Edwards (1965), Liu (1979), and Minagawa and others (1984) |
| Hydrogen and oxygen isotopes in water (USGS labcode 489) | Coplen and others (1991), and Epstein and Mayeda (1953) |
| Radon (USGS labcode 1369) | American Society for Testing and Materials (2002) |
| Pesticides (USGS schedule 2010) | Zaugg and others (1995) |
| Volatile organic compounds (USGS custom method 9090) | Rose and Schroeder (1995) |
| Chlorofluorocarbons | Busenberg and Plummer (1992) |
| Dissolved gases | Busenberg andPlummer (1992) |
| Tritium (USGS labcode 1565) | Ostlund and Dorsey (1975) |
[All units in micrograms per liter; CAS, Chemical Abstract Service]
| Compound | CAS Number | Laboratory reporting level |
|---|---|---|
| alpha-HCH | 319-84-6 | 0.0046 |
| Acetochlor | 34256-82-1 | .006 |
| Alachlor | 15972-60-8 | .0045 |
| 2,6-Diethylaniline | 579-66-8 | .006 |
| Atrazine | 1912-24-9 | .007 |
| Deethylatrazine | 6190-65-4 | .006 |
| Azinphos-methyl | 86-50-0 | .05 |
| Benfluralin | 1861-40-1 | .010 |
| Butylate | 2008-41-5 | .002 |
| Carbaryl | 63-25-2 | .041 |
| Carbofuran | 1563-66-2 | .020 |
| Chlorpyrifos | 2921-88-2 | .005 |
| cis-Permethrin | 54774-45-7 | .006 |
| Cyanazine | 21725-46-2 | .018 |
| Dacthal | 1861-32-1 | .0030 |
| Diazinon | 333-41-5 | .005 |
| Dieldrin | 60-57-1 | .0048 |
| Disulfoton | 298-04-4 | .021 |
| EPTC | 759-94-4 | .0020 |
| Ethalfluralin | 55283-68-6 | .009 |
| Ethoprophos | 13194-48-4 | .005 |
| Fonofos | 944-22-9 | .0027 |
| Lindane | 58-89-9 | .0040 |
| Linuron | 330-55-2 | 0.035 |
| Malathion | 121-75-5 | .027 |
| Parathion-methyl | 298-00-0 | .006 |
| Metolachlor | 51218-45-2 | .013 |
| Metribuzin | 21087-64-9 | .006 |
| Molinate | 2212-67-1 | .0016 |
| Napropamide | 15299-99-7 | .007 |
| p,p'-DDE | 72-55-9 | .0025 |
| Parathion | 56-38-2 | .010 |
| Pebulate | 1114-71-2 | .0041 |
| Pendimethalin | 40487-42-1 | .022 |
| Phorate | 298-02-2 | .011 |
| Prometon | 1610-18-0 | .015 |
| Propachlor | 1918-16-7 | .010 |
| Propanil | 709-98-8 | .011 |
| Propargite | 2312-35-8 | .023 |
| Simazine | 122-34-9 | .005 |
| Tebuthiuron | 34014-18-1 | .016 |
| Terbacil | 5902-51-2 | .034 |
| Terbufos | 13071-79-9 | .017 |
| Thiobencarb | 28249-77-6 | .0048 |
| Triallate | 2303-17-5 | .0023 |
| Trifluralin | 1582-09-8 | .009 |
[µg/L, micrograms per liter; pct, percent; CAS, Chemical Abstract Service; --, none]
| Compound | CAS Number | Laboratory reporting level | Unit |
|---|---|---|---|
| 1,1,1,2-Tetrachloroethane | 630-20-6 | 0.03 | µg/L |
| 1,1,1-Trichloroethane | 71-55-6 | .032 | µg/L |
| 1,1,2,2-Tetrachloroethane | 79-34-5 | .09 | µg/L |
| 1,1,2-Trichloroethane | 79-00-5 | .064 | µg/L |
| 1,1,2-Trichlorotrifluoroethane | 76-13-1 | .06 | µg/L |
| 1,1-Dichloroethane | 75-34-3 | .035 | µg/L |
| 1,1-Dichloroethylene | 75-35-4 | .044 | µg/L |
| 1,1-Dichloropropene | 563-58-6 | .05 | µg/L |
| 1,2,3,4-Tetramethylbenzene | 488-23-3 | .23 | µg/L |
| 1,2,3,5-Tetramethylbenzene | 527-53-7 | .2 | µg/L |
| 1,2,3-Trichlorobenzene | 87-61-6 | .27 | µg/L |
| 1,2,3-Trichloropropane | 96-18-4 | .16 | µg/L |
| 1,2,3-Trimethylbenzene | 526-73-8 | .12 | µg/L |
| 1,2,4-Trichlorobenzene | 120-82-1 | .12 | µg/L |
| 1,2,4-Trimethylbenzene | 95-63-6 | .056 | µg/L |
| 1,2-Dibromo-3-chloropropane | 96-12-8 | .5 | µg/L |
| 1,2-Dibromoethane | 106-93-4 | .036 | µg/L |
| 1,2-Dichlorobenzene | 95-50-1 | .048 | µg/L |
| 1,2-Dichloroethane | 107-06-2 | .13 | µg/L |
| 1,2-Dichloroethane-d4 | 17060-07-0 | .1 | pct |
| 1,2-Dichloropropane | 78-87-5 | .029 | µg/L |
| 1,3,5-Trimethylbenzene | 108-67-8 | .044 | µg/L |
| 1,3-Dichlorobenzene | 541-73-1 | .030 | µg/L |
| 1,3-Dichloropropane | 142-28-9 | .12 | µg/L |
| 1,4-Bromofluorobenzene | 460-00-4 | .1 | pct |
| 1,4-Dichlorobenzene | 106-46-7 | .050 | µg/L |
| 2,2-Dichloropropane | 594-20-7 | .05 | µg/L |
| 2-Butanone | 78-93-3 | 5.0 | µg/L |
| 2-Chlorotoluene | 95-49-8 | .04 | µg/L |
| 2-Hexanone | 591-78-6 | .7 | µg/L |
| 3-Chloropropene | 107-05-1 | .12 | µg/L |
| 4-Chlorotoluene | 106-43-4 | .05 | µg/L |
| 4-Isopropyl-1-methylbenzene | 99-87-6 | .12 | µg/L |
| 4-Methyl-2-pentanone | 108-10-1 | .37 | µg/L |
| Acetone | 67-64-1 | 7.0 | µg/L |
| Acrylonitrile | 107-13-1 | 1.2 | µg/L |
| Benzene | 71-43-2 | .021 | µg/L |
| Bromobenzene | 108-86-1 | .036 | µg/L |
| Bromochloromethane | 74-97-5 | .12 | µg/L |
| Bromodichloromethane | 75-27-4 | 0.048 | µg/L |
| Bromoethene | 593-60-2 | .10 | µg/L |
| Bromoform | 75-25-2 | .10 | µg/L |
| Bromomethane | 74-83-9 | .26 | µg/L |
| n-Butylbenzene | 104-51-8 | .19 | µg/L |
| Carbon disulfide | 75-15-0 | .07 | µg/L |
| Chlorobenzene | 108-90-7 | .028 | µg/L |
| Chloroethane | 75-00-3 | .12 | µg/L |
| Chloroform | 67-66-3 | .024 | µg/L |
| Chloromethane | 74-87-3 | .17 | µg/L |
| cis-1,2-Dichloroethylene | 156-59-2 | .038 | µg/L |
| cis-1,3-Dichloropropene | 10061-01-5 | .09 | µg/L |
| Dibromochloromethane | 124-48-1 | .18 | µg/L |
| Dibromomethane | 74-95-3 | .050 | µg/L |
| Dichlorodifluoromethane | 75-71-8 | .18 | µg/L |
| Dichloromethane | 75-09-2 | .16 | µg/L |
| Diethyl ether | 60-29-7 | .17 | µg/L |
| Diisopropyl ether | 108-20-3 | .10 | µg/L |
| Ethyl methacrylate | 97-63-2 | .18 | µg/L |
| Ethyl tert-butyl ether | 637-92-3 | .05 | µg/L |
| Ethylbenzene | 100-41-4 | .03 | µg/L |
| Hexachlorobutadiene | 87-68-3 | .14 | µg/L |
| Hexachloroethane | 67-72-1 | .19 | µg/L |
| Isopropylbenzene | 98-82-8 | .06 | µg/L |
| m- and p-Xylene | -- | .06 | µg/L |
| Methyl acrylate | 96-33-3 | 2.0 | µg/L |
| Methyl acrylonitrile | 126-98-7 | .57 | µg/L |
| Methyl iodide | 74-88-4 | .35 | µg/L |
| Methyl methacrylate | 80-62-6 | .35 | µg/L |
| Naphthalene | 91-20-3 | .5 | µg/L |
| n-Propylbenzene | 103-65-1 | .042 | µg/L |
| ortho-Ethyl toluene | 611-14-3 | .06 | µg/L |
| ortho-Xylene | 95-47-6 | .07 | µg/L |
| sec-Butylbenzene | 135-98-8 | .06 | µg/L |
| Styrene | 100-42-5 | .042 | µg/L |
| tert-Butyl methyl ether | 1634-04-4 | .17 | µg/L |
| tert-Butylbenzene | 98-06-6 | .10 | µg/L |
| tert-Amyl methyl ether | 994-05-8 | .08 | µg/L |
| Tetrachloroethylene | 127-18-4 | .027 | µg/L |
| Tetrachloromethane | 56-23-5 | .060 | µg/L |
| Tetrahydrofuran | 109-99-9 | 2.2 | µg/L |
| Toluene | 108-88-3 | .05 | µg/L |
| Toluene-d8 | 2037-26-5 | .1 | pct |
| trans-1,2-Dichloroethylene | 156-60-5 | .032 | µg/L |
| trans-1,3-Dichloropropene | 10061-02-6 | .09 | µg/L |
| trans-1,4-Dichloro-2-butene | 110-57-6 | .70 | µg/L |
| Trichloroethylene | 79-01-6 | .038 | µg/L |
| Trichlorofluoromethane | 75-69-4 | .09 | µg/L |
| Vinyl chloride | 75-01-4 | .11 | µg/L |
A reconnaissance steady-state, ground-water-flow model was used to simulate the flow system and to aid in understanding ground-water flow. The finite-difference program MODFLOW (McDonald and Harbaugh, 1988) was used with Ground Water Modeling System (GMS) software as the graphical user interface. MODPATH, a post-processing program developed for MODFLOW, was used to estimate flowpaths and dates of ground-water recharge using the particle- tracking routine (Pollock, 1994). A three-dimensional, conceptual, variable-spaced, cell-centered, finite-difference model grid was developed to emulate hydrologic conditions. Cell dimensions were varied to allow for the discretization needed along the flow system, in rivers, and in areas of low permeability. Construction of the model assumed equilibrium or steady-state conditions. Data collected for this study and existing data (Norvitch and others, 1978; Palen and others, 1989) were used to prepare input files of hydrologic unit thickness, hydraulic properties, and to identify inactive cells of low permeability. Hydrographic data and water levels from existing wells were used to estimate hydraulic head. Boundary flux general-head cells, river cells, and drain cells were used to simulate internal and external boundary conditions. Flow between surficial aquifers and bedrock aquifers was considered negligible (Schoenberg, 1990). Drain cells represented springs and seeps, river cells represented rivers, and general-head cells represented lakes and wetlands. General-head boundaries were used to simulate subsurface flow at the margins of the model. No-flow boundaries were used to simulate the limits of the modeled area where ground-water flow was assumed to be nonexistent or insignificant (McDonald and Harbaugh, 1988). A no-flow boundary at the base of the model represented glacial till overlying bedrock. Calibration of the model was accomplished by comparing water-level altitudes measured in observation wells to model-simulated water-level altitudes and by flow conditions.
Permeability of soils and glacial deposits in the study area are conducive to leaching of contaminants from the land surface to ground water (Lueth, 1974; Chamberlain, 1977). The presence and concentrations of many contaminants in ground water also are affected by the chemical composition of soils. Contaminants are less likely to seep to the water table in areas with peaty soils, due to adsorption by organic materials and small permeabilities that impede ground-water flow and contaminant advection.
The study area is mantled with sand, gravel, and clay deposited as river terraces and overbank or by glacial outwash from the Des Moines glacial lobe during the Pleistocene Epoch (Norvitch and others, 1978; Bloomgren and others, 1989; Meyer and Hobbs, 1989; Meyer, 1993; Anderson, 1993; and Andrews and others, 1998). The terrace overbank and outwash deposits typically are composed of brown to gray, coarse to fine, gravelly sand, with local clayey deposits along streams. Pebbles, cobbles, and sands consist of mafic and felsic igneous and metamorphic minerals, dolomite, and quartz. Those sands and gravels commonly are underlain by silty, sandy clay.
Ground water in the surficial aquifers flows toward the Mississippi River or to tributaries of the Mississippi River (Kanivetsky, 1989; Palen and others, 1989) (fig. 1). The water table in the surficial aquifers generally is within 20 ft of land surface. Helgeson and Lindholm (1977) estimated that recharge to these aquifers can be as great as 9-11 in./yr based on analyses of hydrographs of water levels in shallow wells. Therefore, the surficial sand and gravel aquifers are highly susceptible to contamination from activities at the land surface (Piegat, 1989; Meyer, 1993).
The study area also is underlain by as much as 1,000 ft of sedimentary strata. Sedimentary strata are underlain by so-called "basement" units of metamorphic and igneous rocks of Precambrian age. The primary bedrock aquifer underlying the study area is the Prairie du Chein-Jordan aquifer consisting of fractured sandy dolomite of the Prairie du Chien Group of Ordovician age, and the underlying quartzite sandstone of the Jordan Sandstone of Cambrian age (Setterholm and others, 1991). The Prairie du Chien Group is karstic, with ground-water flow occurring mainly through joints, fractures, and solution cavities, whereas flow in the Jordan Sandstone is primarily through the intergranular pore spaces and some joint partings (Delin and Woodward, 1984). The Prairie du Chien Group and the Jordan Sandstone are in effective hydraulic connection and have traditionally been considered a single aquifer (Delin and Woodward, 1984; Young 1992a; Young, 1992b).
Estimated horizontal hydraulic conductivity values derived from sediment-size analyses ranged from 3.1 to 190 ft/d (table 4), which are typical for materials ranging from glacial till to sand (Freeze and Cherry, 1979). Due to layering of sand and clay, vertical hydraulic conductivities of the surficial sand and gravel aquifers may be as much as five orders of magnitude less than horizontal hydraulic conductivities (Schoenberg, 1990).
Acidity of glacial sediment overlying the water table can affect the degradation and transport of organic compounds from the land surface to the saturated zone, because acidic organic compounds are more likely to be sorbed in alkaline sediments and basic organic compounds are more likely to be sorbed in acidic sediments. Sediment pH had a greater range (4.9–8.2) than pH in ground water, and increased slightly with depth (table 5).
Organic carbon in sediments can affect organic compounds such as pesticides and VOCs, which can leach from the land surface to ground water, because organic compounds can be sorbed to organic substances. Organic carbon also may provide a growth substrate for bacteria, which reduce sulfate and nitrate in ground water. Organic-carbon contents ranged from less than 3.2 to 160 grams per kilogram (g/kg) in the unsaturated zone (shallow) sediments, and from 0.5 to 110 g/kg in sediments near the top of the saturated zone (deep) (table 5). Because low organic-carbon contents were measured in most sediment samples, organic compounds that are transported through surface soils could readily seep through the unsaturated zone to the water table.
