Elemental chemistry of streambed sediments
Implications for sources of trace elements
Namekagon River upstream and downstream of Hayward, Wisconsin
St. Croix River upstream and downstream of Stillwater, Minnesota-Hudson, Wisconsin
Kettle River upstream and downstream of Sandstone and Hinckley, Minnesota
Rush Creek upstream and downstream of Rush City, Minnesota
Osceola Creek upstream and downstream of Osceola, Wisconsin
Implications for water-resource quality
Streambed sediments from 30 sites in the St. Croix River Basin were analyzed for selected chemical elements. Possible occurrences of low-level contamination include: (1) elevated concentrations of arsenic, lead, silver, and to a lesser extent cadmium, copper, and mercury in the Namekagon River downstream of Hayward, Wisconsin; (2) elevated lead concentrations in the St. Croix River downstream of the Stillwater, Minnesota—Hudson, Wisconsin region; (3) slightly elevated concentrations of Arsenic, Cadmium, Copper, Lead, Mercury, Silver, Vanadium, and Zinc in the Kettle River downstream of Sandstone and Hinckley, Minnesota; (4) and substantially elevated mercury concentrations in Rush Creek downstream of Rush City, Minnesota. Elevated concentrations of copper, lead, and to a lesser extent cadmium, in Osceola Creek, downstream of Osceola, Wisconsin may be due to anthropogenic sources or contributions from bedrock that differs geochemically from sediments farther upstream. There does not appear to be large anthropogenic contributions of trace elements—above the background geochemical and atmospherically deposited levels—in the streams sampled for this study. Many of the elements appear to be associated with mineral sources. Concentrations of potentially toxic trace elements in the St. Croix River Basin generally were lower than concentrations associated with frequent adverse effects to aquatic biota. Exceptions were arsenic (7 of 30 samples) and mercury (1 of 30 samples).
Trace elements are natural constituents of the earth's crust; all bedrock and sedimentary deposits contain some levels of trace elements. Human activities such as mining, ore smelting, industrial processes, fossil-fuel combustion, and many other activities, may greatly augment trace element concentrations in the environment. Streambed sediments can be a useful medium for trace element analyses. Many trace elements strongly sorb to sediment particles. Streambed sediments can accumulate chemicals over time, and may be useful archives of past contamination. If significant trace element contamination is introduced to a stream—either transiently or continuously—streambed sediments should accumulate some portion of the elements through chemical and physical sorption processes. Processes that affect the fate of sediment-bound trace elements in rivers include sedimentation, burial of sediments in depositional areas (especially lakes and reservoirs), resuspension (especially during high flow events), desorption, and biological uptake.
Urban development in the St. Croix River Basin and the effects of potentially toxic contaminants and sediment from urban runoff have been identified as major issues in the basin by the St. Croix Basin Planning Team (Holmberg and others, 1997). Previous studies have found elevated concentrations of selected trace elements in the St. Croix River Basin. For example, fish-consumption advisories are established by Minnesota and Wisconsin because of potentially toxic levels of mercury in fish taken from the St. Croix River. A study by Troelstrup and Foley (1993) indicated that the higher concentrations of trace elements in mussel shells in the lower reaches of the St. Croix River Basin, compared to upstream reaches, corresponded to slower growth rates and sizes for those organisms. Troelstrup and Foley (1993) also compiled and analyzed existing water-quality and biological information from the Lower St. Croix National Scenic Riverway, finding concentrations of Cd, Cu, Fe, Mn, and Hg (element symbols are defined in the Methods section) in the water generally were greater in the lower reaches of the St. Croix River relative to the upstream reaches.
A better understanding of variability in trace element concentrations across the St. Croix River Basin, and links to anthropogenic sources could help water-resource managers understand threats to ecosystem health, which include the potential for toxicity to aquatic organisms, including endangered species. The U.S. Geological Survey (USGS), in cooperation with the National Park Service, undertook this study to better understand trace element levels and sources in the St. Croix River Basin.
The purpose of this report is to present the results of elemental chemical analyses of streambed sediment collected during June 19-August 3, 2000 at 30 stream sites in the St. Croix River Basin. Sites upstream and downstream of the larger cities in the basin were sampled to assess potential anthropogenic contributions of trace elements to the St. Croix Riverway. Concentrations of selected trace elements are compared to sediment-quality criteria and guidelines, providing an indication of potential toxicity concerns, which has implications for water-resource quality in the St. Croix River Basin.
Naturally occurring minerals in the St. Croix River Basin are one source of trace elements. Aluminosilicate minerals contain trace levels of many elements. Bedrock in the northeastern portion of the basin and in the Lake Superior Basin, where glacial sedimentary deposits in the St. Croix River Basin originate, is enriched with native copper (Nicholson and others, 1992).
In the absence of anthropogenic inputs of elements, it would be expected that the elemental composition of sediments reflects the composition of the parent geologic material (mineral phase), as modified by weathering and physicochemical reactions, such as dissolution, and precipitation. Some trace elements, such as mercury, typically have exceedingly low concentrations in the mineral phase (Smith, 1990); however, atmospheric transport and deposition from remote natural sources (volcanoes, and mercury volatilization from the oceans) have resulted in elevated concentrations of mercury in pre-industrial strata of lake sediments relative to local geologic sources (Brigham, 1992). The same processes probably contributed to elevated mercury concentrations in surficial soils and organic-enriched streambed sediments in the St. Croix River Basin, even prior to human settlement.
Humans have affected aquatic trace element cycling through direct discharges into streams and atmospheric emissions, which are deposited at varying distances from the source. Nriagu and Pacyna (1988) reported that human activities result in substantial inputs to the environment of Sb, As, Cd, Cr, Cu, Pb, Hg, Mn, Ni, Se, Sn, V, and Zn (element symbols defined in Methods section). Fossil fuel combustion, incineration of municipal and hospital wastes, metal smelting, and other emissions of metals have resulted in widespread releases of numerous elements into the atmosphere. Lead has been widely used as a gasoline additive, although it is no longer used in the United States. Industrial point-source inputs of trace elements to streams have been substantially reduced in recent decades in the United States (see, for example, Anderson and Perry, 1999). Acid mine drainage, and other mining-associated releases, continue to affect streams in certain localities, though with very few instances in the Upper Midwestern states. With reductions in direct discharges into streams, increased attention has focused on non-point sources, such as emissions to the atmosphere.
Proximity to anthropogenic sources has a substantially effect on trace element concentrations in streambed and lake sediments (Iskander and Keeny, 1974; Kroening and others, 2000; Rice, 1999; Verta and others, 1989). Particulate or highly particle-reactive forms of elements tend to be transported relatively short distances before they are deposited (Klein and Russel, 1973; Nriagu and others, 1998). In contrast, gaseous species, such as elemental mercury, can become widely distributed in the atmosphere (Fitzgerald and others, 1998).
Numerous studies point to the wide scale, combined loads of anthropogenic sources of trace elements to the aquatic environment. Sediment cores from lakes distant from point-source contamination have been used to assess changes in atmospheric deposition of trace metals over the past 150-200 years (pre-industrial to near present). Pronounced up-core (from preindustrial to recent sediment strata) increases in mercury concentrations have frequently been reported (Eng-strom and Swain, 1997; Meili, 1991; Swain and others, 1992; Verta and others, 1989). Other elements that often are enriched in more recent, near-surface lake sediments include Cd, Cu, Pb, and Zn (El-Daoushy, 1986; Iskander and Keeny, 1974; Kemp and others, 1978; Nriagu and others, 1979; Verta and others, 1989).
Kroening and others (2000) determined that streambed sediments in urban areas in the Upper Mississippi River Basin generally had higher levels of Sb, As, Cd, Cu, Pb, Hg, Ni, and Zn than non-urban areas. This reflects a national pattern of greater trace element concentrations (Cd, Hg, Cu, Pb, Zn, and to a lesser extent, Cr and Ni) in streambed sediments from urban compared to non-urban streams (Rice, 1999). Urban areas may include numerous sources of trace element emissions to the atmosphere, some portion of which fallout locally, the remainder being transported and deposited regionally or globally. Urban runoff and point discharges also may contribute trace elements to streams.
Kroening and others (2000) analyzed surficial streambed sediments and fish livers at four sites in the St. Croix River Basin. Results showed that concentrations of Cu, Pb, Hg, and Zn in streambed sediments were greater at the most downstream site sampled (located near Hudson, Wisconsin) relative to the other three sites sampled in less developed areas of the St. Croix River Basin.