Inorganic carbon primarily is an indication of carbonate minerals in sediments, which can buffer pH and affect major ion chemistry of water samples. Inorganic carbon concentrations generally were greater in deep sediment samples than shallow sediment samples, indicating progressive dissolution of carbonate minerals as acidic water seeps from the land surface.
[ft/d, feet per day; NA, not applicable]
| Well or multipiezometers | Sampling depth interval (feet) | Estimated horizontal K value (ft/d) |
|---|---|---|
| HN-K (shallow) | 3-5 | 24 |
| HN-K (deep) | 20-22 | 82 |
| UFP-1 (shallow) | 3-5 | 4 |
| UFP-1 (deep) | 13-15 | 150 |
| UFP-2 (shallow) | 3-5 | 102 |
| UFP-2 (deep) | 12-14 | 190 |
| UFP-3 (shallow) | 3-5 | 9 |
| UFP-3 (deep) | 15-17 | 74 |
| UFP-4 (shallow) | 3-5 | 60 |
| UFP-4 (deep) | 7-9 | 180 |
| UFP-5 (shallow) | 3-5 | 5.1 |
| UFP-5 (deep) | 23-25 | 3.1 |
| UFP-6 (shallow) | 3-5 | 9.0 |
| UFP-6 (deep) | 13-15 | 16 |
| Median (shallow) | NA | 9.0 |
| Median (deep) | NA | 82 |
[The 15-digit station identification (ID) number can be used to access water-quality results in the U.S. Geological Survey National Water Information System. Local well or multipiezometer names are shown in figure 1. NA, not analyzed; shaded rows indicate sample in the unsaturated zone; <, less than]
| Well HN-K or multipiezometers | Site identification number | Sampling depth, in feet below land surface | Depth to water table, in feet below land surface | Date of measurement | Sediment pH | Organic carbon content (grams/ kilogram) | Inorganic carbon content (grams/ kilogram) |
|---|---|---|---|---|---|---|---|
| HN-K (shallow) | 450430093220801 | 3-5 | NA | 7/15/1997 | 7.0 | <0.20 | 0.1 |
| HN-K (deep) | 450430093220801 | 20-22 | 17.32 | 7/15/1997 | 7.9 | 2.9 | 9.1 |
| UFP-1 (shallow) | 450424093210501 | 3-5 | NA | 7/15/1997 | 4.9 | 4.2 | 1 |
| UFP-1 (deep) | 450424093210505 | 13-15 | 8.32 | 7/15/1997 | 6.6 | .5 | 4.0 |
| UFP-2 (shallow) | 450425093195303 | 3-5 | NA | 7/10/1997 | 5.6 | 160 | 2.6 |
| UFP-2 (deep) | 450425093195304 | 12-14 | 11.96 | 7/10/1997 | 6.6 | 1.6 | 4.0 |
| UFP-3 (shallow) | 450423093194303 | 3-5 | NA | 7/9/1997 | 6.9 | 4.8 | .4 |
| UFP-3 (deep) | 450423093194305 | 15-17 | 8.80 | 7/9/1997 | 6.8 | 1.0 | .1 |
| UFP-4 (shallow) | 450415092184801 | 3-5 | NA | 7/7/1997 | 7.1 | 13 | .7 |
| UFP-4 (deep) | 450414092184803 | 7-9 | 5.37 | 7/7/1997 | 8.2 | NA | NA |
| UFP-5 (shallow) | 450413093184301 | 3-5 | NA | 7/24/1997 | 7.4 | 3.2 | .1 |
| UFP-5 (deep) | 450413093184305 | 23-25 | 1.63 | 7/24/1997 | 7.2 | 110 | 73 |
| UFP-6 (shallow) | 450406093170204 | 3-5 | NA | 7/9/1997 | 6.9 | 3.3 | .1 |
| UFP-6 (deep) | 450406093170205 | 13-15 | 10.60 | 7/9/1997 | 7.6 | .9 | 1.6 |
| Median for all wells and piezometers (shallow) | NA | NA | NA | NA | 6.9 | 4.2 | 0.1 |
| Median for all wells and piezometers (deep) | NA | NA | 8.80 | NA | 7.0 | 1.3 | 4.0 |
Water-quality constituents summarized in this report include: physical properties; major ions, iron, manganese, nutrients, organic carbon, radon, pesticides, VOCs, and isotopes of nitrogen, hydrogen, and oxygen. Concentrations of chlorofluorocarbons, tritium, and dissolved gases also were determined for recharge-age dating.
Physical properties measured while purging water from access tubes to sampling ports, or during sample collection, included depth to water, specific conductance, pH, water temperature, turbidity, dissolved-oxygen concentration, and alkalinity (milligrams per liter (mg/L) as calcium carbonate (CaCO3)). The depth of the water table below land surface generally was less than 9 ft. Values of pH tended to be alkaline, and specific conductances and alkalinities were substantial, probably due to dissolution of carbonate minerals. Dissolved-oxygen concentrations generally were less than 1 mg/L, except on the upgradient end of the flow system at monitoring well HN-K (fig. 2). Dissolved-oxygen concentrations along much of the flow system were low and indicate that reduction processes such as denitrification, sulfate reduction, and reduction of metallic oxides may affect water quality in this aquifer.
Two-sided p-values from Kruskal-Wallis non-parametric tests (Helsel and Hirsch, 1992) of data from ports sampled three times (July 1997, October 1997, and August 1998) indicated that pH, turbidity, and dissolved-oxygen concentrations were significantly different (fig. 3) between the three samplings (two-sided p-value less than 0.025 means a greater than 95 percent probability that the sample groups are not statistically different). Decreases in dissolved-oxygen concentrations and turbidities with time could indicate stabilization of conditions in the aquifer after drilling and well installation. The reason for greater pH values in the samples collected October 1997, relative to the other two samplings, is unknown.
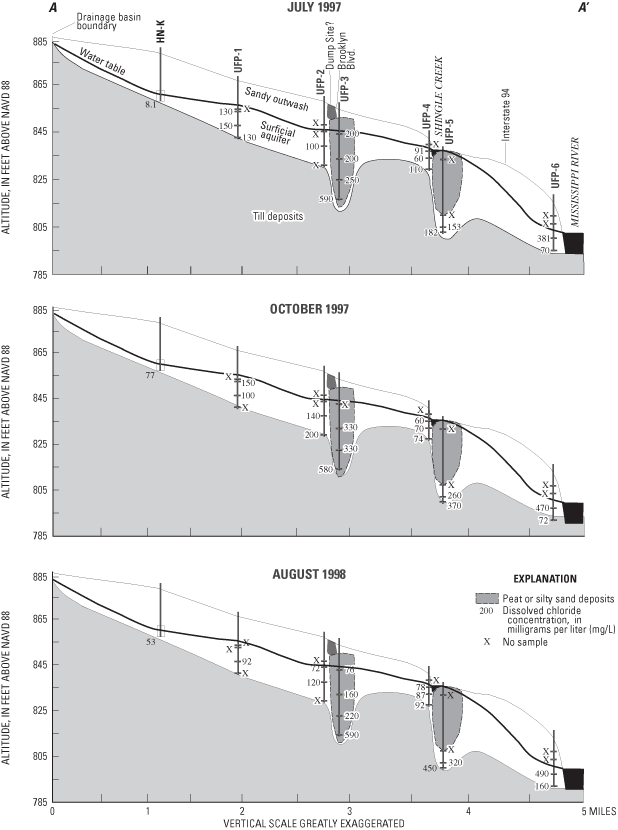
Specific conductances were significantly positively correlated to dissolved-solids concentrations (determined by evaporation of water samples at 180 degrees Celsius) (p=0.0001). Specific conductances and dissolved solids generally increased in the downgradient direction along the flow system, and with depth, except at UFP-6, where ground-water upwelling from underlying aquifers to the Mississippi River, or water originating from the Mississippi River, may have lower specific conductance than ground water in the surficial aquifer (fig. 4).
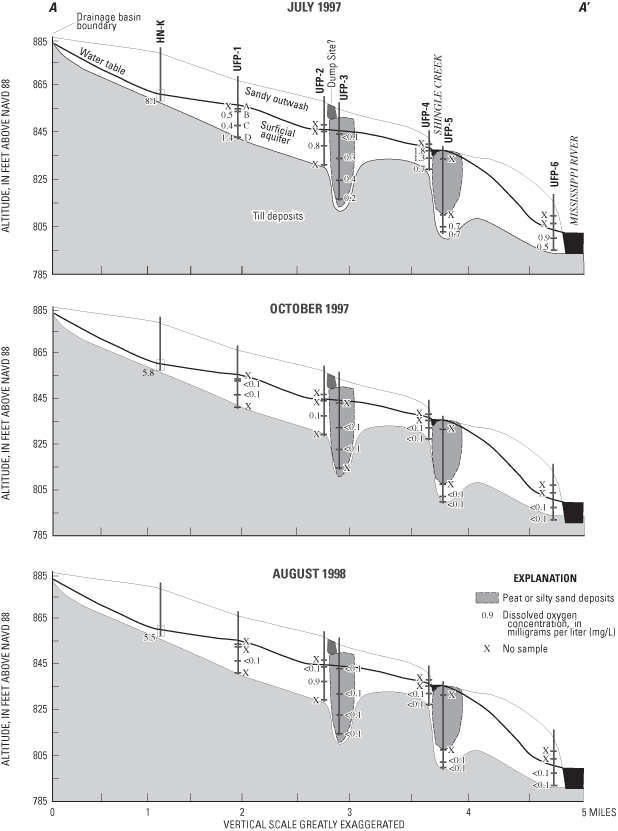
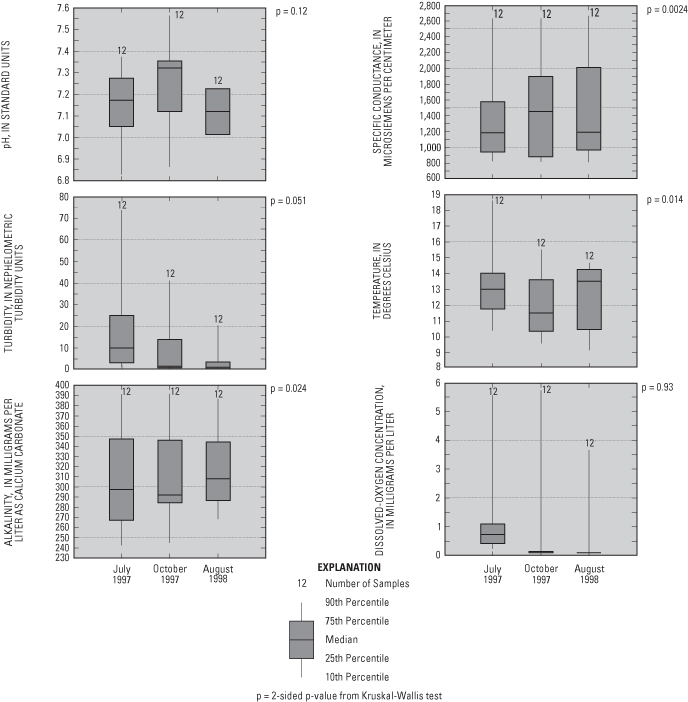
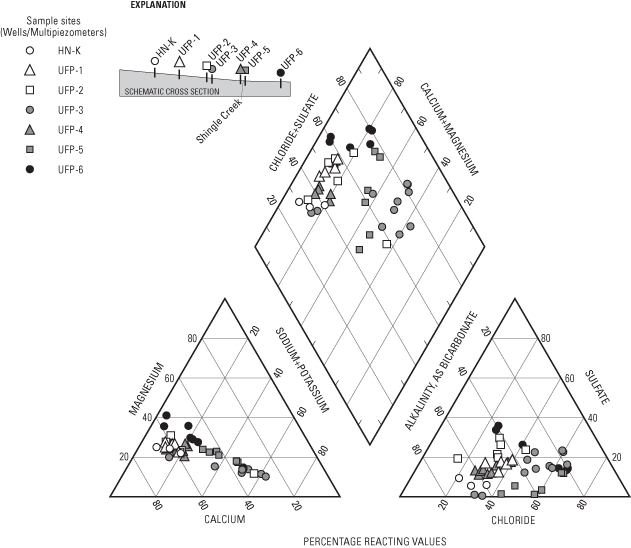
Major ions analyzed in water samples included calcium, magnesium, sodium, potassium, sulfate, chloride, fluoride, bromide (fig. 5), and bicarbonate (alkalinity). The primary major ions in water samples were calcium, magnesium, sodium, bicarbonate, sulfate, and chloride (figs. 5 and 6). Concentrations of these ions generally increase along the flow system (fig. 6) and with depth, with the exception of UFP-6, where ground water upwelling from underlying aquifers or water from the Mississippi River may have lower concentrations of these ions than ground water in the surficial aquifers. Calcium and bicarbonate generally increases along the flow system as a result of dissolution of carbonate minerals in the surficial aquifer. Sodium and chloride may be related to leaching from road salts and fertilizers. Sulfate probably is derived from the oxidation of sulfide minerals, oxidation of organic materials in soils and peat, leachate from concrete, or leachate from small amounts of gypsum in the surficial aquifer. With the exception of bromide, two-sided p-values of the Kruskal-Wallis test comparing major-ion concentrations between samplings were less than 0.025 for the major ions, indicating that concentrations were similar for the three samplings (fig. 5).
Granato and Smith (1999) found a positive correlation of chloride and sodium concentration with specific conductance in highway runoff containing road-salt constituents. A significant positive correlation between chloride and total dissolved-solids concentration (p-value=0.0001) in the urban flow- system study is probably a result of salt applied to de-ice roads. The positive correlation between sodium and total dissolved-solids concentrations also was significant (p-value=0.0001). Chloride concentrations were greatest at UFP-3, UFP-5, and UFP-6 (fig. 7). UFP-3 and UFP-6 are downgradient from Brooklyn Boulevard and Interstate 94, respectively, and UFP-5 is downgradient from Shingle Creek, which has substantial chloride concentrations during winter snowmelt (J.D. Fallon, U.S. Geological Survey, oral commun., January 6, 2005; Granato and Smith, 1999).
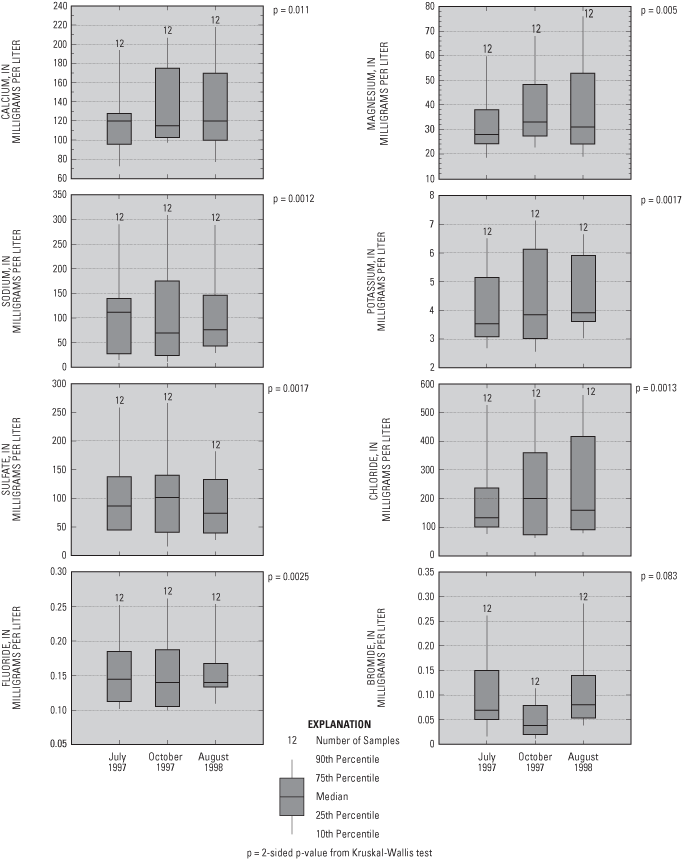
Figure 5. Major ion concentrations in water samples from the flow-system well and multipiezometers.
Because of concerns about stainless steel used in multipiezometers, a full suite of trace metals was not analyzed. Concentrations of dissolved iron and manganese were analyzed. Iron and manganese form insoluble oxide minerals in the presence of oxygen, which tend to dissolve in oxygen-depleted reducing conditions. Therefore, concentrations of these metals tend to increase in oxygen-depleted ground water. Iron (ranging from 0.033 to 18,600 micrograms per liter (µg/L)) and manganese (ranging from 543.7 to 2,606 µg/L) concentrations exceeded the Secondary Maximum Contaminant Levels (SMCLs) set by the U.S. Environmental Protection Agency (USEPA) of 300 µg/L for iron and 50 µg/L for manganese (U.S. Environmental Protection Agency, 1996).
Mineral saturation indices (Parkhurst, 1995) indicate minerals that may be present in an aquifer and minerals that are likely to dissolve or precipitate. If ground water is undersaturated with respect to a mineral, the ground water would theoretically dissolve the mineral if present. Conversely, if ground water is oversaturated with respect to a mineral, then the mineral would theoretically precipitate from the ground water. Equilibrium equations indicate that ground-water samples were oversaturated with respect to the iron and manganese oxide and hydroxide minerals hematite (Fe2O3), goethite (FeO(OH)), pyrolusite (MnO2), hausmannite (Mn2O4), and magnetite (MnO(OH)). Oversaturation determined for these minerals may be due to (1) overestimates of the concentrations of these metals due to the presence of colloids (particles with diameters less than the filter pore size of 0.45 microns), (2) high activation energies of mineral formation, or (3) overestimation of redox states that were calculated from the ratios of nutrient compound concentrations. Most of the water samples were oversaturated or nearly saturated with respect to the silicate minerals quartz (SiO2) and chalcedony (cryptocrystalline quartz) and the carbonate minerals rhodochrosite (MnCO3), aragonite (CaCO3), calcite (CaCO3), and dolomite (Ca,Mg(CO3)2), probably as a result of the presence or predominance of most of these minerals in clasts or grains of the surficial aquifer and underlying aquifers.
Concentrations of nitrite plus nitrate as nitrogen rarely contained a substantial nitrite component and these nutrients hereinafter are termed nitrate. Concentrations of nitrate in drinking water exceeding the USEPA Maximum Contaminant Level (MCL) of 10 mg/L as nitrogen have been associated with blue-baby syndrome (methemoglobinemia), and with increased rates of stomach cancer, birth defects, miscarriage, and leukemia (Dorsch and others, 1984: Forman and others, 1985; National Research Council, 1985; Fan and others, 1987). Nitrogen and phosphorus in ground water that discharges to surface water also can lead to eutrophication, which depletes oxygen in lakes and rivers, leading to increased mortality of oxygen- and temperature-sensitive native aquatic species.
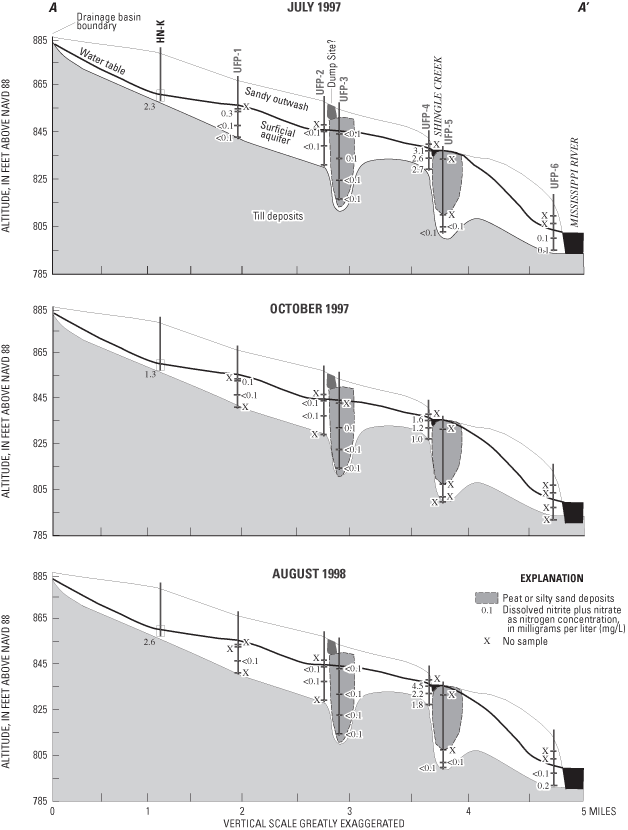
Most nitrate concentrations were less than 3 mg/L (fig. 8) and ranged from less than 0.050 to 4.15 mg/L. Nitrate in ground water principally is derived from animal wastes, soils, fertilizers, and nitrate-mineral deposits. Nitrate-mineral deposits do not occur in the study area. Most nitrate is oxidized (nitrified) in soils from the ammonia form of nitrogen (NH4 +), which is leached from animal wastes, soils, and fertilizers. Rainwater also is a source of nitrate to soils and ground water. Nitrate concentrations in rainfall in the Upper Midwest ranged from 1.0 to 1.5 mg/L in 1995 (National Atmospheric Deposition Program/National Trends Network, 1997). Nitrate can be removed from water by the processes of denitrification and assimilation. Denitrification is a process in which nitrate is reduced to gases such as nitrous oxide (N2O) or nitrogen gas (N2). Bacteria or reduced forms of metals in oxygen-depleted water facilitate denitrification. Assimilation is the uptake of nitrate and other nutrients by plant roots or by biota such as algae and bacteria. Nitrite (NO2) is an unstable intermediate product of nitrification, and does not commonly occur in substantial concentrations in ground water.
Two-sided p-values from the Kruskal-Wallis test indicate that concentrations of nitrogen compounds were similar in water samples collected during the three sampling events (fig. 8). Phosphorus compound concentrations increased during the last sampling (August 1998).
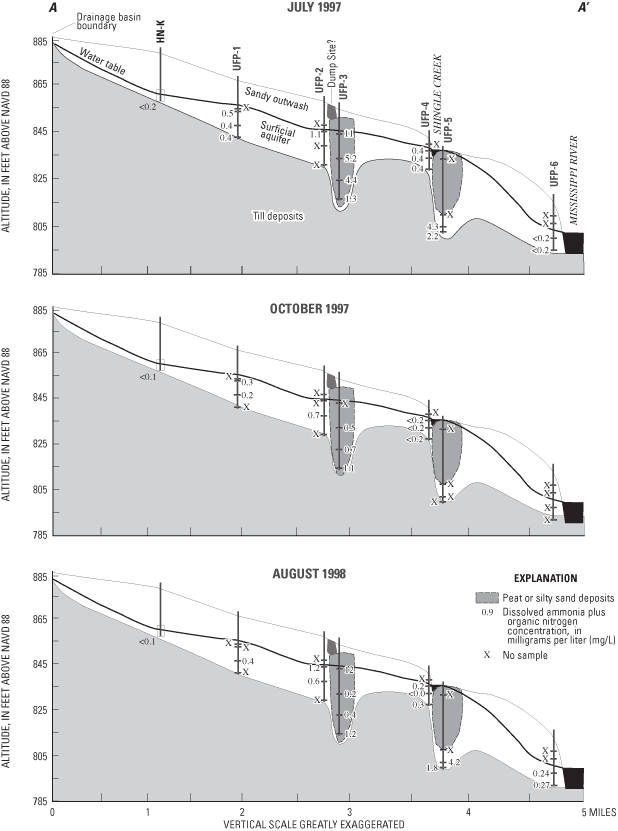
Concentrations of ammonia plus organic nitrogen (ranging from 0.10 to 5.20 mg/L) (reduced forms) were similar to those of nitrate (oxidized form), but concentrations of these constituents differed along the flow system (figs. 9 and 10). Nitrate concentrations generally were greatest in water samples collected from the upgradient end of the flow system and near Shingle Creek (fig. 9). As ground water flows through the aquifer, nitrate probably is both diluted and denitrified. Where conditions were reducing (deeper in the aquifer), reduced forms of nitrogen (ammonium plus organic nitrogen) were the dominant nitrogen compounds, for example UFP-3 (fig. 10).
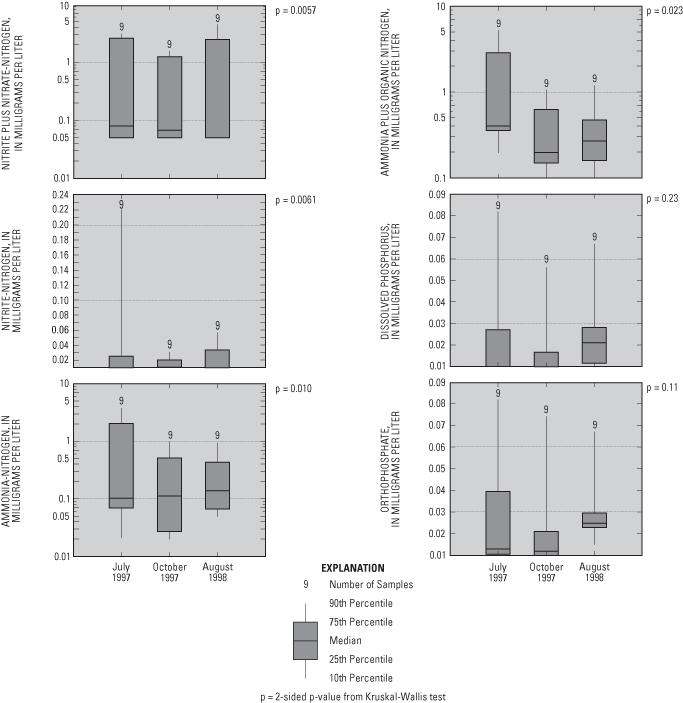
Figure 8. Nutrient concentrations in water samples from the flow-system well and multipiezometers.
The isotopic ratio of 15N/14N of nitrogen in nitrate molecules can indicate the source of nitrate and whether denitrification has occurred. The ratio between these two isotopes is reported as δ15N in parts per thousand (ppt) and is expressed as follows:
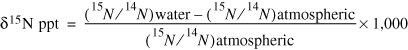 (2)
(2)
Atmospheric nitrogen gas consists principally of 14N (~99.632 percent, δ15N=~0) (Nier, 1955). The nitrogen-isotope ratio of nitrate derived from synthetic fertilizers generally is near that of atmospheric nitrogen gas (δ15N= -3 to +2 ppt) (Krietler, 1975). Nitrate leached from soils is somewhat enriched with 15N (δ15N=+2 to +8 ppt) as a result of preferential uptake of lighter 14N by plants and microbes (Krietler, 1975). Nitrate derived from animal wastes has greater amounts of 15N (δ15N=+10 to +20 ppt) (Krietler, 1975) as a result of preferential uptake of 14N and preferential evaporation of 14N as ammonia gas from human and animal wastes. Denitrification also increases δ15N values because denitrifying bacteria preferentially uptake lighter nitrate molecules due to greater thermodynamic efficiency.
Because analyses of nitrogen isotope ratios requires a minimum concentration of nitrate (3 mg/L in this case), water samples with nitrate concentrations less than that concentration were not analyzed for isotope ratios. Four of the 15 samples collected during July 1997 had sufficient nitrate concentrations to be analyzed. Nitrogen-isotope ratios (δ15N) in those water samples ranged from 6.5 to 10.3 ppt, indicating that the sources of nitrogen primarily were soils or animal wastes, or that denitrification that had increased nitrogen isotope ratios from an original, much lighter, fertilizer end member.
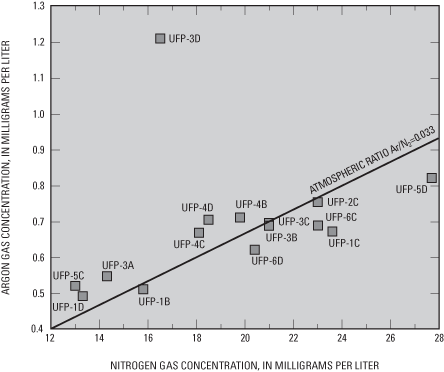
Concentrations of methane (CH4), carbon dioxide (CO2), nitrogen (N2), oxygen (O2), and argon (Ar) were determined to evaluate the redox state of water and the related likelihood of degradation of CFC concentrations used for age-dating of ground-water recharge. As oxygen is depleted by biotic or abiotic reactions in aquifers, bacteria can derive energy (through oxidation of carbon substrate) by removing oxygen atoms from nitrate (denitrification), sulfate (sulfate reduction), and carbon dioxide (methanogenesis). Greater ratios of the concentrations of dissolved nitrogen gas to argon gas (fig. 11) than the ratio of those gases in the atmosphere can indicate production of excess amounts of nitrogen gas in water by denitrification. In water samples from most of the sampling ports, nitrogen to argon ratios were similar to the atmospheric ratio, indicating little denitrification. Water samples most likely to have undergone denitrification, as indicated by that dissolved gas ratio, were from ports UFP-1C, 5D, 6C, and 6D (fig. 11), which are located in deeper parts of the surficial aquifers. Nitrate concentrations were less than 0.2 mg/L in water samples from those same ports. Methane concentrations exceeding 0.5 mg/L in water samples from multipiezometer wells UFP-3 and 5 probably were related to very low redox conditions at those wells.
The ratios of the isotopes of hydrogen (2H and 1H) and oxygen (18O and 16O) in water molecules can be used to determine the extent of evaporation in water samples based on comparison to the average ratios of those isotopes in precipitation (the meteoric water line). As with nitrogen isotopes, the ratios of the isotopes (R) are expressed in delta units (δ) as ppt as shown in the following equation:
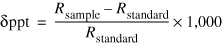 (3)
(3)
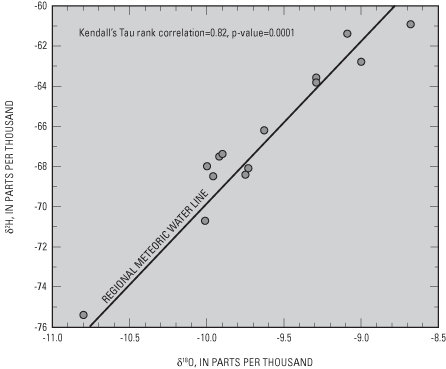
The standard is an arbitrary standard, known as Vienna Standard Mean Ocean Water (VSMOW). Because precipitation over continental areas originally is evaporated from ocean or lake water, it tends to be lighter than VSMOW as indicated by negative δ ppt values. For the most part, isotope ratios of hydrogen and oxygen (δ2H and δ18O) were well correlated with the regional meteoric water line (Craig, 1961) (fig. 12), indicating little evaporation of precipitation at the land surface prior to recharge. These results indicate rapid recharge in the study area.