Randy Ferrin and Barry Long (National Park Service), and Sharon Kroening (U.S. Geological Survey) were instrumental in initiating this study, and Randy Ferrin provided valuable input into preparation of this report. I also thank the following U.S. Geological Survey personnel: Luke Stuewe provided careful assistance with sampling and sample processing, Laurel Woodruff and Tim Cowdery offered expertise on geology of the region; Barbara Scudder and Bernard (Bernie) Lenz offered helpful review suggestions.
The St. Croix River Basin drains 20,176 km2 in Minnesota and Wisconsin, and includes greater than 15 major tributaries to the Namekagon and St. Croix Rivers (fig. 1). The Namekagon River and St. Croix River form the St. Croix Scenic Riverway, a National Scenic Riverway of the National Park Service.
| Figure 1. Location of the St. Croix River Basin, surficial geology, ecoregions, the St. Croix National Scenic Riverway, and sampling sites. |
Noncalcareous Superior Lobe glacial sediments overlie much of the St. Croix River Basin (fig. 1). Calcareous Des Moines Lobe glacial sediments of the Grantsburg sublobe overrode the Superior Lobe sediments in the western portion of the basin (Wright, 1972) (fig. 1), resulting in substantial mixing of Des Moines Lobe and Superior Lobe sediments.
The St. Croix Scenic Riverway is a relatively well-preserved and biologically diverse aquatic resource. More than 60 State and Federally listed endangered and threatened species have been found in the Riverway. Among the 40 species of freshwater mussels found, 15 are State listed and two are Federally listed as endangered. There are 10 species of fish that are State listed as threatened or endangered.
Although the water quality generally is considered to be good in the upper St. Croix and Namekagon Rivers, activities in the tributaries and their watersheds are affecting the overall health of the St. Croix Scenic Riverway (Troelstrup and others, 1993). Due to its proximity to the Minneapolis/St. Paul metropolitan area, there are continued increases in use and developmental pressure from population growth in adjacent counties.
Streambed sediments were sampled at 30 sites in the St. Croix River Basin (fig. 1, table 1), following techniques of Shelton and Capel (1994). Samples were collected at 9 sites on the St. Croix River, 3 sites on the Namekagon River, and 18 sites on other tributaries. For tributary streams, typically two sites were sampled—one upstream and one downstream of a city. Samples were collected during June 19-August 3, 2000 during low-flow conditions.
Table 1. Streambed sediment sampling sites,
St. Croix River Basin, 2000. [Minn., Minnesota; Wisc., Wisconsin. Sites were sampled once between June 19–August 3, 2000.] |
||||
| Map number (figure 1) |
Site identification number |
Site name | Latitude (ddmmss) |
Longitude (ddmmss) |
|---|---|---|---|---|
| 1 |
05331775 | St. Croix River near Woodland Corner, Wisc. | 46°07'00" | 92°07'53" |
| 2 |
05331833 | Namekagon River at Leonards, Wisc. | 46°10'18" | 91°19'50" |
| 3 |
05331873 | Namekagon River above Spring Lake Creek near Hayward, Wisc. | 45°59'22" | 91°30'33" |
| 4 |
05333400 | Namekagon River near Woodland Corner, Wisc. | 46°05'02" | 92°06'47" |
| 5 |
05333500 | St. Croix River near Danbury, Wisc. | 46°04'28" | 92°14'50" |
| 6 |
05333983 | Yellow River above Spooner, Wisc. | 45°49'17" | 91°51'58" |
| 7 |
05334270 | Yellow River near Spooner, Wisc. | 45°50'12" | 92°02'03" |
| 8 |
05336500 | Kettle River near Sandstone, Minn. | 46°10'48" | 92°49'58" |
| 9 |
05337020 | Kettle River at State Highwy 48, near Hinckley, Minn. | 46°00'40" | 92°50'24" |
| 10 |
05337150 | Snake River near McGrath, Minn. | 46°13'05" | 93°14'25" |
| 11 |
05338400 | Snake River at Pine City, Minn. | 45°49'53" | 92°57'58" |
| 12 |
05338955 | Wood River at North Williams Road near Grantsburg, Wisc. | 45°47'07" | 92°37'52" |
| 13 |
05338975 | Wood River at State Highway 70 near Grantsburg, Wisc. | 45°46'22" | 92°42'29" |
| 14 |
05339705 | Rush Creek at Co. Rd. 54 above Rush City, Minn. | 45°40'56" | 92°58'52" |
| 15 |
05339715 | Rush Creek at Co. Rd. 5 below Rush City, Minn. | 45°40'21" | 92°54'42" |
| 16 |
05340170 | North Branch Sunrise River near North Branch, Minn. | 45°30'48" | 92°59'47" |
| 17 |
05340195 | Sunrise River at Sunrise, Minn. | 45°32'48" | 92°51'22" |
| 18 |
05340420 | St. Croix River at Nevers Dam Site, near Wolf Creek, Wisc. | 45°32'13" | 92°43'27" |
| 19 |
05340500 | St. Croix River at St. Croix Falls, Wisc. | 45°24'25" | 92°38'49" |
| 20 |
05340552 | St. Croix River at Franconia, Minn. | 45°21'40" | 92°42'04" |
| 21 |
053405543 | Osceola Creek at 248th St. above Osceola, Wisc. | 45°19'22" | 92°40'17" |
| 22 |
0534055489 | Osceola Creek below Cascade Falls at Osceola, Wisc. | 45°19'16" | 92°42'28" |
| 23 |
05341111 | Apple River at Co. Rd. H near Balsam Lake, Wisc. | 45°26'16" | 92°21'58" |
| 24 |
05341499 | Apple River in Apple Falls flowage below Somerset, Wisc. | 45°08'40" | 92°41'38" |
| 25 |
05341550 | St. Croix River at Stillwater, Minn. | 45°03'22" | 92°48'11" |
| 26 |
05341560 | St. Croix River at Lakeland Shores, Minn. | 44°57'18" | 92°45'10" |
| 27 |
05341770 | St. Croix River at Afton, Minn. | 44°54'00" | 92°46'45" |
| 28 |
05341875 | Kinnickinnic River at 100th St. near River Falls, Wisc. | 44°53'28" | 92°35'54" |
| 29 |
05342000 | Kinnickinnic River near River Falls, Wisc. | 44°49'50" | 92°44'00" |
| 30 |
05344490 | St. Croix River at Prescott, Wisc. | 44°44'57" | 92°48'16" |
At each wadeable site, samples were collected and composited from 5-10 depositional zones where fine-grained sediments accumulate during low flow. At each depositional zone, several samples of the upper 2-5 cm of streambed sediment were scooped by hand with a 5-cm diameter Teflon tube, and placed into a Teflon bowl for compositing. At deeper, nonwadeable sites, a large (23 x 23 cm) stainless steel Ekman dredge (Wild-life Supply Co.) was deployed from a canoe to retrieve streambed sediment. Surficial streambed sediments were sampled from the dredge using a Teflon tube, taking care to avoid sampling material that was within 1 cm of the walls of the dredge.
Each sample was thoroughly stirred. A portion of each sample was sieved using a new, acid-cleaned 63-µm (micrometer) Nylon mesh sieve (Gilson Co.) held in an acrylic sieve frame (Gilson Co.), using less than 250 mL (milliliters) of native water (stream water taken from the sampling site) to aid in sieving. After settling, supernatant water was decanted, and samples were transferred to clean paper containers suitable for soil and sediment trace element samples (Star Paper Tube, Inc., Salt Lake City, Utah), oven-dried at 50°C, and shipped for laboratory analysis. Both sieved and unsieved sediments were retained for archive purposes.
Samples were submitted to the USGS-Central Region Mineral Resource Team, then transferred to XRAL Laboratory Services (Don Mills, Ontario, Canada) for analysis. A list of analytes, and a summary of analytical procedures, is given in table 2. Further details of analytical procedures are given in the Appendix. The USGS-Central Region Mineral Resource Team routinely assesses quality-assurance procedures of contract laboratories (Murphy and others, 1997).
| Table 2. Elements analyzed, NWIS code, detectable
ranges (by method), and summary of field-replicate-sample analyses, St.