Radon (222Rn) is a radioactive noble gas created by the decay of radium isotopes 223, 224, and 226 (Hem, 1985). Radon is soluble in water and readily volatilizes, with a half-life of 3.8 days. The primary health threat posed by radon in ground water is through atmospheric exposure (Wanty and Nordstrom, 1993). Radon can be absorbed by lung tissues, and alpha particles emitted during radioactive decay can damage those tissues. Prolonged respiration of high concentrations of radon gas have been related to increased incidence of lung cancer (National Research Council, 1988). Because of this risk, the USEPA has proposed a MCL for drinking water of 300 picocuries per liter (pCi/L) in states with an active indoor air program and an alternative MCL of 4,000 pCi/L in states that do not have an active indoor air program (U.S. Environmental Protection Agency, 2004).
Radon concentrations were analyzed in samples collected from multipiezometers during July 1997. Radon concentrations in those 15 samples ranged from 195 to 374 pCi/L, some exceeding the proposed MCL of 300 pCi/L in samples from ports UFP-3D, UFP-4B, UFP-4D, and UFP-6C. These radon concentrations generally were less than those reported in water samples from the deeper aquifers in the surrounding area (Fong and others, 1998).
Several different classes of pesticides, including triazines, organophosphorus, organochlorines, and carbamates, were analyzed in ground-water samples (table 2). Most pesticides are designed to decompose in the soil zone. Many pesticides sorb to organic carbon or clay in soil and are degraded by abiotic and biotic processes in soils and the unsaturated zone, and have relatively low solubilities in water (Rao and Alley, 1993). Soil and unsaturated-zone properties including hydraulic conductivity, organic-carbon content, pH, and cation-exchange capacity may affect leaching of a pesticide to ground water. Pesticides are more likely to be sorbed to less permeable soils with greater organic-carbon contents. Information about uses and application of pesticides in the study area is presented by Andrews and others (1998).
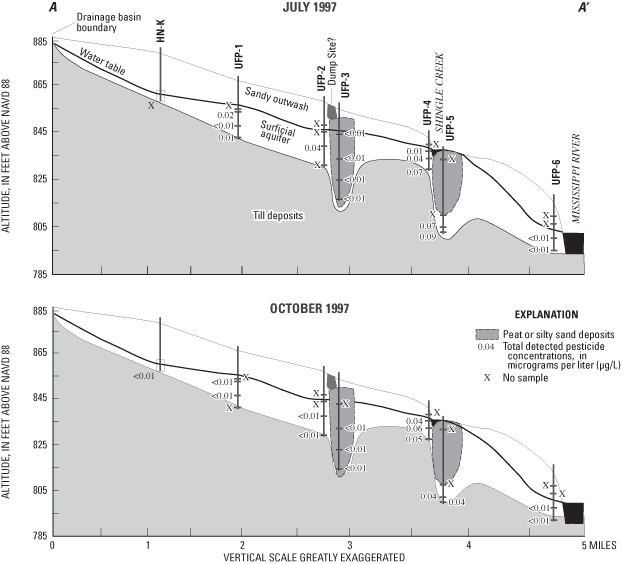
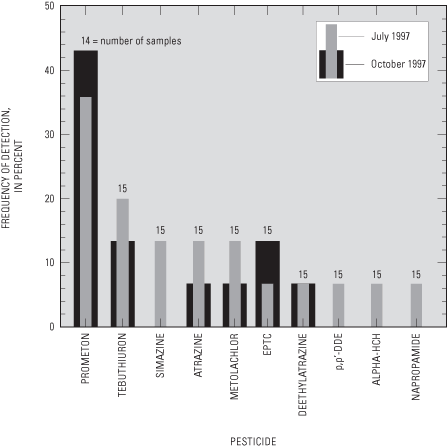
Water samples from 8 of the 15 ports sampled during July 1997 and in 5 of the 15 ports sampled during October 1997 had detectable concentrations of one or more pesticides (fig. 13). None of the concentrations of the 10 pesticides detected exceeded USEPA MCLs or Health Advisory Limits (HALs). Pesticide concentrations greater than the method detection limits, but less than the laboratory reporting levels, were reported as estimated concentrations.
Prometon, a nonselective, long-lasting herbicide used to control weeds and grasses on rights-of-way (Sine, 1993), was the most frequently detected pesticide (fig. 14). There is no MCL established for prometon, but the USEPA has established a lifetime HAL of 100 µg/L for drinking water (U.S. Environmental Protection Agency, 1996), which was not exceeded in any of the water samples (maximum concentration detected was 0.0598 µg/L). Prometon is relatively slow to degrade in soils, with an estimated half-life of 200 to 500 days (Rao and Alley, 1993; Weber, 1994). Prometon has a relatively low sorption coefficient to organic carbon in soils, making it more likely to leach to ground water (Weber, 1994).
Some herbicide compounds associated with agricultural applications were detected in some water samples. The source of these herbicides is not known. They may be associated with atmospheric deposition or agricultural applications prior to suburban development in the study area (Andrews and others, 1998). Tebuthiuron also is a herbicide labeled for use on rights-of-way and was the detected in water samples from three of the ports during July 1997 (UFP-5C, UFP-5D, and UFP-2C) (fig. 14) (maximum concentration of 0.0418 µg/L). Tebuthiuron has a greater solubility and a lower sorption coefficient than prometon and atrazine. Its half-life in soil is estimated to be 360 days (Weber, 1994). The USEPA established a lifetime HAL of 500 µg/L for tebuthiuron in drinking water (U.S. Environmental Protection Agency, 1996). Simazine, an herbicide commonly applied to corn, was also detected in water samples from two of the ports sampled during July 1997 (UFP-4C, UFP-4D). Metolachlor (a maximum concentration of 0.0418 µg/L), a pre-emergence herbicide used on corn, soybeans, grain sorghum, and potatoes (Sine, 1993), was detected in water samples from two of the ports (UFP-4B and UFP-4D) sampled during July 1997 and in water from port UFP-4C during October 1997. Metolachlor is classified as a possible human carcinogen with a lifetime HAL of 70 µg/L for drinking water (U.S. Environmental Protection Agency, 1996). Metolachlor is relatively soluble in water, is moderately volatile, has a relatively low sorption coefficient to organic carbon, and has a half-life of 40 days (Weber, 1994).
Atrazine, used to control annual grasses in corn (Winkelmann and Klaine, 1991), and its metabolite deethylatrazine were detected in water samples collected during July 1997 from two ports (UFP-1B and UFP-4D) and from well HN-K. During October 1997, atrazine and deethylatrazine were detected in a water sample from well HN-K. The maximum concentration detected was 0.00885 µg/L.
EPTC, an herbicide applied to beans, forage legumes, potatoes, and corn (Sine, 1993), was detected in a sample from UFP-5D during July 1997, and in water from UFP-5C and UFP-5D during October 1997 (estimated maximum concentration of 0.00262 µg/L). EPTC is highly volatile, moderately soluble in water, has a relatively low partition coefficient to organic carbon in soils, has a moderately short half-life of 7 days, and is readily degraded by soil micro-organisms (Weber, 1994). EPTC in ground water may be related to seepage from Shingle Creek during periods of high streamflow. Average estimated application rates for EPTC in 1987 were 39.9 and 56.9 lb/mi2 in Anoka and Hennepin Counties (Gianessi and Puffer, 1991), respectively, making EPTC the most heavily-applied registered pesticide in or adjacent to the study area. Due to the lack of cropland in the study area, actual applications of this compound are probably less than the averages for the counties.
The compound p,p'-DDE, an impurity and breakdown product of the insecticide DDT (Verschueren, 1983), can damage nervous systems of humans and other animals and is listed by the USEPA as a probable human carcinogen (Agency for Toxic Substances and Disease Registry, 2003). The compound p,p'-DDE was detected in water from port UFP-2C during July 1997, and in water from well HN-K during October 1997 (estimated maximum concentration of 0.00206 µg/L). The insecticide alpha-HCH, was detected in the water sample collected from UFP-1B during July 1997 (estimated concentration of 0.0012 µg/L). Napropamide, an herbicide used to control annual grasses and weeds in vegetables and turf (Extension Toxicology Network, 2003), was detected (0.00915 µg/L) in the water sample collected from UFP-4B during July 1997.
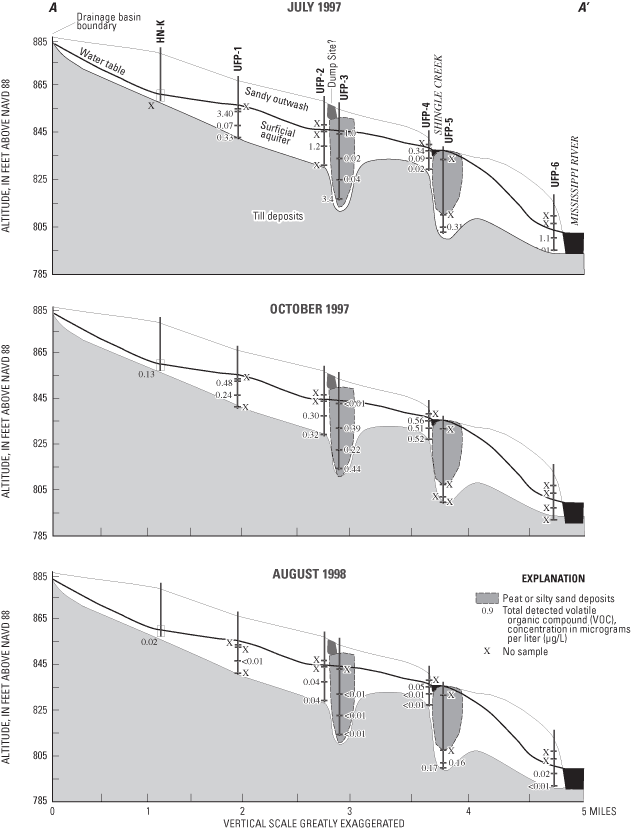
Volatile organic compounds (VOCs) are carbon-containing compounds that evaporate rapidly at normal temperatures and pressures. VOCs are contained in many commercial products including gasoline, paint, adhesives, solvents, wood preservatives, dry-cleaning agents, pesticides, cosmetics, correction fluids, and refrigerants. VOCs may reach ground water through spills and leaks at or near the land surface, from atmospheric dispersion into ground water, and through recharge of rainwater containing VOCs sorbed during passage through the atmosphere. Concentrations of 88 VOCs (table 3) were analyzed in water samples from the flow-system well and multipiezometers. None of the concentrations of the 26 detected VOCs exceeded MCLs set by the USEPA for drinking water (U.S. Environmental Protection Agency, 1996). VOC concentrations greater than the method detection limits, but less than laboratory reporting levels, were reported as estimated concentrations.
VOC concentrations generally were greatest at UFP-1 through UFP-4 (fig. 15). During July 1997, rainfall had seeped into an underground tank containing fuel oil, located approximately 100 ft to the northwest of UFP-1, forcing oil to the land surface (Anoka-Hennepin School District Staff, oral commun., 1997). Minor amounts of VOCs related to leakage of oil from that tank may have led to the detection of several VOCs in water samples from UFP-1 during July and October 1997. Detection of VOCs in water from UFP-3 may be associated with the proximity of a dump site (located about 300 ft west/upgradient) (Chris Malec, Minnesota Pollution Control Agency, oral commun., 2003), or nearby roadways.
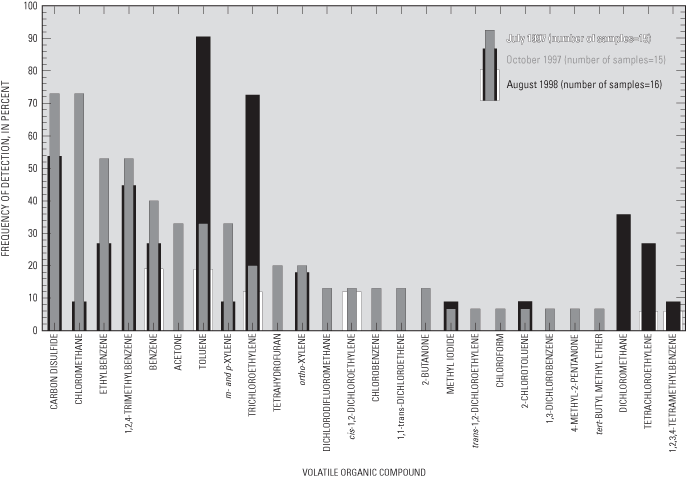
Twenty-three VOCs, most of which were solvents or fuel constituents, were detected in water samples collected during July 1997 (fig. 16) and 14 VOCs were detected in water samples collected during October 1997 (fig. 16). Only six VOCs were detected in water samples collected from the multipiezometers during August 1998 (fig. 16) due, at least in part, to a practice begun in 1998 of diluting samples by a factor of 2 prior to analysis at the laboratory. That practice probably contributed to the lack of detection of several VOCs, limiting comparability of data from the third sampling event to the previous sampling events. For more information on the uses and sources of each of the detected VOCs, refer to Andrews and others (1998) or Verschueren (1983).
The most frequently detected VOCs included carbon disulfide, chloromethane, toluene, and trichloroethylene. Carbon disulfide is a colorless liquid with a slightly pungent, sulfurous odor used in the manufacture of rayon, cellophane, carbon tetrachloride, rubber, soil disinfectants, soil conditioners, grain fumigants, herbicides, paper, pharmaceuticals, and electronic vacuum tubes (Versheuren, 1983). Carbon disulfide also is used as a solvent for phosphorus, sulfur, bromide, iodine, selenium, fats, and resins (Verscheuren, 1983). In addition to a wide variety of industrial uses, carbon disulfide is naturally emitted by bacteria in many soils, particularly soils rich in organic matter (Versheuren, 1983). At airborne concentrations exceeding 100 parts per million, carbon disulfide can cause illness or be toxic to humans, but no MCL for drinking water has been established for the compound. The substantial frequency of detection of low concentrations of this compound in ground water (maximum estimated concentration of 0.01 µg/L) indicates a widespread source of this compound, such as emissions from soils. Lack of knowledge about anthropogenic emissions or measurement of airborne concentrations of this compound precludes assessment as to whether the origins of this compound in ground water are natural or anthropogenic in the study area.
Chloromethane (CH3Cl), also known as methyl chloride, is used in manufacturing silicones, tetraethyllead, synthetic rubber, methyl cellulose, refrigerants, chloromethane, chloroform, carbon tetrachloride, and fumigants (Versheuren, 1983). Chloromethane also is used as a low-temperature solvent, in medicines, as a fluid in thermostatic equipment, as an extractant, a propellant, and in herbicides, and also is present in cigarette smoke (Versheuren, 1983). Low concentrations of chloromethane may have been created through reactions between naturally occurring organic matter in the water samples and the drops of 50 percent hydrochloric acid used to preserve the samples analyzed for VOCs. The USEPA has determined that chloromethane is a possible human carcinogen and has established an HAL for this compound of 3 µg/L for drinking water (U.S. Environmental Protection Agency, 1996), which was not exceeded in any of the water samples (maximum estimated concentration of 0.08 µg/L).
Toluene (C6H5CH3), also known as methyl benzene, can be emitted by petroleum refining and coal tar distillation and is used in the manufacture of benzene derivatives, saccharin, medicines, dyes, perfumes, TNT, and detergents (Verscheuren, 1983). Toluene is a component of gasoline and is used as a solvent for paints, coatings, gums, resins, rubber, lacquer, and adhesives (Verscheuren, 1983). In 1993, in Anoka and Hennepin Counties, 99,202 and 443,311 lbs, respectively, of this compound were released to air according to Toxic Release Inventory records (U.S. Environmental Protection Agency, written commun., 1995). Similar concentrations of toluene in ground water and equilibrium concentrations in air indicate that dispersion from the atmosphere or entrainment in rainfall may be the sources of this compound in ground water in the study area. The maximum estimated concentration in samples collected for this study was 0.22 µg/L.
The chlorinated alkene trichloroethene (CCl2CHCl), also known as TCE, is used for dry cleaning and metal degreasing; as a solvent for fats, greases, waxes, caffeine, and dyes; and as a refrigerant, fumigant, and anesthetic (Verscheuren, 1983). In 1993, in Anoka and Hennepin Counties, 55,417 and 421,458 lbs, respectively, of TCE were released to air according to records of the Toxic Release Inventory (U.S. Environmental Protection Agency, electronic commun., 1995). In ground water, TCE degrades to less chlorinated alkene compounds such as dichloroethene (DCE) and vinyl chloride. The maximum estimated concentration in water samples collected for this study was 0.015 µg/L.
A reconnaissance steady-state, ground-water-flow model was used to simulate the flow system in the study area and along the sampling transect to aid in understanding ground-water flow. The finite-difference program MODFLOW (McDonald and Harbaugh, 1988) was used with Ground-Water Modeling System (GMS) software (Environmental Modeling Systems, Incorporated, 2002) as the graphical user interface. MODPATH, a post-processing program developed for MODFLOW, was used to estimate flowpaths and dates of ground-water recharge using the particle tracking routine (Pollock, 1994).
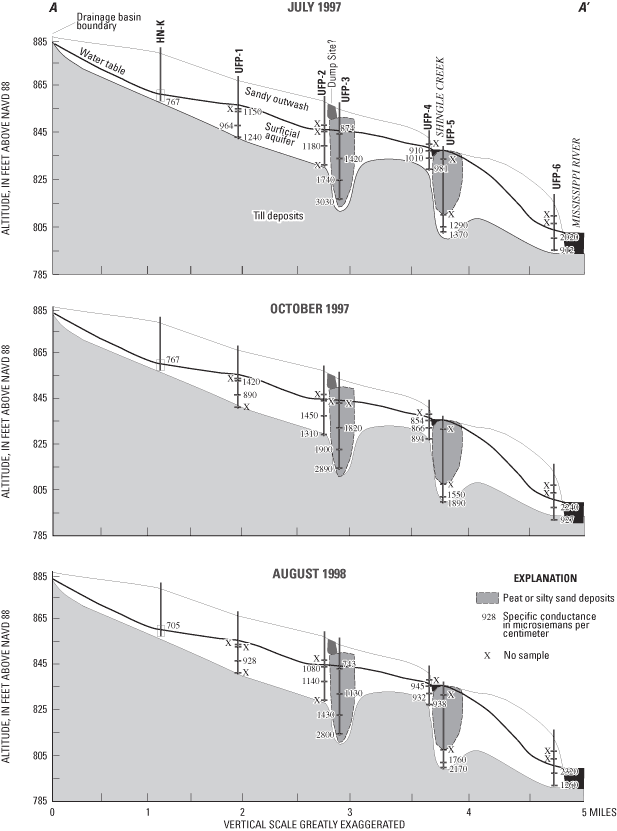
A three-dimensional, conceptual, variable-spaced, cell-centered, finite difference model grid was developed to emulate hydrologic conditions along the flow system (fig. 17). The grid contained sixteen 10-foot-thick layers of cells, 35 cells in the east-west direction, and 33 cells in the north-south direction. Cell dimensions varied to allow for the discretization needed along the flow system and in rivers and areas of low permeability. Construction of the model assumed equilibrium or steady-state conditions when inflow to the system equals outflow from the system. Hydrologic conditions from 1996 and 1997 were considered to be acceptable estimates of the flow system at equilibrium for purposes of the model because pumpage from the system was minimal and precipitation was near average.
Data collected for this study and existing data (Norvitch and others, 1978; Palen and others, 1989) were used to prepare input files of hydrologic unit thickness, hydraulic properties, and to identify inactive cells of low permeability. Peat and silty sand deposits of low permeability were incorporated into appropriate model cells.
Hydrographic data and water levels from existing wells were used to calibrate the model to hydraulic head. Boundary flux general-head cells, river cells, and drain cells were used to simulate internal and external boundary conditions. Flow between surficial aquifers and bedrock aquifers was considered negligible (Schoenberg, 1990).
Drain cells represented springs and seeps, river cells represented rivers, and general-head cells represented lakes and wetlands. River cells allowed leakage through the bottom of the river based on assumed differences in water levels and assumed streambed conductance. River cells provided or received as much water as the model required to reach a mathematical solution. Drain cells allowed leakage from the uppermost layers to springs or seeps. It was assumed that the stage in the springs or seeps reflected the altitude of the water table and that flow in the springs and seeps was predominantly from ground water. General-head boundaries were used to simulate subsurface flow at the margins of the model. They allowed ground water to enter or leave the simulated system based on relative head differences between the model cells and the regional potentiometric surface. No-flow boundaries were used to simulate the limits of the modeled area where ground-water flow was assumed to be nonexistent or insignificant (McDonald and Harbaugh, 1988). A no-flow boundary at the base of the model represented glacial till overlying bedrock.
River and general-head cells were assigned a value for the altitude of the respective river, lake, or wetland and for conductance. Altitude of the river, lake, or wetland bed was determined from USGS 7.5-minute topographic maps (U.S. Geological Survey, 1967). A bed thickness of 1 ft and a vertical hydraulic conductivity of 0.5 ft/d were used in the streambed conductance calculations (McDonald and Harbaugh, 1988). Drain cells were assigned values in a similar way.
Hydraulic heads in active model cells were assumed to represent steady-state conditions. Grain-size information from samples obtained during drilling were used to estimate horizontal and vertical hydraulic conductivities assigned. Evapotranspiration was not simulated. Net recharge (average infiltration minus average evapotranspiration) to the uppermost active cells was assumed to be 0.002 ft/d (Lindholm, 1980). The final estimates of hydraulic conductivity (table 6) for the aquifer were substantially greater than estimates based on samples from drill cuttings. This difference may indicate that the modeled value for recharge was less than actual recharge, that the thickness of the aquifer was not precisely known, or that soil-sample permeabilities were not representative estimates of average aquifer hydraulic conductivity.
Calibration of the model was accomplished by comparing water-level altitudes measured in observation wells to model-simulated water-level altitudes and comparing average baseflow to model-simulated flux. The mean absolute error was 2.61 ft. Simulated hydraulic heads were greater than measured hydraulic heads on average. This difference is within the range of annual fluctuation of the water table for surficial aquifers in Minnesota. Mass balance in recharge compared to discharge calculated by the model was within less than 0.01 percent. Recharge was from precipitation and from certain reaches of Shingle and Ryan Creeks. Discharge was to other reaches of the creeks and to the Mississippi River. Measured and simulated ground-water flow generally flowed to the east toward the Mississippi River. Simulated discharge to streams was used to help evaluate the model by comparing these values with stream discharge in Shingle Creek measured during baseflow. Average measured baseflow in Shingle Creek near well HN-U (fig. 1) varied from about 3 to about 6 ft3/s (Mitton and others, 1978; 1999), whereas model-simulated discharge to the river at this location was 6.3 ft3/s.
Table 6. Hydraulic properties of the modeled study area, Twin Cities metropolitan area, Minnesota.
| Stratigraphic unit (figure 1) | Horizontal conductivity (ft/d) | Vertical/horizontal anisotrophy ratio |
|---|---|---|
| Aquifer (terrace and overbank deposits and outwash) | 500 | 0.2 |
| Silty sand | 10 | .5 |
| Peat | 0.004 | .003 |
The steady-state ground-water simulation used herein represents a simplification of actual ground-water conditions, but this tool can provide insights about ground-water flow based on what is known about the aquifer system. A finite-difference method is an approximation of the ground-water flow equations, and in certain conditions the method may not be able to approximate the equations accurately. This model assumed steady-state hydrologic conditions and horizontal homogeneity of aquifer properties. Hydraulic conductivity probably varies throughout the aquifer, and in the model, most areas were assigned generalized (constant over a broad area) hydraulic conductivity values. Streambed hydraulic properties also were estimated, and it is possible that the conductances change throughout the modeled streambeds. Streamflow data for calibration were sparse and the effects from pumping wells were assumed to be negligible on ground water and streamflow.
Simulated ground-water flow and model-simulated hydraulic heads, coupled with measured ground-water recharge-age dates, were used to aid in understanding ground-water flow. Concentrations and ratios of CFCs can be used to estimate the year that the sampled water reached the water table (for water that has been recharged since the 1940s) (Thompson and Hayes, 1979). CFCs are dissolved in water as it falls through the atmosphere and flows through the unsaturated zone to the water table (Johnston and others, 1998). Because of the relatively inert nature of CFCs, their atmospheric concentrations and ratios can be maintained for long periods in water containing dissolved oxygen. In water with little to no dissolved oxygen, bacteria may metabolize CFCs, affecting estimated ground-water recharge dates.
CFC concentrations at the upgradient end of the flow system (well HN-K) and in the vicinity of Shingle Creek indicate that ground water was recharged about 1990 (fig. 18). However, CFC concentrations in samples from other multipiezometers indicate older water (fig. 18). Because of possible anaerobic degradation of CFCs, recharge dates at UFP-5 were estimated by comparing tritium concentrations at other ports with CFC-determined dates. The peak of tritium concentrations in rainfall, which coincided with the peak of nuclear testing during the early to mid-1960s, is reflected in the graph comparing tritium concentrations to CFC-determined dates of recharge from water samples collected during July 1997 from the flow-system well and multipiezometers (fig. 19a). The presence of many chemical constituents is related to dates of recharge and residence time in the surficial aquifers. For example, water from the ports near the water table and those with the greatest hydraulic conductivity generally were recharged most recently (figs. 18, 19b). Concentrations of calcium and magnesium, which are likely to be dissolved from the major minerals in the aquifer, tended to be less in more recently recharged ground water (fig. 19c). Concentrations of nutrients also related to dates of recharge. The largest concentrations of nitrate only occurred in ground water recharged since 1985 (fig. 19d).
Prometon, metolachlor, and simazine were only detected in ground water recharged after 1985, and the greatest detected total pesticide concentrations were in water recharged after 1975 (fig. 19e). Unlike pesticides, VOC concentrations were generally greater in older ground water (fig. 19f). Although the total detected concentrations of VOCs and date of recharge were not significantly correlated (p-value=0.49), the greatest detected concentrations of VOCs were in water samples recharged before 1975.
Reverse particle tracking was done at the flow-system well and multipiezometers to obtain an approximate particle flowpath traveltime (table 7, fig.17). A porosity of 0.2 was assumed for all model cells for particle tracking. An example of a simulated flow path through the flow system transect is shown in figure 17. The model predictions of recharge-age dates for HN-K, UFP-4, UFP-5 (port D) and UFP-6 (port D) were similar to the CFC/tritium dates (table 7). At well UFP-6 (port C), however, the model did not predict an accurate date, possibly due to misrepresentation of the model simulation near the boundary representing the Mississippi River. Simulated recharge-age at UFP-1, UFP-2, UFP-3, and UFP-5 (port C) were less consistent when compared with dates from water samples. Water from well UFP-1, located downgradient from a tributary to Shingle Creek, had a CFC/tritium age that was older than the corresponding model-computed age date, indicating that interaction between the creek and ground water was not as great as was simulated by the model. At UFP-2 (port C), the CFC/tritium age date also was older than the dates estimated from the model.
At UFP-3, completed in the low permeability silty sand and peat, the CFC/tritium date may have substantial error because of interference from carbon and methane in water from that well. The CFC/tritium dates were much older in the water in UFP-3 than the model predicted dates. Well UFP-5, completed in silty sand and peat deposits, also contained substantial carbon and methane, which may have affected CFC-calculated recharge-age dates. UFP-5A is completed in a peat unit and simulated flow indicates very slow traveltime indicating that ground water at that site probably was recharged at relatively early dates.
[TU, tritium units; CFC, chlorofluorocarbon; --, no sample]
| Site identifier (local well or multipiezometer name) | CFC/Tritium Date | Tritium (TU) | Model date from reverse particle tracking | Model comments |
|---|---|---|---|---|
| 450430093220801 (HN-K) | 1990 | 45.0 | 1992 | -- |
| 450424093210502 (UFP-1B) | 1972 | 37.8 | 1994 | Stream fed |
| 450424093210504 (UFP-1C) | 1959 | 26.9 | 1990 | -- |
| 450424093210505 (UFP-1D) | 1956 | 42.2 | 1986 | -- |
| 450425093195303 (UFP-2C) | 1977 | 31.7 | 1990 | -- |
| 450425093195304 (UFP-2D) | -- | -- | 1969 | -- |
| 450423093194303 (UFP-3B) | 1976 | 33.6 | 1986 | -- |
| 450423093194304 (UFP-3C) | 1973 | 39.4 | 1985 | -- |
| 450423093194305 (UFP-3D) | 1954 | 26.2 | 1976 | -- |
| 450415092184801 (UFP-4B) | 1995 | 23.0 | 1994 | -- |
| 450415092184802 (UFP-4C) | 1989 | 36.2 | 1989 | -- |
| 450414092184803 (UFP-4D) | 1985 | 36.5 | 1986 | -- |
| 450413093184301 (UFP-5A) | -- | -- | 1950 or older | Very slow traveltimes through peat unit |
| 450413093184303 (UFP-5B) | -- | -- | 1981 | -- |
| 450413093184304 (UFP-5C) | 1990 | 34.6 | 1979 | -- |
| 450413093184305 (UFP-5D) | 1973 | 38.7 | 1975 | -- |
| 450406093170204 (UFP-6C) | 1961 | 70.1 | 1990 | -- |
| 450406093170205 (UFP-6D) | 1966 | 91.8 | 1965 | -- |
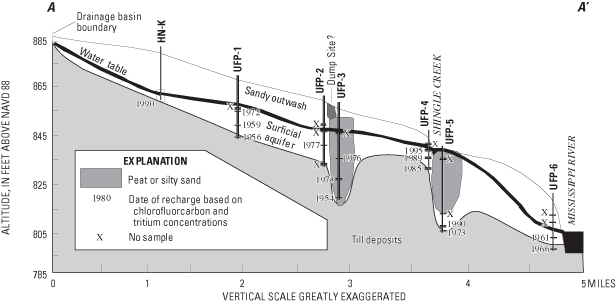
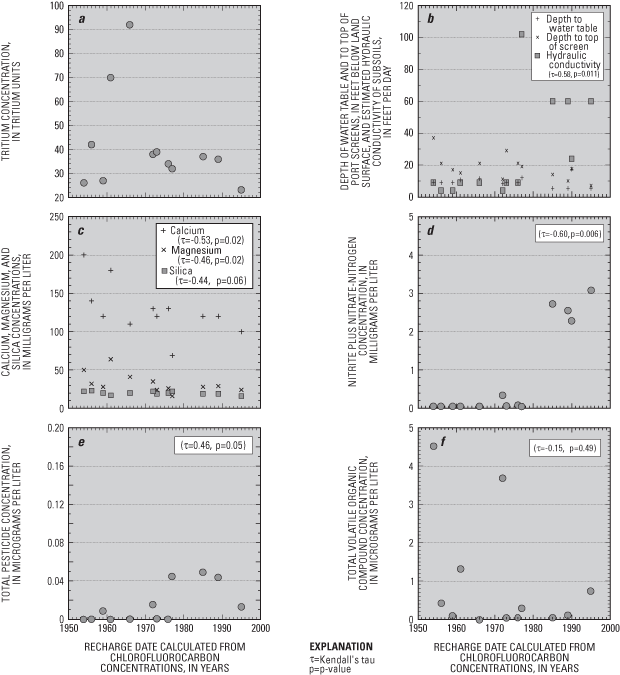
Surficial sand and gravel aquifers, similar to those in the study area for this report, are sources of ground-water discharge that sustain base flow to streams. In addition, these aquifers commonly are important sources of drinking water to supply wells because they are near land surface, and because drilling wells into these aquifers is relatively easy and inexpensive. However, surficial aquifers generally are highly susceptible to contamination because precipitation and contaminants can quickly infiltrate to the water table from the land surface.