Croix River Basin, 2000 [Concentrations in micrograms per gram dry sediment, unless listed as % (percent, by mass, of dry sediment). A. ICP-AES (inductively coupled plasma—atomic emission spectroscopy, following sample digestion using hydrochloric, nitric, perchloric, and hydrofluoric acids at low temperature. (Carbon is analyzed by separate methods, as described in Appendix 1.) B. ICP-AES, following digestion using hydrochloric acid and hydrogen peroxide digestion, then extraction with diisobutyl ketone). C. Atomic absorption spectroscopy (for mercury, follows digestion and reduction to elemental mercury; for arsenic and selenium, follows hydride generation). Where analytes were determined by multiple methods, the preferred method (indicating data used for data analysis herein) is indicated by boldface. NWIS, U.S. Geological Survey National Water Information System data base; N>LOQ, number of pairs of replicate samples, where the sample concentration exceeds the limit of quantitation (five times the lower reporting limit); RPD, absolute value of the relative percent difference, calculated only for replicate pairs for which the sample concentration exceeded the limit of quantitation; >=, greater than or equal to; --, not applicable.] |
|||||||||
| Detectable range | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Element | Symbol | NWIS code | N›L0Q | Minimum RPD |
Maximum RPD |
Mean RPD |
|||
| A | B | C | |||||||
| Aluminum | Al | 34790 | 0.005-50% | -- | -- | 5 | 0.44 | 3.8 | 1.9 |
| Antimony | Sb | 34795 | -- | 1.0-6,000 -- | 0 | -- | -- | -- | |
| Arsenic | As | 34800 | 10-50,000 |
1.0-6,000 | 0.6-20 | 5 | 1.16 | 29.9 | 11.2 |
| Barium | Ba | 34805 | 1-35,000 | -- | -- | 5 | 0.19 | 5.8 | 2.6 |
| Beryllium | Be | 34810 | 1-5,000 | -- | -- | 0 | -- | -- | -- |
| Bismuth | Bi | 34816 | 10-50,000 | 1.0-6,000 | -- | 0 | -- | -- | -- |
| Cadmium | Cd | 34825 | 2-25,000 | 0.05-500 | -- | 5 | 3.2 | 55.3 | 15.2 |
| Calcium | Ca | 34830 | 0.005-50% | -- | -- | 5 | 0.24 | 3.6 | 2.3 |
| Cerium | Ce | 34835 | 5-50,000 | -- | -- | 5 | 0.0 | 28.0 | 10.4 |
| Chromium | Cr | 34840 | 2-50,000 | -- | -- | 5 | 0.0 | 33.8 | 17.0 |
| Cobalt | Co | 34845 | 2-25,000 | -- | -- | 2 | 0.0 | 7.4 | 3.7 |
| Copper | Cu | 34850 | 2-15,000 | 0.05-500 | -- | 5 | 3.0 | 23.2 | 10.3 |
| Europium | Eu | 34855 | 2-5,000 | -- | -- | 0 | -- | -- | -- |
| Gallium | Ga | 34860 | 4-50,000 | -- | -- | 0 | -- | -- | -- |
| Gold | Au | 34870 | 8-50,000 | 0.10-1,500 | -- | 1 | 52.6 | 52.6 | 52.6 |
| Holmium | Ho | 34875 | 4-5,000 | -- | -- | 0 | -- | -- | -- |
| Iron | Fe | 34880 | 0.02-25% | -- | -- | 5 | 0.0 | 5.9 | 2.4 |
| Lanthanum | La | 34885 | 2-50,000 | -- | -- | 5 | 0.0 | 13.3 | 3.4 |
| Lead | Pb | 34890 | 4-50,000 | 1.0-6,000 | -- | 5 | 0.0 | 12.5 | 8.1 |
| Lithium | Li | 34895 | 2-50,000 | -- | -- | 4 | 0.0 | 8.7 | 4.9 |
| Magnesium | Mg | 34900 | 0.005-5% | -- | -- | 5 | 1.8 | 4.8 | 2.8 |
| Manganese | Mn | 34905 | 4-50,000 | -- | -- | 5 | 0.0 | 12.1 | 3.2 |
| Mercury | Hg | 34910 | -- | -- | >=0.02 | 4 | 0.0 | 9.5 | 3.4 |
| Molybdenum | Mo | 34915 | 2-50,000 | 0.10-900 | -- | 2 | 0.0 | 15.4 | 7.7 |
| Neodymium | Nd | 34920 | 9-50,000 | -- | -- | 0 | -- | -- | -- |
| Nickel | Ni | 34925 | 3-50,000 | -- | -- | 5 | 0.0 | 11.1 | 5.2 |
| Niobium | Nb | 34930 | 4-50,000 | -- | -- | 0 | -- | -- | -- |
| Phosphorus | P | 34935 | 0.005-50% | -- | -- | 5 | 3.5 | 6.9 | 5.2 |
| Potassium | K | 34940 | 0.01-50% | -- | -- | 5 | 0.0 | 5.2 | 2.0 |
| Scandium | Sc | 34945 | 2-50,000 | -- | -- | 0 | -- | -- | -- |
| Selenium | Se | 34950 | -- | -- | 0.2-4 | 5 | 0.0 | 9.5 | 3.4 |
| Silver | Ag | 34955 | 2-1000 | 0.08-400 | 3 | 85.7 | 159 | 133 | |
| Sodium | Na | 34960 | 0.005-50% | -- | -- | 5 | 0.0 | 6.1 | 3.3 |
| Strontium | Sr | 34965 | 2-15,000 | -- | -- | 5 | 1.4 | 5.0 | 2.5 |
| Tantalum | Ta | 34975 | 40-50,000 | -- | -- | 0 | -- | -- | -- |
| Thorium | Th | 34980 | 6-50,000 | -- | -- | 0 | -- | -- | -- |
| Tin | Sn | 34985 | 5-50,000 | -- | -- | 0 | -- | -- | -- |
| Titanium | Ti | 49274 | 0.005-25% | -- | -- | 5 | 1.7 | 4.3 | 2.8 |
| Uranium | U | 35000 | 100-100,000 | -- | -- | 0 | -- | -- | -- |
| Vanadium | V | 35005 | 2-30,000 | -- | -- | 5 | 0.0 | 4.8 | 3.1 |
| Ytterbium | Yb | 35015 | 1-5,000 | -- | -- | 0 | -- | -- | -- |
| Yttrium | Y | 35010 | 2-25,000 | -- | -- | 5 | 0.0 | 5.7 | 3.1 |
| Zinc | Zn | 35020 | 2-15,000 | 0.05-500 | -- | 5 | 0.97 | 4.0 | 2.3 |
| Carbon, total | TC | 49267 | 0.05-30% | -- | -- | 5 | 0.0 | 4.6 | 2.4 |
| Carbonate carbon | CO3 | 49269 | 0.01-50% | -- | -- | 5 | 0.0 | 35.3 | 13.7 |
Reproducibility was assessed by submitting five sets of split samples for analysis. Of the elements that were detected at greater than 5 times the lower limit of detection, most of the replicate samples had absolute values of relative percent differences (RPDs) below 15 percent (table 2), the stated laboratory criterion for acceptable data (Appendix). Exceptions (RPDs exceeding 15 percent) are one sample pair each for As, Cd, Cu, Au, and Mo; two samples for Ce and carbonate carbon; and 3 samples for Cr and Ag. Ag was the only commonly detected element that yielded poor precision for all replicates (table 2).
Aluminosilicate minerals are key components of fine (clay and silt-sized) inorganic sediments. These minerals are largely composed of lithophil elements (after Gold-schmidt's classification of the elements, Goldschmidt, 1954; Sposito, 1989). Many of the elements measured for this study are lithophils. Within an area of relatively uniform geologic source material, and in the absence of significant contamination, concentrations of lithophil elements would be expected to correlate with Al content (denoted by r[Al], where r is the Pearson correlation coefficient). Among the lithophils analyzed in this study, many correlate strongly (Ba, Ce, La, Li, Mg, Nb, K, Sc, Na, Sr, Th, and Ti; r[Al] > 0.65, p<0.0001) or modestly (Nd, Yb, and Y; 0.35< r[Al] <0.64, 0.0001<p<0.05) with Al content (fig. 2; table 3). The abundance of aluminosilicates or clay minerals may be controlling the concentrations of many lithophilic elements in streambed sediments in the St. Croix River Basin, particularly those elements with strong correlation to Al content.
|
| Figure 2. Concentrations of selected elements compared to aluminum concentrations in streambed sediments from the St Croix River Basin, 2000. |
| Table 3. Summary of element
concentrations from streambed sediment samples (less than 63 micrometer
fraction) and correlation
with aluminum content, St. Croix River Basin, 2000. [Concentrations in micrograms per gram dry sediment, unless listed as % (percent, by mass, of dry sediment); <, less than; N>LRL, number of samples with concentrations greater than the lower reporting limit; N, number of samples; r(Al), Pearson correlation coefficient of element concentration with aluminum content (boldfaced values are significant at the 95 percent confidence level (p<0.05)); --, not calculated.] |
|||||||
| Element | Symbol | Minimum | Median | Maximum | N>LRL | N | r(Al) |
|---|---|---|---|---|---|---|---|
| Aluminum | Al |
1.220% |
4.06% |
5.31% |
30 |
30 |
-- |
| Antimony | Sb |
<1.0 |
<1.0 |
<1.0 |
0 |
30 |
-- |
| Arsenic | As |
3.5 |
11.4 |
33.4 |
30 |
30 |
-0.105 |
| Barium | Ba |
266 |
458 |
686 |
30 |
30 |
0.694 |
| Beryllium | Be |
<1.0 |
<1.0 |
2.0 |
1 |
30 |
-- |
| Bismuth | Bi |
<1.000 |
<1.00 |
<1.00 |
0 |
30 |
-- |
| Cadmium | Cd |
0.34 |
0.54 |
0.86 |
30 |
30 |
0.336 |
| Calcium | Ca |
0.905% |
1.32% |
2.86% |
30 |
30 |
0.108 |
| Carbon, Organic | OC |
3.84% |
7.75% |
21.7% |
30 |
30 |
-0.939 |
| Carbon, Total | CT |
4.42% |
7.95% |
21.8% |
30 |
30 |
-0.940 |
| Carbonate carbon | CO3 |
0.04% |
0.13% |
0.90% |
30 |
30 |
0.239 |
| Cerium | Ce |
32 |
59 |
82 |
30 |
30 |
0.844 |
| Chromium | Cr |
35 |
52.5 |
71.0 |
30 |
30 |
-.245 |
| Cobalt | Co |
5 |
11 |
22 |
30 |
30 |
0.556 |
| Copper | Cu |
17 |
28.5 |
48 |
30 |
30 |
-0.171 |
| Europium | Eu |
<2 |
<2 |
2 |
5 |
30 |
-- |
| Gallium | Ga |
7 |
13 |
25 |
30 |
30 |
0.570 |
| Gold | Au |
<0.1 |
0.3 |
1.5 |
22 |
29 |
0.347 |
| Holmium | Ho |
<4 |
<4 |
7 |
9 |
30 |
-- |
| Iron | Fe |
2.13% |
4.73% |
9.97% |
30 |
30 |
0.295 |
| Lanthanum | La |
14 |
25 |
32 |
30 |
30 |
0.900 |
| Lead | Pb |
13 |
19.5 |
59 |
30 |
30 |
-.103 |
| Lithium | Li |
9 |
17 |
24 |
30 |
30 |
0.835 |
| Magnesium | Mg |
0.395% |
0.73% |
1.22% |
30 |
30 |
0.664 |
| Manganese | Mn |
582 |
1900 |
8450 |
30 |
30 |
-.028 |
| Mercury | Hg |
0.06 |
0.12 |
0.49 |
30 |
30 |
-.027 |
| Molybdenum | Mo |
<0.1 |
0.3 |
1.4 |
19 |
30 |
-.244 |
| Neodymium | Nd |
10 |
23 |
30 |
30 |
30 |
0.636 |
| Nickel | Ni |
17 |
30 |
38 |
30 |
30 |
0.578 |
| Niobium | Nb |
7 |
15.5 |
25 |
30 |
30 |
0.727 |
| Phosphorus | P |
0.11% |
0.16% |
0.35% |
30 |
30 |
-.144 |
| Potassium | K |
0.42% |
1.35% |
2.04% |
30 |
30 |
0.831 |
| Scandium | Sc |
4 |
9 |
13 |
30 |
30 |
0.741 |
| Selenium | Se |
0.6 |
1.45 |
10.4 |
30 |
30 |
-.572 |
| Silver | Ag |
<0.08 |
0.4 |
1.9 |
29 |
30 |
-.479 |
| Sodium | Na |
0.275% |
0.69% |
1.05% |
30 |
30 |
0.784 |
| Strontium | Sr |
49 |
98 |
134 |
30 |
30 |
0.811 |
| Tantalum | Ta |
<40 |
<40 |
<40 |
0 |
30 |
-- |
| Thorium | Th |
<6 |
7 |
11 |
24 |
30 |
0.672 |
| Tin | Sn |
<50 |
<50 |
<50 |
0 |
30 |
-- |
| Titanium | Ti |
0.171% |
0.33% |
0.57% |
30 |
30 |
0.666 |
| Uranium | U |
<100 |
<100 |
<100 |
0 |
30 |
-- |
| Vanadium | V |
47 |
95.5 |
161 |
30 |
30 |
0.041 |
| Ytterbium | Yb |
1 |
2 |
3 |
30 |
30 |
0.418 |
| Yttrium | Y |
11 |
19 |
26 |
30 |
30 |
0.627 |
| Zinc | Zn |
51 |
97 |
151 |
30 |
30 |
0.388 |
The strong, positive correlations of Na and Sr with Al content in the St. Croix River Basin are in contrast to the Red River of the North Basin (RRB), for which Na showed no significant relation to Al, and Sr was negatively correlated with Al (Brigham and others, 1998). These two lithophil elements were considered indicative of evaporite minerals in the RRB, but are more likely to be associated with aluminosilicates in the St. Croix River Basin, due to their strong correlation with Al and other lithophilic elements.
Co, Ga, Ni, and Zn showed modest correlations with Al content in the St. Croix River Basin. These elements are chalcophils (ore loving) (Gold-schmidt, 1954), and tend to associate with sulfide mineral phases in comparison to lithophils, which associate with silicates. Co and Ni also are siderphils (iron loving) and lithophils. Depending on temperature and oxidation state during rock formation, these two elements may associate with sulfides, iron ores, or silicate materials. Furthermore, processes such as sulfide inclusion, isomorphic substitution, and adsorption to mineral surfaces may result in their association of chalcophil or siderophil elements with aluminosilicate minerals (Sposito, 1989).
Organic carbon dilutes Al content, as indicated by its strong negative correlation with Al (fig. 2; table 3). Se and Ag correlate negatively with Al content, which suggests that the source of Se and Ag is not aluminosilicates. Also, Se and Ag were the only two elements that correlated positively with organic carbon (r= 0.66 and 0.50, respectively), which suggests that these elements are enriched in surficial soils and sediments, possibly due to atmospheric deposition from industrial emissions. Anthropogenic emissions to the atmosphere of Se (Nriagu and Pacyna, 1988) and Ag (Agency for Toxic Substances and Disease Registry, 1990, chapter 5) are known to occur, which supports the hypothesis that concentrations of these elements are enhanced at the earth's surface, relative to deeper, organic-carbon-poor sediments. Given this hypothesis, it is surprising that other atmospherically transported elements, such as Pb and Hg, did not correlate significantly with organic carbon.
Cr and V (lithophils) and Fe (a siderophil; Goldschmidt, 1954) had no significant relation to Al content in St. Croix River Basin streambed sediments, but strongly correlated to Al content in RRB sediments (for V, this correlation was strong when samples from areas of the RRB with Cretaceous marine shales were excluded) (Brigham and others, 1998). One possible explanation for the lack of correlation of Cr, Fe, and V to Al content is that the St. Croix River Basin is over-lain by mixed glacial deposits from two distinct glacial lobes (calcareous Des Moines Lobe and siliceous Superior Lobe sediments), whereas the RRB is predominantly overlain by deposits from one glacial lobe (Des Moines Lobe).
Other elements that do not correlate (r[Al] <0.35) with Al content are As, Cd, Ca, Cu, Au, Pb, Mn, Hg, Mo, P, and carbonate carbon. Strong correlations between As and concentrations of Fe, Mn, and P were observed. The strong correlation of As (a siderophil/chalcophil element; Gold-schmidt, 1954) with Fe suggests a geologic source of As. Concentrations of As, Cd, Cu, Pb, and Hg may be slightly elevated in the St. Croix River Basin due to anthropogenic sources because of poor correlation with elements indicative of almuminosilicates.