Ground-water vulnerability to contamination is a combination of an aquifer's natural susceptibility to contamination and the potential availability of chemicals that could be transported to aquifers through infiltration. Identifying areas that are vulnerable to ground-water contamination, as well as understanding how contaminants move through and are transformed in aquifers, can help water-resource managers make land-use decisions that minimize the potential for contamination. The surficial aquifers described in this report are susceptible to contamination because the water table is near land surface, the aquifers have a high hydraulic conductivity, and contaminants from urban land uses are potentially available. Results from this study provide insights about factors and processes that affect ground-water quality of susceptible aquifers in urban areas.
Land use is an important factor in assessing the potential for ground-water contamination. During the last 40 years, the study area has been converted from an agricultural area to an area that is suburban. These changes have resulted in uses of complex mixtures of chemicals that are potential sources of ground-water contamination. Water samples from the aquifer had small concentrations of many anthropogenic compounds. Some nutrient and pesticide compounds may be relicts of past agricultural activity. Small concentrations of sodium, chloride, nutrients, and certain herbicides are more likely to be related to applications of chemicals used in urban and suburban areas. Small concentrations of VOCs detected during sampling probably are likely to be due to industrial and commercial activities, and to disposal of wastes from these activities.
In addition to directly affecting the presence and distribution of chemicals in ground water, land uses also are related to each other, to hydrologic characteristics in the study area, and to the presence and distribution of contaminants in ground water. For example, residential land uses comprise about one-half of the land area surrounding the flow-system well and multipiezometers in the eastern part of the study area. In relatively undeveloped areas of the western part of the study area, the water table is near land surface and home builders avoid those areas because they are prone to flooding or seepage of water into basements. Industrial development and road builders also avoid those areas because of poor drainage. Conversely, parks are present in greater percentages in those areas due to the attractiveness of water features, the low cost of the land, and the unsuitability of the land for other purposes. These factors result in differences in the kinds of compounds and concentrations of compounds detected locally in the aquifer.
Some compounds such as nitrate and dissolved oxygen are greatest in water samples from the upgradient end of the flow system and near the water table. As ground water flows through the aquifer, nitrate is diluted and denitrified. Specific conductance, dissolved solids, and ammonia increase and dissolved oxygen decreases along the flow system and with depth due to increase in residence time of water within the aquifer and dissolution of aquifer materials. Calcium and bicarbonate also increase along the flow system probably as a result of dissolution of carbonate minerals in the aquifer. Some constituent concentrations varied with depth and with location due to variability in the flow system. Distributions of sodium, chloride, certain pesticides, and VOCs were complex and appear to be related to local sources, such as roadways, petroleum spills, dump sites, and interactions with streams and wetlands receiving runoff that contains contaminants that locally affect ground water.
Although land use has a substantial effect on ground-water quality, the distribution of contaminants in the aquifer is affected by a complex combination of factors and processes that include sources of natural and anthropogenic contaminants, three-dimensional advective flow, physical and hydrologic settings, age, evolution, and transformation of chemical compounds along a flow system. Water quality is stratified vertically and along the flow system as indicated by the age of ground water, and physical and chemical transformations in soil and water systems. Older and more reduced ground water are likely to occur near the base and downgradient parts of the aquifer. Results from this study also indicate that monitoring that focuses on wells completed near the water table is useful in identifying the first arrival of contaminants to the aquifer but deeper wells are necessary to understand how ground-water contaminants are transported throughout flow systems, including to surface waters and deeper aquifers.
As part of a national analysis of the effects of urban land use on ground-water quality for the National Water-Quality Assessment Program, the U.S. Geological Survey sampled wells along a flow system in surficial glacial aquifers in the northwestern part of the Twin Cities metropolitan area during 1997 and 1998. In addition, a reconnaissance, steady-state ground-water model was developed to estimate flowpaths and dates of ground-water recharge using a particle-tracking routine.
Sediment samples from drilling were analyzed for sediment size, organic-carbon content, and pH. Sediment samples had high horizontal hydraulic conductivities (ranging from 3.1 to 190 ft/d, based on sediment-size analysis of well cuttings), small organic carbon concentrations (ranging from less than 0.2 to 160 g/kg), and mostly alkaline pH values (ranging from 4.9 to 8.2).
Water samples were analyzed for physical properties; concentration of major ions, iron, manganese, nutrients, organic carbon, radon, pesticides, volatile organic compounds (VOCs), chlorofluorocarbons, and tritium; and isotopes of nitrogen, hydrogen, and oxygen. Most of the water samples had small dissolved-oxygen concentrations (less than 1 milligram per liter (mg/L)). Calcium, magnesium, sodium, bicarbonate, sulfate, and chloride were the primary dissolved constituents in water samples. Mineral saturation indices indicated oversaturation with respect to oxides and hydroxides of iron and manganese. Iron and manganese concentrations in water samples exceeded Secondary Maximum Contaminant Levels set by the U.S. Environmental Protection Agency (USEPA).
Concentrations of nitrite plus nitrate as nitrogen (nitrate) were less than the USEPA Maximum Contaminant Level of 10 mg/L. Nitrate concentrations generally were greatest in water samples collected from the upgradient end of the flow system and near the water table. Nitrogen-isotope ratios indicated sources of nitrogen primarily were soils or animal wastes, and that denitrification may have increased nitrogen isotope ratios.
Isotope ratios of hydrogen and oxygen indicated that little evaporation of precipitation at the land surface occurs before recharge, thus indicating rapid recharge in the study area. Radon concentrations in some samples exceeded the proposed USEPA Maximum Contaminant Level of 300 picocuries per liter.
Small concentrations of pesticides were detected in the shallow parts of the aquifers. The herbicide prometon was the most frequently detected pesticide. Herbicides applied to control grasses and weeds in corn (atrazine, simazine, and metolachlor) also were frequently detected in water samples. All pesticide detections were less than USEPA Maximum Contaminant Levels or Health Advisory Limits. Twenty-six VOCs were detected in water samples, and all concentrations were below USEPA Maximum Contaminant Levels for drinking water.
Chlorofluorocarbon compounds and tritium concentrations were used to estimate dates of recharge of ground-water samples. In general, shallower ground-water samples were more recently recharged, although most water sampled from the aquifer water was recharged after 1955.
A three-dimensional, variable-spaced, cell-centered finite difference model was developed to estimate flowpaths and recharge age-dates of ground-water in the study area. Water-level altitudes, measured in observations wells, were compared to model-simulated water-level altitudes. Simulated hydraulic heads were within the range of fluctuation of measured ground-water heads. Mass balance in recharge compared to discharge calculated by the model was within less than 0.01 percent. Recharge was from precipitation and from certain reaches of Shingle and Ryan Creeks. Discharge was to other reaches of the creeks and to the Mississippi River. Measured and simulated ground water generally flows to the east toward the Mississippi River.
The model predictions of recharge-age dates at most sampling location and depths were similar to the CFC/tritium dates. At multipiezometer UFP-6 (port C) the model did not predict an accurate date possibly due to misrepresentation of the model simulation near the boundary representing the Mississippi River. Water from well UFP-1, located downgradient from a tributary to Shingle Creek, had a CFC/tritium age that was older than the corresponding model-computed age date, indicating that interaction between the creek and ground water was not as great as was simulated by the model. At some multipiezometers (UFP-3 and UFP-5) completed in low permeability silty sand and peat deposits, CFC/tritium dates may have substantial error because of interference from carbon and methane in water from those multipiezometer ports or degradation of CFC's in the flow system. Regardless of these problems, the conclusion that can be drawn for age-dating results is that ground-water age increases with depth in the aquifer and downgradient in the flow system.
Although land use has a substantial potential affect on ground-water quality, the distribution of contaminants in the aquifer is affected by a complex combination of factors and processes that include sources of natural and anthropogenic contaminants, three-dimensional advective flow, physical and hydrologic settings, age and evolution of ground water, and transformation of chemical compounds along the flow system. Water quality is stratified vertically and along the flow system, as indicated by the age of the ground water, the age of the land use, and physical and chemical transformations of the soil and water systems. Some compounds such as nitrate and dissolved oxygen are greatest in water samples from the upgradient end of the flow system and near the water table. Other constituents, such as specific conductance, dissolved solids, and ammonia increase along the flow system and with depth due to increase in residence time of water within the aquifers, reducing conditions, and dissolution of aquifer materials.
Agency for Toxic Substances and Disease Registry, 2003, ToxFAQs for DDT, DDE, and DDD: accessed December 29, 2003 at URL http://www.atsdr.cdc.gov/tfacts35.html
American Society for Testing and Materials, 2002, Standard test method for radon in drinking water: Annual Book of ASTM Standards, v. D 5072-98, p. 681-683.
Anderson, H.W., 1993, Effects of agricultural and residential land use on ground-water quality, Anoka Sand Plain aquifer, east-central Minnesota: U.S. Geological Survey Water-Resources Investigations Report 93-4074, 62 p.
Andrews, W.J., Fong, A.L., Harrod, Leigh, and Dittes, M.E., 1998, Water-quality assessment of part of the Upper Mississippi River Basin, Minnesota and Wisconsin—Ground-water quality in an urban part of the Twin Cities Metropolitan Area, Minnesota, 1996: U.S. Geological Survey Water-Resources Investigations Report 97-4248, 54 p.
Baker, D.G., Nelson, W.W., and Kuehnast, E.L., 1979, Climate of Minnesota Part XII, The hydrologic cycle and soil water: Agricultural Experiment Station Technical Bulletin 322, University of Minnesota, 24 p.
Bloomgren, B.A., Cleland, J.M., and Olsen, B.M., 1989, Depth to bedrock and bedrock topography, in Balaban, N.H., ed., Geologic atlas—Hennepin County, Minnesota: Minnesota Geological Survey, County Atlas Series, C4, 8 pl.
Bremner, J.M., 1965, Isotope-ratio analysis of nitrogen in nitrogen-15 tracer investigations, in Black, C.A., and Evans, D.D., eds., Methods of soil analysis (pt. 2): Madison, Wisc., American Society of Agronomy, p. 1256-1286.
Bremner, J.M., and Edwards, A.P., 1965, Determination and isotope-ratio analysis of different forms of nitrogen in soils—I. Apparatus and procedure for distillation and determination of ammonium: Soil Science Society Proceedings, v. 29, p. 504-507.
Brenton, R.W., and Arnett, T.L., 1993, Methods of analysis by the U.S. Geological Survey National Water-Quality Laboratory—Determination of dissolved organic carbon by UV-promoted persulfate oxidation and infrared spectrometry: U.S. Geological Survey Open-File Report 92-480, 12 p.
Busenberg, Eurybiades, and Plummer, L.N., 1992. Use of chlorofluorocarbons (CCl3F and CCl2F2) as hydrologic tracers and age-dating tools—The alluvium and terrace system of central Oklahoma: Water Resources Research, v. 28, no. 9, p. 2257-2283.
Chamberlain, L.M., 1977, Soil survey of Anoka County, Minnesota: U.S. Soil Conservation Service, 92 p., 71 pl.
Coplen, T.B., Wildman, J.D., and Chen, J., 1991, Improvements in the gaseous hydrogen-water equilibration technique for hydrogen isotope ratio analysis: Analytical Chemistry, v. 63, p. 910-912.
Craig, H., 1961, Isotopic variations in meteoric waters: Science, v. 133, p. 1833-1834.
Delin, G.N., and Landon, M.K., 1996, Multiport well design for sampling ground water at closely-spaced intervals: Ground Water, v. 34, no. 6, p. 1098-1104.
Delin, G.N., and Woodward, D.G., 1984, Hydrogeologic setting and the potentiometric surfaces of regional aquifers in the Hollandale Embayment, southeastern Minnesota: U.S. Geological Survey Water-Supply Paper 2219, 56 p.
Dorsch, M.M., Scragg, R.K., McMichael, A.U., Baghurst, P.A., and Dyer, K.F., 1984, Congenital malformations and maternal drinking water supply in rural South Australia— A case-control study: American Journal of Epidemiology, v. 119, p. 473-480.
Environmental Modeling Systems, Incorporated, 2002, The Department of Defense groundwater modeling system online reference manual available on the World Wide Web accessed December 11, 2002, at URL http://www.ems-i.com/gmshelp/gmsv40.htm.
Epstein, S., and Mayeda, T., 1953, Variation in 18O content of water from natural sources: Geochimica et Cosmochimica Acta, v. 4, p. 213-223.
Extension Toxicology Network, 2003, EXTOXNET: accessed September 30, 2003, at URL http://ace.orst.edu/info//extoxnet/ghindex.html
Fan, A.M., Wilhite, C.C., and Book, S.A., 1987, Evaluation of the nitrate drinking water standard with reference to infant methemoglobinemia and potential reproductive toxicity: Regulatory Toxicology and Pharmacology, v. 7, p. 135-137.
Farnsworth, D.K., Thompson, E.S., and Peck, E.L., 1982, Annual free water surface (FWS) evaporation from shallow lakes, 195670, map 3 of evaporation atlas for the contiguous 48 United States: National Oceanic and Atmospheric Administration Technical Report NWS 33, scale 1:4,800,000.
Fishman, M.J., and Friedman, L.C., eds., 1989, Methods for determination of inorganic substances in water and fluvial sediments: U.S. Geological Survey Techniques for Water-Resources Investigations, book 5, chap. A1, 545 p.
Fong, A.L., Andrews, W.J., and Stark, J.R., 1998, Water-quality assessment of part of the Upper Mississippi River Basin, Minnesota and Wisconsin—Ground-water quality in the Prairie du Chien-Jordan aquifer, 1996: U.S. Geological Survey Water-Resources Investigations Report 98-4248, 45 p.
Forman, David, Al-Dabbagh, S., and Doll, R., 1985, Nitrates, nitrites, and gastric cancer in Great Britain: Nature, v. 313, p. 620-625.
Freeze, R.A., and Cherry, J.A., 1979, Groundwater: Englewood Cliffs, New Jersey, Prentice-Hall, Inc., 604 p.