Many of the trace elements in streambed sediment appear to be closely associated with parent mineral sources, with possible low-level anthropogenic contributions of several elements at some sites. In comparison to streams draining highly urbanized parts of the Minneapolis-St. Paul metropolitan area (Kroening and others, 2000), and compared to other highly urbanized stream basins across the Nation (Callender and Rice, 2000; Rice, 1999), streams in the St. Croix River Basin generally had low levels of the trace elements that are commonly elevated from anthropogenic sources (Cd, Cr, Cu, Pb, Hg, Ni, and Zn). Emission of trace elements to the atmosphere, and subsequent transport and deposition, contribute low levels of several elements to relatively pristine areas (see Previous Investigations section).
In five cases, there were higher concentrations of selected trace elements at a site downstream of a city or urban area, compared to a site on the same stream, upstream of the urban area. These cases are discussed below (fig. 3; table 4). There does not appear to be large anthropogenic contributions of trace elements—above the background geochemical and atmospherically deposited levels—in the streams sampled for this study. There is no basinwide pattern of increased trace element concentrations downstream of urban areas. Possible causes of elevated trace element concentrations are speculative. Rigorous determination of specific sources was beyond the scope of this study.
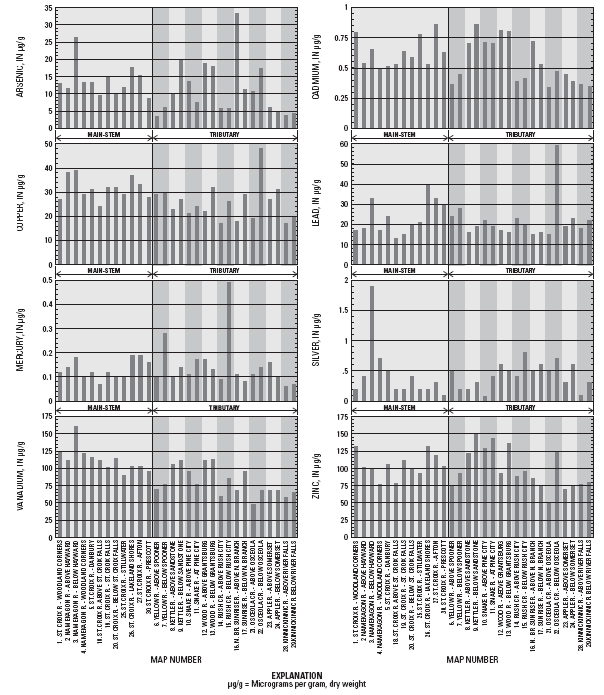 |
| Figure 3. Concentrations of selected elements in streambed sediments of the St. Croix River Basin, 2000. (Map number corresponds to Figure 1 and Table 1). |
| Table 4. Concentrations
of selected trace elements detected in streambed sediments in the St. Croix
River Basin, 2000 and
reference values for comparison. [<, less than; --, no guidelines established; Minn., Minnesota; Wisc., Wisconsin; ISQG (Interim Sediment Quality Guidelines) and PEL (Probable Effects Level) are Canadian reference values (Canadian Council of Mininsters of the Environment, 1995, 1999); Effects Range-Low (ERL) and Effects Range-Median (ERM) values are given by Long and others (1995). Consensus-based Probable Effects Concentration (PEC) is given by MacDonald and others (2000). All concentrations are in micrograms per gram dry sediment. The highest three concentrations for each element are in boldface. Element symbols are defined in table 2.) |
|||||||||||||
| Element concentration | |||||||||||||
| Map number (figure 1) |
Site Identification number |
Site name | |||||||||||
| As | Cd | Cr | Cu | Pb | Hg | Ni | Se | Ag | V | Zn | |||
| 1 | 05331775 | St. Croix River near Woodland Corner, Wisc. | 13.0 | 0.79 | 61 | 27 | 17 | 0.12 | 36 | 1.4 | 0.20 | 123 | 132 |
| 2 | 05331833 | Namekagon River at Leonards, Wisc. | 11.5 | 0.54 | 68 | 38 | 18 | 0.14 | 22 | 2.7 | 0.40 | 111 | 101 |
| 3 | 05331873 | Namekagon River above Spring Lake Creek near Hayward, Wisc. | 26.2 | 0.65 | 68 | 39 | 33 | 0.18 | 23 | 3.5 | 1.90 | 161 | 98 |
| 4 | 05333400 | Namekagon River near Woodland Corner, Wisc. | 13.3 | 0.49 | 40 | 29 | 17 | 0.10 | 25 | 1.6 | 0.70 | 121 | 77 |
| 5 | 05333500 | St. Croix River near Danbury, Wisc. | 13.3 | 0.51 | 71 | 31 | 24 | 0.12 | 33 | 1.6 | 0.50 | 115 | 105 |
| 6 | 05333983 | Yellow River above Spooner, Wisc. | 3.5 | 0.36 | 66 | 29 | 24 | 0.10 | 22 | 3.4 | 0.50 | 69 | 73 |
| 7 | 05334270 | Yellow River near Spooner, Wisc. | 6.2 | 0.45 | 68 | 30 | 28 | 0.28 | 22 | 3.4 | 0.20 | 76 | 92 |
| 8 | 05336500 | Kettle River near Sandstone, Minn. | 9.9 | 0.70 | 47 | 23 | 16 | 0.10 | 32 | 0.6 | 0.20 | 106 | 122 |
| 9 | 05337020 | Kettle River at State Hwy 48, near Hinckley, Minn. | 19.8 | 0.86 | 55 | 27 | 19 | 0.14 | 38 | 0.6 | 0.30 | 111 | 151 |
| 10 | 05337150 | Snake River near McGrath, Minn. | 13.5 | 0.71 | 51 | 21 | 22 | 0.11 | 28 | 0.6 | <0.08 | 96 | 128 |
| 11 | 05338400 | Snake River at Pine City, Minn. | 7.5 | 0.70 | 53 | 24 | 19 | 0.17 | 30 | 0.9 | 0.40 | 76 | 143 |
| 12 | 05338955 | Wood River at North Williams Road near Grantsburg, Wisc | 18.7 | 0.81 | 50 | 22 | 17 | 0.17 | 35 | 1.2 | 0.60 | 111 | 92 |
| 13 | 05338975 | Wood River at State Highway 70 near Grantsburg, Wisc. | 18.0 | 0.80 | 44 | 32 | 16 | 0.13 | 34 | 1.0 | 0.50 | 113 | 136 |
| 14 | 05339705 | Rush Creek at Co. Rd. 54 above Rush City, Minn. | 5.8 | 0.39 | 35 | 17 | 23 | 0.09 | 22 | 0.9 | 0.40 | 59 | 88 |
| 15 | 05339715 | Rush Creek at Co. Rd. 5 below Rush City, Minn. | 5.7 | 0.41 | 45 | 26 | 20 | 0.49 | 25 | 1.3 | 0.80 | 85 | 96 |
| 16 | 05340170 | North Branch Sunrise River near North Branch, Minn. | 33.4 | 0.72 | 48 | 18 | 15 | 0.11 | 33 | 2.3 | 0.50 | 68 | 86 |
| 17 | 05340195 | Sunrise River at Sunrise, Minn. | 11.3 | 0.53 | 55 | 29 | 16 | 0.08 | 32 | 1.1 | 0.60 | 95 | 75 |
| 18 | 05340420 | St. Croix R. at Nevers Dam Site, near Wolf Creek, Wisc. | 9.6 | 0.53 | 68 | 24 | 13 | 0.07 | 30 | 1.4 | 0.20 | 111 | 78 |
| 19 | 05340500 | St. Croix River at St. Croix Falls, Wisc. | 15.1 | 0.64 | 58 | 32 | 15 | 0.12 | 36 | 1.3 | 0.20 | 101 | 111 |
| 20 | 05340552 | St. Croix River at Franconia, Minn. | 10.2 | 0.59 | 66 | 32 | 20 | 0.10 | 35 | 0.9 | 0.40 | 114 | 100 |
| 21 | 053405543 | Osceola Creek at 248th St. above Osceola, Wisc. | 10.7 | 0.34 | 41 | 19 | 15 | 0.11 | 17 | 10.4 | 0.50 | 47 | 51 |
| 22 | 0534055489 | Osceola Creek below Cascade Falls at Osceola, Wisc. | 17.4 | 0.47 | 51 | 48 | 59 | 0.14 | 28 | 2.8 | 0.70 | 68 | 124 |
| 23 | 05341111 | Apple River at Co. Rd. H near Balsam Lake, Wisc. | 6.2 | 0.45 | 39 | 27 | 19 | 0.16 | 24 | 1.5 | 0.30 | 68 | 69 |
| 24 | 05341499 | Apple River in Apple Falls flowage below Somerset, Wisc. | 4.6 | 0.39 | 57 | 31 | 23 | 0.10 | 26 | 2.1 | 0.60 | 68 | 74 |
| 25 | 05341550 | St. Croix River at Stillwater, Minn. | 11.8 | 0.78 | 43 | 29 | 21 | 0.10 | 32 | 1.0 | 0.20 | 89 | 93 |
| 26 | 05341560 | St. Croix River at Lakeland Shores, Minn. | 17.5 | 0.53 | 58 | 37 | 40 | 0.19 | 35 | 1.5 | 0.20 | 103 | 131 |
| 27 | 05341770 | St. Croix River at Afton, Minn. | 15.4 | 0.86 | 52 | 33 | 33 | 0.19 | 30 | 1.5 | 0.30 | 102 | 119 |
| 28 | 05341875 | Kinnickinnic River at 100th St. near River Falls, Wisc. | 3.8 | 0.36 | 46 | 17 | 18 | 0.06 | 23 | 3.2 | 0.10 | 58 | 77 |
| 29 | 05342000 | Kinnickinnic River near River Falls, Wisc. | 4.4 | 0.35 | 42 | 20 | 22 | 0.07 | 24 | 1.8 | 0.30 | 65 | 79 |
| 30 | 05344490 | St. Croix River at Prescott, Wisc. | 8.6 | 0.63 | 55 | 28 | 30 | 0.16 | 32 | 1.1 | 0.10 | 95 | 103 |
| Median, all samples | 11.4 | 0.54 | 52.5 | 28.5 | 19.5 | 0.12 | 30 | 1.45 | 0.40 | 95.5 | 97 | ||
| Interim Sediment Quality Guideline (ISQG) | 5.9 | 0.6 | 37.5 | 35.7 | 35 | 0.17 | -- | -- | -- | -- | 123 | ||
| Probable Effect Level (PEL) | 17.0 | 3.5 | 90 | 197 | 91. 3 | 0.486 | -- | -- | -- | 315 | |||
| Effects range--Low (ERL) | 8.2 | 1.2 | 81 | 34 | 46.7 | 0.15 | 20.9 -- | 1.0 | -- | 150 | |||
| Effects range--Median (ERM) | 70 | 9.6 | 370 | 270 | 218 | 0.71 | 51.6 | -- | 3.7 | -- | 410 | ||
| Consensus-based Probable Effects Concentration (PEC) | 33 | 4.98 | 111 | 149 | 128 | 1.06 | 48.6 -- | -- | -- | 459 | |||
As, Pb, and Ag concentrations were substantially higher in the Namekagon River downstream of Hayward, Wisconsin (site 3) than upstream (site 2). In addition, Cd, Cu, and Hg concentrations were slightly higher downstream. Site 3 had high concentrations of As, Cr, Cu, Pb, and Se compared to most other sites in the basin, and had the maximum concentrations of Ag and V. The Ag concentration at site 3 is twice as high as the next highest Ag concentration. This elevation in trace element concentrations may result from anthropogenic sources in the Hayward area. Concentrations of As, Cr, and Cu were moderately higher at site 3 than at most other sites. Several tributary sites in northwestern Wisconsin had comparable concentrations of Cr.
The co-occurrence of relatively high concentrations of As, Cr, and Cu suggests the possibility of contamination from chromated copper arsenate, a preservative for green-treated lumber. This type of wood is commonly used for small boat docks and yard landscape projects. Also, field notes from this sampling site indicated presence of woody debris, including sawn wood, on the stream bottom. However, the evidence for contamination from chromated copper arsenate is not overwhelming.
The V concentrations is somewhat elevated downstream of Hayward compared to other sites, which could indicate inputs from particulate emissions from the combustion of petroleum. Another potential source of trace elements to site 3 is the use of fly-ash cinders to seal cracks in stoplogs in the Hayward dam. Although some fly ashes have high trace element concentrations compared to typical uncontaminated sediments, metal concentrations for the fly ash used in the Hayward dam1 generally are lower than those determined in streambed sediments. The trace element concentrations at site 3 are not so highly elevated that one cannot rule out the possibility that these concentrations simply reflect natural differences in sediment composition in the upper Namekagon River, compared to the rest of the St. Croix River Basin.
1 As reported in an interagency memo entitled "Plan to comply with article 401 of the Hayward License (FERC Project No. 2417) fly ash/cinders monitoring plan."
The Pb concentration increased by nearly a factor of two from the St. Croix River at Stillwater, Minnesota (site 25) to Lakeland Shores, Minnesota (site 26). Elevated Pb concentrations at site 26 could be attributed to past use of leaded gasoline additives because site 26 is most prone to the effects of urbanization and automobile traffic (it is downstream of the Stillwater, Minnesota-Hudson, Wisconsin area, and approximately 0.5 km downstream of Interstate Highway 94). Pb concentrations decrease at the next two downstream sites on the St. Croix River.
Concentrations of all of the elements shown in figure 3 were modestly higher at the downstream site on the Kettle River (site 9) than the upstream site (site 8), despite nearly identical concentrations of Al, carbonate carbon, and organic carbon at these two sites. Anthropogenic influences from Sandstone and Hinckley may account for the elevation in trace element concentrations.
The Hg concentration downstream of Rush City (site 15) was nearly 5 times higher than at the upstream site (site 14), and was the highest Hg concentration determined in this study. No other trace element concentrations appeared to be anomalous at site 15, indicating that the source of elevated Hg was specific to that element. Given that the Hg concentration at this site was substantially higher than at all other sites in the basin, it is likely that there is a local source of Hg to Rush Creek, in addition to the lower-level atmospheric deposition of Hg that occurs in mid-continental North America (Swain and others, 1992). Possible sources of Hg include outflow from wastewater treatment facilities, leakage from landfills, or other industrial or waste disposal operations.
Concentrations of Cu and Pb, and to a lesser extent Cd, As, Ni, and V were substantially higher downstream of Osceola, Wisconsin (site 22) than upstream (site 21). It is possible that concentrations at site 22 reflect anthropogenic sources of these elements from runoff from Osceola, Wisconsin. However, it also is likely that the upstream-downstream differences in concentrations of Cu, Pb, and Cd may be attributable to the geologic setting of the sites. Downstream of Osceola, Osceola Creek bisects a dolomitic sandstone, which may strongly influence the sediment chemistry. The concentration of carbonate carbon (a component of dolomite) was high at site 22 (0.81 percent), compared to site 21 (0.10 percent). Trace substitution of Cu and Pb into the carbonate mineral matrix, or cooccurrence of Cu- and Pb-containing minerals, is a possible explanation for the inter-site differences observed.
Numerous studies have assessed the effects of potentially toxic sediment-bound trace elements on aquatic biota. From these studies, various reference values or guidelines have been published, and are useful for comparison purposes, and to indicate potential areas of concern.
The Canadian Council of Ministers of the Environment (1995, 1999) have developed Interim Sediment Quality Guidelines (ISQGs) and Probable Effect Levels (PELs) for several elements. ISQGs are concentrations below which toxic effects are infrequently observed, and are intentionally set low, to be protective for a broad range of Canadian waters (Canadian Council of Mininsters of the Environment, 1995). In contrast, the PEL is defined as the level (concentration) above which adverse effects are predicted to occur frequently, and is higher than the ISQGs. Because site-specific sediment properties are known to influence toxicity, these guidelines are not absolute indications of toxicity, but are useful for comparison to measured concentrations. As new toxicity data are gathered, ISQGs will be replaced by Sediment Quality Guidelines (SQGs) when the data substantiate establishment of more permanent guidelines.
Based on an extensive literature review, Long and others (1995) established Effects Range Low (ERL) and Effects Range Median (ERM) values for numerous contaminants, including potentially toxic trace elements, in marine and estuarine sediments. Adverse biological effects due to the contaminant of interest are rarely observed at concentrations below the ERL. Concentrations between the ERL and ERM represent conditions where adverse effects may occasionally occur, and concentrations equal to and exceeding the ERM represent conditions where adverse effects occur frequently. It is not known how well metal toxicity in marine and estuarine environments compares to freshwater environments.