Gianessi, L.P., and Puffer, C.A., 1991, Herbicide use in the United States: Resources for the Future, Washington, DC, variously paged.
Granato, G.E., and Smith, K.P., 1999, Estimating concentrations of road-salt constituents in highway-runoff from measurements of specific conductance: U.S. Geological Survey Water-Resources Investigation Report 99-4077, 22 p.
Guy, H.P., 1969, Laboratory theory and methods for sediment analysis: U.S. Geological Survey Techniques of Water-Resources Investigations, book 5, chap. C1, 58 p.
Helgeson, J.O., and Lindholm, G.F., 1977, Geology and water-supply potential of the Anoka Sand-Plain aquifer, Minnesota: Minnesota Department of Natural Resources Technical Paper no. 6, 17 p.
Helsel, D.R., and Hirsch, R.M., 1992, Statistical methods in water resources: Elsevier, New York, 522 p.
Hem, J.D., 1985, Study and interpretation of the chemical characteristics of natural water (3d ed.): U.S. Geological Survey Water-Supply Paper 2254, 263 p.
Johnston, C.T., Cook, P.G., Frape, S.K., Plummer, L.N., Busenberg, Eurybiades, and Blackport, R.J., 1998, Ground-water age and nitrate distribution within a glacial aquifer beneath a thick unsaturated zone: Groundwater, v. 36, no. 1, p. 171-180.
Kanivetsky, Roman, 1989, Water-table system, in Balaban, N.H., ed., Geologic atlas—Hennepin County, Minnesota: Minnesota Geological Survey, County Atlas Series, C4, 8 pl.
Koterba, M.T., Wilde, F.D., and Lapham, W.W., 1995, Ground-water data-collection protocols and procedures for the National Water-Quality Assessment Program—Collection and documentation of water-quality samples and related data: U.S. Geological Survey Open-File Report 95-399, 113 p.
Krietler, C.W., 1975, Determining the source of nitrate in ground water by nitrogen isotope studies: University of Texas at Austin, Bureau of Economic Geology, Report of Investigations, no. 83, 57 p.
Lapham, W.W., Wilde, F.D., and Koterba, M.T., 1995, Ground-water data-collection protocols and procedures for the National Water-Quality Assessment Program—Selection, installation, and documentation of wells and collection of related data: U.S. Geological Survey Open-File Report 95-398, 69 p.
Lindholm, G.F., 1980, Ground-water appraisal of sand plains in Benton, Sherburne, Stearns, and Wright Counties, central Minnesota: U.S. Geological Survey Water-Resources Investigations Report 80-1285, 103 p.
Liu, Kon-Kee, 1979, Geochemistry of inorganic nitrogen in two marine environments—The Santa Barbara Basin and the ocean off Peru: University of California at Los Angeles, Ph.D. Dissertation, 354 p.
Lueth, R.A., 1974, Soil survey of Hennepin County, Minnesota: Soil Conservation Service, 159 p., 81 pl.
McDonald, M.G., and Harbaugh, A.W., 1988, A modular three-dimensional finite-difference ground-water flow model: U.S. Geological Survey Techniques of Water-Resources Investigations, book 6, chap. A1, 586 p.
Metropolitan Council, 2003, 1990 and 2000 census population and household statistics: accessed August 3, 2003 at URL http://www.metrocouncil.org/Census/9000_pophh.pdf
Meyer, G.N., 1993, Geologic sensitivity of the uppermost aquifer to pollution, in Meyer, G.N., Falteisek, J., Meints, J., and Dahlman, B., Regional hydrogeologic assessment, Anoka Sand Plain, Anoka, Chisago, Isanti, and Sherburne Counties, Minnesota: Minnesota Department of Natural Resources Regional Hydrogeologic Assessment, RHA-1, 3 pl.
Meyer, G.N., and Hobbs, H.C., 1989, Surficial geology, in Balaban, N.H., ed., Geologic atlas—Hennepin County, Minnesota: Minnesota Geological Survey, County Atlas Series, C4, 8 pl.
Miller, R.G., 1985, Multiple comparisons, in Kotz, S., and Johnson, N.L., eds., Enclyclopedia of Statistical Sciences: New York, John Wiley and Sons, v. 1, p. 679-689.
Minagawa, Masao, Winter, D.A., and Kaplan, I.R., 1984, Comparison of Kjeldahl and combustion methods for measurement of nitrogen isotope ratios in organic matter: Analytical Chemistry, v. 56, p. 1859-1861.
Minnesota Department of Health, 1994, Rules handbook— A guide to Minnesota rules, Chapter 4725.6450, Wells and borings, August, 1994: St. Paul, Minnesota Department of Health, p. 393-401.
Minnesota State Climatologist, 1995, Historical data retrieval and climate summaries: accessed June 15, 1995, at URL http://climate.umn.edu/doc/historical.htm
Mitton, G.B., Guttormson, K.G., Larson, W.W., Stratton, G.W., and Wakeman, E.S., 1998, Water-resources data, Minnesota water year 1997: U.S. Geological Survey Water-Data Report MN-97-1, 461 p.
Mitton, G.B., Guttormson, K.G., Stratton, G.W., and Wakeman, E.S., 1999, Water-resources data, Minnesota water year 1998: U.S. Geological Survey Water-Data Report MN-98-1, 410 p.
Moore, D.S., and McCabe, G.P., 1993, Analysis of variance, chap. 10 of Introduction to the Practice of Statistics (2d ed.): New York, W.H. Freeman and Company, p. 712-789.
National Atmospheric Deposition Program/National Trends Network, 1997, Nitrate ion concentration, 1995: accessed October 1, 1997, at URL http://nadp.sws.uiuc.edu/isopleths/maps1995/no3.gif
National Research Council, 1985, The health effects of nitrate, nitrite, and N-nitroso compounds: Washington, D.C., National Academy of Press, 723 p.
National Research Council, 1988, Health risks of radon and other internally deposited alpha-emitters: Washington, D.C., National Academy Press, 624 p.
Nier, A.D., 1955, Determination of isotopic masses and abundances by mass spectrometry: Science, v. 121, p. 737-744.
Norvitch, R.F., Ross, T.G., and Briekrietz, Alex, 1978, Water resources outlook for the Minneapolis-Saint Paul metropolitan area, Minnesota: Twin Cities Metropolitan Council, 219 p.
Ostlund, H.G., and Dorsey, H.G., 1975, Rapid electrolytic enrichment of hydrogen gas proportional counting of tritium: International Conference on Low Radioactivity Measurement and Applications, High Tatras, Czechoslovakia, October, 1975 [Proceedings], 6 p.
Palen, B.M., Kanivetsky, Roman, and Christopher, R.A., 1989, Water-table hydrogeology, in Balaban, N.H., ed., Geologic atlas—Hennepin County, Minnesota: Minnesota Geological Survey, County Atlas Series, C4, 8 pl.
Parkhurst, D.L., 1995, Users guide to PHREEQE-C computer program for speciation, reaction-path, advective-transport, and inverse geochemical calculations: U.S. Geological Survey Water-Resources Investigations Report 95-4227, 143 p.
Piegat, James, 1989, Sensitivity of ground-water systems to pollution, in Balaban, N.H., ed., 1989, Geologic atlas—Hennepin County, Minnesota: Minnesota Geological Survey, County Atlas Series, C4, 8 pl.
Pollock, D.W., 1994, User's guide for MODPATH MODFLOW PLOT, version 3—A particle tracking post-processing package for MODFLOW, the U.S. Geological Survey finite difference ground-water-flow model: U.S. Geological Survey Open-File Report 94-404, 249 p.
Rao, P.S.C., and Alley, W.M., 1993, Pesticides, in Alley, W.M., ed., Regional ground-water quality: New York, Van Nostrand Reinhold, p. 345-382.
Rose, D.L., and Schroeder, M.P., 1995, Methods of analysis by the U.S. Geological Survey National Water-Quality Laboratory—Determination of volatile organic compounds in water by purge and trap capillary gas chromatography/mass spectrometry: U.S. Geological Survey Open-File Report 94-708, 26 p.
Schoenberg, M.E., 1990, Effects of present and projected ground-water withdrawals on the Twin Cities aquifer system, Minnesota: U.S. Geological Survey Water-Resources Investigations Report 90-4001, 165 p.
Setterholm, D.R., Runkel, A.C., Cleland, J.M., Tipping, R.G., Mossler, J.H., Kanivetsky, R.M., and Hobbs, H.C., 1991, Geologic factors affecting the sensitivity of the Prairie du Chien-Jordan aquifer: Minnesota Geological Survey Open-File Report 91-5, 18 p.
Sine, C., ed., 1993, Farm chemical handbook 93: Willoughby, Ohio, Meister Publishing Company, variously paged.
Thompson, G.M., and Hayes, J.M., 1979, Trichlorofluoromethane in groundwater—A possible tracer and indicator of groundwater age: Water Resources Research, v. 15, no. 3, p. 546-554.
U.S. Bureau of Census, 1991, Census of population and housing, 1990: Washington, DC, Public law (P.L.) 94-171, data from CD Rom (Iowa, Minnesota, North Dakota, South Dakota, and Wisconsin).
U.S. Environmental Protection Agency, 1996, Drinking water regulations and health advisories: U.S. Environmental Protection Agency, Office of Water, EPA 822-B9-60-02, October 1996, 9 p.
U.S. Environmental Protection Agency, 2004, Proposed radon in drinking water rule: U.S. Environmental Protection Agency, accessed November 19, 2004, at URL http://www.epa.gov/safewater/radon/fact.html
U.S. Geological Survey, 1967, revised 1980, 7.5 minute series topographic, Minnesota, Minneapolis north quadrangle, scale 1:24,000.
Verschueren, Karel, 1983, Handbook of environmental data on organic chemicals: New York, Van Nostrand Reinhold, 1310 p.
Wanty, R.B., and Nordstrom, D.K., 1993, Natural radionuclides, in Alley, W.M., ed., Regional ground-water quality: New York, Van Nostrand Reinhold, p. 423-441.
Weber, J.B., 1994, Properties and behavior of pesticides in soil, in Honeycutt, R.C, and Schabacker, D.J., eds., Mechanisms of pesticide movement into ground water: Boca Raton, Fla., Lewis Publishers, p. 15-41.
Wershaw, R.L., Fishman, M.J., Grabbe, R.R., and Lowe, L.E., 1987, Methods for the determination of organic substances in water and fluvial sediments: U.S. Geological Survey Techniques of Water-Resources Investigations, book 5, chap. A3, 80 p.
Winkelmann, D.A., and Klaine, S.J., 1991, Atrazine metabolite behavior in soil-core microcosms—Formation, disappearance, and bound residues, in Somasundarum, L., and Coats, J.R., eds., Pesticide transformation products—Fate and significance in the environment: American Chemical Society, Symposium Series 59, Washington, D.C., p. 75-92.
Young, H.L., 1992a, Summary of ground-water hydrology of the Cambrian-Ordovician aquifer system n the northern Midwest, United States: U.S. Geological Survey 1405-A, 55 p.
Young, H.L., 1992b, Hydrogeology of the Cambrian-Ordovician aquifer system in the northern Midwest: U.S. Geological Survey Professional Paper 1405-B, 99 p.
Zaugg, S.D., Sandstrom, M.W., Smith, S.G., and Fehlberg, K.M., 1995, Methods of analysis by the National Water-Quality Laboratory—Determination of pesticides in water by C-18 solid-phase extraction and capillary column gas chromatography/mass spectrometry with selected-ion monitoring: U.S. Geological Survey Open-File Report 95-181, 60 p.
A total of 18 quality-assurance/quality-control (QA/QC) samples were collected in accordance with NAWQA protocols (Koterba and others, 1995). These samples included three VOC source-solution blanks; one field blank for major ions, nutrients, and VOCs; two field blanks for major ions, nutrients, pesticides and VOCs; three VOC trip blanks; three pesticide spikes; four VOC spikes; and two replicates.
Blanks collected were one of three types: source solution, trip, or field. Source solution blanks consist of specially-prepared deionized, distilled, nitrogen-purged water, which is decanted directly to sample bottles and shipped to the laboratory for analysis. The purpose of source solution blanks is to serve as a reference for the field blanks. Trip blanks consist of vials filled with VOC free blank water prepared at the USGS National Water-Quality Laboratory, which are sealed and kept in the sampling van for a defined period of time. Trip blanks verify that VOC vials are not being contaminated during storage, sampling, and shipment to the laboratory. One VOC trip blank (composed of three vials) was placed in the sampling van during each of the 3 weeks that samples were collected. Those trip blanks were subsequently shipped to the laboratory with other samples. Field blanks consist of three types of water prepared to be free of analyzed major ions and nutrients, pesticides, and VOCs, that have been pumped through the sampling system after standard post-sampling cleaning. Field blanks are used to check the efficacy of cleaning procedures, and that field methods, sample shipment, and laboratory procedures are not contaminating samples.
Two source solution blanks for VOCs were submitted on December 16, 1997, and one source-solution blank was submitted on August 13, 1998. Only 3 of the 88 analyzed VOCs were detected in the source solution blanks and those compounds were only detected at concentrations at or below the laboratory reporting level (table 8).
Trip blanks for VOCs were submitted on July 7, 1997; October 27, 1997; and August 12, 1998. The only VOC detected in trip blanks was 1,4-dichlorobenzene, which was detected at an estimated concentration of 0.01 µg/L (less than the reporting limit) in the trip blank submitted on July 7, 1997.
Calcium and magnesium were detected at small concentrations in most of the field blanks (table 9), but at concentrations much less than were typical for ground-water samples (fig. 5). Ammonia nitrogen and orthophosphorus were detected in low concentrations in the field blank collected on August 31, 1998. No pesticides were detected in any of the field blanks. Organic carbon was detected in all field blanks, with elevated concentrations in the blank collected on October 30, 1997 (table 9). Sixteen of the 88 analyzed VOCs were detected in one or more of the three field blanks. Three of those compounds—acetone, 1,4-dichlorobenzene, and toluene, also were detected in one or more of the VOC source solution blanks (table 8). Most of the detected concentrations of VOCs were less than the laboratory reporting levels. The field blank collected on October 30, 1997, had large concentrations of 2-butanone and toluene. The concentration of toluene also was elevated in the field blank collected on July 17, 1997. Contamination of these samples by those compounds may have occurred during collection, processing, and storage prior to analysis.