More recent guidelines are the Probable-Effects Concentration (PEC, or consensus-based PEC) guidelines for freshwater sediments (MacDonald and others, 2000). Conceptually, the PEC values are similar to ERM and PEL values described above. That is, PECs "may be used to identify sediments that are likely to be toxic to sediment-dwelling organisms" (MacDonald and others, 2000, p. 29).
Element concentrations frequently exceeded the lowest reference values—the ISQG and ERL values—in the St. Croix River Basin (table 4). All elements for which an ISQG exists exceeded the ISQG value at one or more sites. All elements for which an ERL exists, except Cd and Cr, exceeded the ERL at one or more sites.
The higher reference values—above which there are frequent literature reports of adverse effects on aquatic biota—were rarely exceeded. The PEL for As was exceeded in 7 of 30 samples. Hg exceeded its PEL in only 1 sample—site 15, downstream of Rush City, Minnesota. No other trace element concentrations exceeded PEL values; ERM values were not exceeded in any samples. Concentrations were less than one-half of the ERMs for all trace elements, except in 1 sample for Hg, 1 sample for Ag, and 19 samples for Ni. One concentration exceeded a PEC—the As concentration at site 16 (33.4 µg/g) was slightly higher than the PEC for As (33 µg/g).
This study examined trace element concentrations in streambed sediment samples collected in the St. Croix River Basin during June 19–August 3, 2000. Streambed sediments were sampled at 30 sites in the St. Croix River Basin, sieved to <63 micrometers, and analyzed for a suite of chemical elements.
Possible occurrences of low-level trace element concentrations that could be attributed to anthropogenic sources include: (1) elevated concentrations of As, Pb, Ag, and to a lesser extent Cd, Cu, and Hg in the Namekagon River downstream of Hayward, Wisconsin; (2) elevated Pb concentrations in the St. Croix River downstream of the Still-water-Hudson, Wisconsin region; (3) slightly elevated concentrations of As, Cd, Cu, Pb, Hg, Ag, V, and Zn in the Kettle River downstream of Sandstone-Hinckley, Minnesota; and (4) substantially elevated Hg concentrations in Rush Creek downstream of Rush City, Minnesota. Elevated concentrations of Cu, Pb, and to a lesser extent Cd, in Osceola Creek, downstream of Osceola, Wisconsin may be due to anthropogenic sources or contributions from bedrock that differs geochemically from sediments farther upstream. There does not appear to be large anthropogenic contributions of trace elements—above the background geochemical and atmospherically deposited concentrations—in the streams sampled for this study, nor is there a basinwide pattern of increased trace element concentrations downstream of urban areas. Many trace elements appear to be associated with parent mineral sources, with possible low-level anthropogenic contributions of several elements at some sites.
To assess implications for resource quality, several sets of reference values or sediment-quality guidelines were used for comparison. These include the Interim Sediment Quality Guidelines (ISQG) and Probable Effects Level (PEL); the Effects Range-Low and Effects Range-Median (ERM); and the concensus-based Probable-Effects Concentration (PEC). ISQGs and PELs are concentrations below which toxic effects are rarely observed. Concentrations frequently exceeded these low reference values. All elements for which an ISQG exists exceeded the ISQG at one or more sites, and all elements for which an ERL exists, except Cd and Cr exceeded the ERL at one or more sites.
Concentrations exceeding PEL, ERM, or PEC values have been associated with frequent adverse effects in aquatic biota. Concentrations of potentially toxic trace elements in the St. Croix River Basin generally were lower than PEL values and always lower than ERM values. Exceptions were As, which exceeded its PEL in 7 of 30 samples, and Hg, which exceeded its PEL in only 1 sample. Only one concentration exceeded a PEC: the As concentration at the North Branch Sunrise River site (33.4 µg/g) was slightly higher than the PEC for As (33 µg/g).
Agency for Toxic Substances and Disease Registry (ATSDR), 1990, Toxicological profile for silver: U.S. Department of Health and Human Services, Public Health Service., variously paged, accessed on August 20, 2001, URL: http://www.atsdr.cdc.gov/toxprofiles/tp146.html
Anderson, J., and Perry, J., 1999, Comparison of temporal trends in ambient and compliance trace element and PCB data in pool 2 of the Mississippi River, USA, 1985-95: Environmental Management, v. 24, no. 4, p. 497-507.
Brigham, M.E., 1992, Accumulation of mercury in Minnesota, Alaska, and Wisconsin lakes: Minneapolis, Minnesota, University of Minnesota, Master's Thesis, 216 p.
Brigham, M.E., Goldstein, R.M., and Tornes, L.H., 1998, Trace elements and organic chemicals in stream-bottom sediments and fish tissues, Red River of the North Basin, Minnesota, North Dakota, and South Dakota 1992-95: U.S. Geological Survey Water-Resources Investigations Report 97-4043, 32 p.
Callender, E., and Rice, K.C., 2000, The urban environmental gradient—Anthropogenic influences on the spatial and temporal distributions of lead and zinc in sediments: Environmental Science and Technology, v. 34, p. 232-238.
Canadian Council of Mininsters of the Environment, 1995, Protocol for the derivation of Canadian sediment quality guidelines for the protection of aquatic life: Environment Canada CCME EPC-98E, 38 p.
Canadian Council of Ministers of the Environment, 1999, Canadian sediment quality guidelines for the protection of aquatic life, 5 p.: accessed on April 11, 2001, at URL http://www.ec.gc.ca/
El-Daoushy, F., 1986, Scandinavian limnochronology of sediments and heavy metals: Hydrobiologia, v. 143, p. 267-276.
Engstrom, D.R., and Swain, E.B., 1997, Recent declines in atmospheric mercury deposition in the Upper Midwest: Environmental Science and Technology, v. 31, no. 4, p. 960-967.
Fenneman, N.M., 1938, Physiography of eastern United States: New York, McGraw-Hill Book Company, 691 p.
Fitzgerald, W.F., Engstrom, D.R., Mason, R.P., and Nater, E.A., 1998, The case for atmospheric mercury contamination in remote areas: Environmental Science and Technology, v. 32, no. 1, p. 1-7.
Goldschmidt, V.M. (Alex Muir, ed.), 1954, Geochemistry: London, Oxford at the Clarendon Press, 730 p.
Hoobs, H.C., and Goebel, J.E., 1982, Geologic map of Minnesota—Quaternary geology: Minnesota Geological Survey State Map Series S-1, 1 sheet.
Holmberg, K.L., Perry, J., Ferrin, R.S., and Sharrow, D.L., 1997, Water resources plan—St. Croix National Scenic Riverway, Minnesota and Wisconsin: National Park Service, 155 p.
Iskander, I.K., and Keeny, D.R., 1974, Concentration of heavy metals in sediment cores from selected Wisconsin lakes: Environmental Science and Technology, v. 8, p. 165-170.
Kemp, A.L.W., Williams, J.D.H., Thomas, R.L., and Gregory, M.L., 1978, Impact of man's activities on the chemical compositions of the sediment of Lakes Superior and Huron: Water, Air and Soil Pollution, v. 10, p. 381-402.
Klein, D.H., and Russel, P., 1973, Heavy metals—Fallout around a power plant: Environmental Science and Technology, v. 7, no. 4, p. 357-358.
Kroening, S.E., Fallon, J.D., and Lee, K.E., 2000, Water-quality assessment of part of the Upper Mississippi River Basin, Minnesota and Wisconsin—Trace elements in streambed sediment and fish livers, 1995-96: U.S. Geological Survey Water-Resources Investigations Report 00-4031 26 p.
Long, E.R., MacDonald, D.D., Smith, S.L., and Calder, F.D., 1995, Incidence of adverse biological effects within ranges of chemical concentrations in marine and estuarine sediments: Environmental Management, v. 19, no. 1, p. 81-97.
MacDonald, D.D., Ingersoll, C.G., and Berger, T.A., 2000, Development and evaluation of consensus-based sediment quality guidelines for freshwater ecosystems: Archives of Environmental Contamination and Toxicology, v. 39, p. 20-31.
Meili, M., 1991, Fluxes, pools, and turnover of mercury in Swedish forest lakes: Water, Air, and Soil Pollution, v. 56, p. 719-727.