[all units are in micrograms per liter; NA, not applicable; <, less than; e, estimated]
| Compound | Reporting level/ method detection limit | December 17, 1997 | December 17, 1997 | August 13, 1998 |
|---|---|---|---|---|
| Acetone | 7.0/4.90 | <5.0 | <5.0 | e3.0 |
| 1,4-Dichlorobenzene | 0.50/.050 | e.02 | e.02 | <.05 |
| Toluene | .05/.05 | e.04 | e.02 | e.02 |
[mg/L, milligrams per liter; µg/L, micrograms per liter; na, not analyzed; --, none; >, less than
| Compound and concentration units | Laboratory reporting level/method detection limit | July 17, 1997 | October 30, 1997 | August 31, 1998 |
|---|---|---|---|---|
| Specific conductance (microsiemens per centimeter at 25 degrees Celsius) | 0.5/-- | 3.0 | 3.0 | 3.0 |
| pH | --/-- | 7.6 | 7.7 | 7.3 |
| Calcium (mg/L) | .02/-- | .15 | .14 | .05 |
| Magnesium (mg/L) | .001/.01 | <.01 | .023 | .004 |
| Acid neutralizing capability (mg/L as CaCO3) | 0.5/-- | 2.0 | 3.0 | .02 |
| Dissolved solids (mg/L) | 1.0/10 | 4.0 | 2.0 | <10 |
| Ammonia-nitrogen (mg/L) | .015/.01 | <.01 | <.015 | .042 |
| Dissolved orthophosphorus-phosphorus (mg/L) | .01/-- | <.01 | <.01 | .013 |
| Organic carbon (mg/L) | .1/-- | .2 | 13 | .3 |
| Acetone (µg/L) | 5.0 /-- | 16 | 9.0 | 3.0 |
| 1,2,4-trimethylbenzene (µg/L) | .056/-- | .01 | .01 | <.06 |
| 1,4-dichlorobenzene (µg/L) | .05/-- | <.05 | .1 | <.05 |
| Benzene (µg/L) | .021/0.1 | .04 | .06 | <.03 |
| Carbon disulfide (µg/L) | .07/0.37 | .05 | <.08 | <.08 |
| Chloroform (µg/L) | .024/-- | .04 | <.05 | <.05 |
| Ethylbenzene (µg/L) | .03/-- | <.03 | .01 | <.03 |
| Methyl iodide (µg/L) | .35/0.2 | na | .2 | <.2 |
| Bromomethane (µg/L) | .26/-- | <.1 | .1 | <.1 |
| Dichloromethane (µg/L) | .16/-- | <.4 | .5 | <.4 |
| 2-Butanone (µg/L) | 5.0/-- | <2.0 | 28 | <2.0 |
| m- and p-Xylene (µg/L) | .06/-- | .02 | <.06 | <.06 |
| ortho-Xylene (µg/L) | .07/-- | <.06 | .01 | <.06 |
| Styrene (µg/L) | .042/-- | <.04 | .01 | <.04 |
| Toluene (µg/L) | .05 /-- | .52 | .25 | .02 |
| Trichloroethylene (µg/L) | .038/-- | 00<.04 | .01 | <.04 |
Spiked samples are collected to assess analytical recovery and precision of pesticides and VOCs. Water samples from UFP-5C on July 24, 1997, and from UFP-2D on October 30, 1997, were spiked with known volumes of solutions containing known concentrations of pesticides. Most pesticides had mean recoveries ranging from about 50 to 120 percent (table 10). Of the compounds with poor recoveries, deethylatrazine was the only pesticide compound that was frequently detected in environmental samples. The relatively poor recoveries may indicate that its frequency of occurrence and reported concentrations may have been greater than described in this report. The pesticide-spiked sample collected on October 30, 1997, generally had poorer recoveries than the samples collected on July 24, 1997 (table 10).
Three water samples collected from well UFP-5C on July 24, 1997, and one water sample collected from well UFP-2D on October 30, 1997, were spiked with identical volumes of VOC solutions. Mean recoveries in VOC spike samples generally ranged from 70 to 110 percent (table 11). As with the pesticide-spiked samples, recoveries generally were less in the sample spiked on October 30, 1997.
In addition to spike samples, surrogates comprised of compounds similar in character to the standard analytes were added to every pesticide (diazinon-d10, alpha D6 HCH, and terbutylazine) and VOC (1,2-dichloroethane-d4, toluene-d8, 1,4-bromofluorobenzene) sample before analysis to assess recoveries (tables 10 and 11).
| Pesticide | Well UFP-5C (July 24, 1997) | Well UFP-5C (July 24, 1997) | Well UFP-2D (October 30, 1997) |
|---|---|---|---|
| Acetochlor | 90 | 98 | 49 |
| Alachlor | 82 | 95 | 47 |
| Atrazine | 88 | 94 | 64 |
| Deethylatrazine | 19 | 21 | 12 |
| Azinphos-methyl | 51 | 43 | 2 |
| Benfluralin | 99 | 99 | 56 |
| Butylate | 94 | 92 | 54 |
| Carbaryl | 110 | 110 | 40 |
| Carbofuran | 110 | 110 | 57 |
| Chlorpyrifos | 90 | 90 | 57 |
| Cyanazine | 73 | 78 | 34 |
| DCPA (dacthal) | 89 | 91 | 60 |
| p,p'-DDE | 72 | 74 | 43 |
| Diazinon | 110 | 110 | 62 |
| Dieldrin | 280 | 310 | 230 |
| Disulfoton | 28 | 50 | 34 |
| EPTC | 100 | 98 | 81 |
| Ethalfluralin | 90 | 87 | 60 |
| Ethoprop | 100 | 105 | 67 |
| Fonofos | 94 | 95 | 62 |
| alpha-HCH | 94 | 95 | 54 |
| Lindane | 88 | 87 | 45 |
| Linuron | 110 | 120 | 58 |
| Malathion | 91 | 86 | 35 |
| Parathion, methyl | 150 | 150 | 74 |
| Metolachlor | 94 | 98 | 53 |
| Metribuzin | 100 | 110 | 51 |
| Molinate | 110 | 110 | 78 |
| Napropamide | 88 | 93 | 68 |
| Parathion | 0 | 0 | 98 |
| Pebulate | 92 | 87 | 67 |
| Pendimethalin | 35 | 16 | 91 |
| cis-Permethrin | 39 | 39 | 19 |
| Phorate | 58 | 71 | 40 |
| Prometon | 82 | 77 | 45 |
| Propachlor | 93 | 88 | 35 |
| Propanil | 110 | 110 | 79 |
| Propargite | 79 | 76 | 31 |
| Simazine | 82 | 88 | 58 |
| Tebuthiuron | 81 | 73 | 27 |
| Terbacil | 54 | 64 | 31 |
| Terbufos | 71 | 68 | 23 |
| Thiobencarb | 99 | 100 | 80 |
| Triallate | 110 | 110 | 75 |
| Trifluralin | 110 | 100 | 66 |
| Surrogates | |||
| Diazinon-d10 | 104 | 110 | 69 |
| Alpha D6 HCH | 103 | 111 | 82 |
| Terbuthylazine | 105 | 106 | 98 |
| Compound | Well UFP-5C (July 24, 1997) | Well UFP-5C (July 24, 1997) | Well UFP-5C (July 24, 1997) | Well UFP-2D (October 30, 1997) |
|---|---|---|---|---|
| 1,1,1-2-Tetrachloroethane | 77 | 94 | 98 | 72 |
| 1,1,1-Trichloroethane | 81 | 110 | 110 | 73 |
| 1,1,2,2-Tetrachloroethane | 95 | 110 | 110 | 85 |
| 1,1,2-Trichloroethane | 91 | 110 | 110 | 82 |
| 1,1,2-Trichlorotrifluoromethane | 98 | 120 | 130 | 62 |
| 1,1-Dichloroethane | 82 | 100 | 110 | 79 |
| 1,1-Dichloropropene | 89 | 110 | 120 | 80 |
| 1,2,3,5-Tetramethylbenzene | 75 | 94 | 100 | 69 |
| 1,2,3-Trichlorobenzene | 91 | 110 | 110 | 78 |
| 1,2,3-Trichloropropane | 97 | 110 | 110 | 66 |
| 1,2,3-Trimethylbenzene | 78 | 96 | 100 | 78 |
| 1,2,4-Trichlorobenzene | 74 | 91 | 91 | 63 |
| 1,2,4-Trimethylbenzene | 74 | 94 | 100 | 69 |
| 1,2-Dibromo-3-chloropropane | 91 | 100 | 100 | 66 |
| 1,2-Dibromoethane | 82 | 99 | 100 | 76 |
| 1,2-Dichlorobenzene | 88 | 100 | 100 | 80 |
| 1,2-Dichloroethane | 88 | 100 | 100 | 80 |
| 1,2-Dichloropropane | 83 | 100 | 110 | 76 |
| 1,3,5-Trimethylbenzene | 74 | 96 | 100 | 66 |
| 1,3-Dichlorobenzene | 74 | 94 | 98 | 64 |
| 1,3-Dichloropropane | 74 | 94 | 98 | 64 |
| 1,4-Dichlorobenzene | 73 | 92 | 97 | 90 |
| 2,2-Dichloropropane | 61 | 77 | 88 | 42 |
| 2-Butanone | 93 | 100 | 93 | 110 |
| 2-Chlorotoluene | 83 | 100 | 100 | 66 |
| 2-Hexanone | 100 | 110 | 110 | 100 |
| 3-Chloropropene | 110 | 120 | 140 | 48 |
| 4-Chlorotoluene | 78 | 92 | 100 | 67 |
| 4-Methyl-2-pentanone | 88 | 97 | 97 | 79 |
| Acetone | 120 | 130 | 130 | 87 |
| Acrylonitrile | 83 | 83 | 83 | 83 |
| Benzene | 88 | 110 | 110 | 83 |
| Bromobenzene | 78 | 95 | 100 | 70 |
| Bromochloromethane | 77 | 98 | 98 | 76 |
| Bromodichloromethane | 78 | 97 | 97 | 80 |
| Bromoethene | 110 | 130 | 140 | 75 |
| Bromoform | 72 | 85 | 88 | 75 |
| Bromomethane | 97 | 97 | 130 | 32 |
| n-Butylbenzene | 63 | 86 | 91 | 57 |
| Carbon disulfide | 130 | 140 | 130 | 79 |
| Chlorobenzene | 82 | 100 | 110 | 76 |
| Chloroethane | 61 | 52 | 88 | 61 |
| Chloroform | 77 | 98 | 100 | 75 |
| Chloromethane | 120 | 120 | 140 | 54 |
| cis-1,2-Dichloroethene | 91 | 110 | 110 | 84 |
| cis-1,3-Dichloropropene | 77 | 98 | 100 | 65 |
| Dibromomethane | 80 | 99 | 100 | 77 |
| Dibromochloromethane | 85 | 100 | 100 | 85 |
| Dichlorodifluoromethane | 160 | 140 | 190 | 65 |
| Dichloromethane | 100 | 130 | 120 | 100 |
| Diethyl ether | 94 | 110 | 110 | 63 |
| Diisopropyl ether | 75 | 97 | 110 | 86 |
| Ethyl methacrylate | 81 | 93 | 97 | 70 |
| tert-Ethyl-butyl ether | 83 | 98 | 98 | 89 |
| Ethylbenzene | 82 | 100 | 100 | 74 |
| Hexachlorobutadiene | 74 | 97 | 97 | 58 |
| Hexachloroethane | 70 | 91 | 97 | 55 |
| Isopropylbenzene | 81 | 100 | 110 | 66 |
| m-and p-Xylene | 83 | 100 | 110 | 71 |
| Methyl acrylate | 97 | 110 | 110 | 96 |
| Methyl acrylonitrile | 92 | 100 | 100 | 104 |
| Methyl iodide | 70 | 97 | 120 | 89 |
| Methyl methacrylate | 82 | 91 | 93 | 84 |
| tert-butyl methyl ether | 87 | 110 | 110 | 87 |
| n-Propylbenzene | 73 | 94 | 98 | 64 |
| Naphthalene | 120 | 140 | 140 | 71 |
| ortho-Ethyl toluene | 80 | 98 | 100 | 62 |
| ortho-Xylene | 83 | 100 | 110 | 71 |
| sec-Butylbenzene | 65 | 83 | 91 | 53 |
| Styrene | 70 | 86 | 96 | 71 |
| tert-Amyl methyl ether | 77 | 96 | 96 | 77 |
| tert-Butylbenzene | 73 | 93 | 97 | 63 |
| Tetrachloroethylene | 94 | 110 | 110 | 100 |
| Tetrachloromethane | 74 | 96 | 100 | 68 |
| Tetrahydrofuran | 75 | 74 | 90 | 75 |
| Toluene | 82 | 100 | 100 | 77 |
| trans-1,2-Dichloroethene | 94 | 120 | 120 | 79 |
| trans-1,3-Dichloropropene | 81 | 95 | 100 | 71 |
| trans-1,4-Dichloro-2-butene | 75 | 86 | 100 | 51 |
| Trichloroethylene | 90 | 110 | 120 | 82 |
| Trichlorofluoromethane | 98 | 120 | 130 | 62 |
| Vinyl chloride | 120 | 130 | 140 | 57 |
| Surrogates | ||||
| 1,2-Dichloroethane-d4 | 100 | 100 | 110 | 97 |
| Toluene, D8 | 100 | 100 | 100 | 96 |
| 1,4-Bromofluorobenzene | 100 | 100 | 100 | 85 |
Replicates for this study are ground-water samples collected sequentially at the same well and are analyzed for the same compounds using the same methods. The purpose of replicate samples is to assess variability that may result during sample collection and laboratory analysis. One set of replicate samples was collected from port UFP-5C on July 24, 1997, and from UFP-3C on October 29, 1997 (table 12). Precision is calculated from two replicate samples, expressed as relative percent difference (RPD):
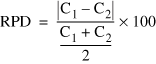 (4)
(4)
where C1 is the larger of the two concentrations and C2 is the smaller of the two concentrations.
If the estimated concentration of a constituent in one sample of a pair was less than the laboratory reporting level, the reporting level was substituted for that concentration to calculate the RPD. If both constituent concentrations in the replicate pair were less than the reporting level, then the RPD was not calculated and the concentrations were assumed to be the same.
Constituents with concentrations varying by more than 10 percent between the samples and replicates are shown in table 12. The relatively few compounds (of the more than150 compounds measured) with greater than 10 percent relative percent differences and the small relative differences of those constituents, in terms of concentration, indicate that results were highly reproducible.
[mg/L, milligrams per liter; µg/L, micrograms per liter; <, less than]
| Compound and concentration units | Laboratory reporting level | Concentration | Replicate concentration | Relative percent difference |
|---|---|---|---|---|
| UFP-5C July 24, 1997 | ||||
| Dissolved phosphorus (mg/L) | 0.01 | 1.44 | 1.67 | 15 |
| Dissolved orthophosphorus (mg/L) | .01 | 1.34 | 1.69 | 23 |
| Organic carbon (mg/L) | .5 | 8.3 | 9.3 | 11 |
| Tebuthiuron (µg/L) | .01 | .042 | <.01 | 123 |
| Benzene (µg/L) | .03 | .05 | .04 | 22 |
| Carbon disulfide (µg/L) | .01 | .08 | .06 | 29 |
| UFP-3C October 29, 1997 | ||||
| Bromide (mg/L) | .01 | .08 | .09 | 12 |
| Dissolved phosphorus (mg/L) | .01 | .013 | <.01 | 26 |
| Dissolved orthophosphorus (mg/L) | .01 | .23 | .17 | 30 |
| 1,4-Dichlorobenzene (µg/L) | .2 | .12 | .08 | 40 |
| Toluene (µg/L) | .22 | .08 | .06 | 29 |
For more information concerning the research in this report, contact:
Director
U.S. Geological Survey
Water Science Center of Minnesota
2280 Woodale Drive
Mounds View, MN 55112
Document Accessibility: Adobe Systems Incorporated has information about PDFs and the visually impaired. This information provides tools to help make PDF files accessible. These tools convert Adobe PDF documents into HTML or ASCII text, which then can be read by a number of common screen-reading programs that synthesize text as audible speech. In addition, an accessible version of Acrobat Reader 6.0, which contains support for screen readers, is available. These tools and the accessible reader may be obtained free from Adobe at Adobe Access.
| AccessibilityFOIAPrivacyPolicies and Notices | |
 |
|