Murphy, C., Briggs, P., Adrian, B., Wilson, S., Hageman, P., and Theodorakos, P., 1997, Chain of custody—Recommendations for acceptance and analysis of evidentiary geochemical samples: U.S. Geological Survey Circular 1138, 26 p.
Nicholson, S.W., Cannon, W.F., and Schulz, K.J., 1992, Metallogeny of the Midcontinent rift system of North America: Precambrian Research, v. 58, p. 355-386.
Nriagu, J.O., Kemp, A.L.W., Wong, H.K.T., and Harper, N., 1979, Sedimentary record of heavy metal pollution in Lake Erie: Geochimica et Cosmochimica, v. 16 43, p. 247-258.
Nriagu, J.O., and Pacyna, J.M., 1988, Quantitative assessment of worldwide contamination of air, water and soils by trace metals: Nature, v. 333, no. 12, p. 134-139.
Nriagu, J.O., Wong, H.K.T., Lawson, G., and Daniel, P., 1998, Saturation of ecosystems with toxic metals in Sudbury Basin, Ontario, Canada: Science of the Total Environment, v. 223, no. 2/3, p. 99-117.
Olcott, P.G., 1992, Ground-water atlas of the United States, Segment 9, Iowa, Michigan, Minnesota, and Wisconsin: U.S. Geological Survey Hydrologic Investigations Atlas 730-J, 31 p., scales 1:250,000 and 1:500,000.
Omernik, J.M. and Gallant, A.L., 1988, Ecoregions of the Upper Midwest States: United States Environmental Protection Agency, EPA/600/3-88/037, 56 p.
Rice, K.C., 1999, Trace-element concentrations in streambed sediment across the conterminous United States: Environmental Science and Technology, v. 33, p. 2499-2504.
Shelton, L.R., and Capel, P.D., 1994, Guidelines for collecting and processing samples of stream bed sediment for analysis of trace elements and organic contaminants for the National Water-Quality Assessment Program: U.S. Geological Survey Open-File Report 94-458, 20 p.
Smith, E.A., 1990, Mercury geochemistry of four watersheds in northeast Minnesota: Duluth, Minnesota, University of Minnesota-Duluth, Master's Thesis, 107 p.
Sposito, G., 1989, The chemistry of soils: New York, Oxford University Press, 277 p.
Swain, E.B., Engstrom, D.R., Brigham, M.E., Henning, T.A., and Brezonik, P.L., 1992, Increasing rates of atmospheric mercury deposition in midcontinental North America: Science, v. 257, p. 784-787.
Troelstrup, N.H., Jr., and Foley, J.L., 1993, Examination of mussel growth and shell chemistry as indicators of water quality within the Lower St. Croix National Scenic Riverway: Minnesota-Wisconsin Boundary Area Commission Deliverables B5, 70 p.
Troelstrup, N.H., Jr., Foley, J.L., and Perry, J.A., 1993, Changing patterns of water quality and biology within the Lower St. Croix National Scenic Riverway: Minnesota-Wisconsin Boundary Area Commission B2 and B6, 162 p.
Verta, M., Tolonen, K., and Simola, H., 1989, History of heavy metal pollution in Finland as recorded by lake sediments: Science of the Total Environment, v. 87/88, p. 1-18.
Wright, H.E., Jr., 1972, Quaternary history of Minnesota, in Sims, P.K., and Morey, G.B., eds., Geology of Minnesota—A centennial volume: Minneapolis, Minnesota, Minnesota Geological Survey, p. 515-547.
Analytical procedures are described in this appendix. The following information was obtained from the U.S. Geological Survey, Central Region Mineral Resource Team web site (URL: https://miner-als.cr.usgs.gov/intranet/chem/labmethods.html) accessed on March 8, 2001. Method numbers are those used by the Central Region Mineral Resource Team.
Method 1. Forty-element ICPAES. Forty major, minor, and trace elements are determined in geological materials by inductively coupled plasma-atomic emission spectrometry (ICP-AES). The sample is decomposed using a mixture of hydrochloric, nitric, perchloric, and hydrofluoric acids at low temperature. The digested sample is aspirated into the ICP-AES discharge where the elemental emission signal is measured simultaneously for the 40 elements. Calibration is performed by standardizing with digested rock reference materials and a series of multi-element solution standards. Data are deemed acceptable if recovery for all 40 elements is ±15% at five times the Lower Limit of Determination (LOD) and the calculated Relative Standard Deviation (RSD) of duplicate samples is no greater than 15%.
Method 2. Low-level ICP analysis of 10 elements, following digestion and extraction. A widely used extraction technique (Aliquat/MIBK) has been modified and adapted for use with inductively coupled plasma-atomic emission spectrometry for the analysis of geologic materials. Hydro-chloric acid-hydrogen peroxide solubilizes metals that are not tightly bound in the silicate lattice of rocks, soils, and stream sediments. The metals are extracted by a 10% Aliquat-336/diisobutyl ketone solution as organic halides. The separated organic phase is pneumatically aspirated into a multichannel ICP instrument where the concentrations of the extracted metals (Ag, As, Au, Bi, Cd, Cu, Mo, Pb, Sb, and Zn) are determined simultaneously. It is important to note that this procedure is a partial digestion, and depending on an element's availability, results may be biased low when compared to other methods of analyses.
Depending on the type of sample, there may be a significant discrepancy between the proposed value and the laboratory value. This is primarily due to the availability of the metal in the sample. Since this is a partial digestion, those metals tightly bound in highly resistant minerals will not be extracted. Data will be deemed acceptable if recovery for all 10 elements is ±20% on spikes at five times the LOD and the calculated percent RSD of duplicate samples is no greater than 15%.
Method 3a. Total Carbon. Total carbon in geologic materials is determined by the use of an automated carbon analyzer. A weighed sample is combusted in an oxygen atmosphere at 1370°C to oxidize carbon to carbon dioxide. Moisture and dust are removed and the carbon dioxide gas is measured by a solid state infrared detector. The operating range for total carbon is from 0.05% to about 30%. Data will be deemed acceptable if recovery of total carbon is ±15% at five times the LOD and the calculated percent RSD of duplicate samples is no greater the 15%.
Method 3b. Carbonate Carbon. Carbonate carbon in geologic materials is determined as carbon dioxide by coulometric titration. The sample is treated with hot 2N perchloric acid and the evolved carbon dioxide is passed into a cell containing a solution of monoethanolamine. The carbon dioxide, quantitatively absorbed by the monoethanolamine, is coulometrically titrated using platinum and silver/potassium-iodide electrodes. The lower reporting limit is 0.01% carbon dioxide and samples containing up to 50% carbon dioxide may be analyzed. Sample size is adjusted from 0.5 g for the range 0.01–5% carbon dioxide, 0.1 g for the range 5– 10% carbon dioxide, and 0.02 g for greater than 10% carbon dioxide. Data will be deemed acceptable if recovery for carbonate carbon is ±15% at five times the lower LOD and the calculated percent RSD of duplicate samples is no greater than 15%.
Method 8. Mercury. Mercury is determined by weighing out 0.1 g of sample and digesting with a mixture of sulfuric acid, nitric acid, 5% potassium permanganate, and 5% potassium peroxydisulfate in a water bath for one hour. The excess of potassium permanganate is reduced with hydroxylamine sulfate solution and then Hg (II) is reduced with stannous chloride. The Hg vapor is separated and measured using a LEEMAN PS200 automated mercury analyzer. The method offers a lower reporting limit of 0.02 ppm mercury in solid-phase samples. Samples exceeding the working range of 0.02–.8 ppm mercury require dilution. Data will be deemed acceptable if recovery of mercury is ±20% at five times the LOD and the calculated percent RSD of duplicate samples is no greater than 20%.
Method 9. Arsenic (other elements not determined for this study, using this method). Arsenic, antimony, and thallium are determined by weighing 0.1 g of sample into a zirconium crucible. Approximately 0.75 g of sodium peroxide is added and mixed. The mixture is heated in a muffle furnace set at 750°C for four minutes. The sample is cooled then 15 mL of water and 5 ml of concentrated HCl is added. The mixture is shaken and 0.25 mL of an ascorbic acid KI solution is added, then diluted with 20% HCl and let to stand overnight. Arsenic and selenium are then measured using hydride generation atomic absorption spectrometry.
The optimum concentration ranges without sample dilution for these elements in various solid phase sample media are: As-0.6 ppm–20 ppm, and Sb-0.6 ppm –20 ppm. Data will be deemed acceptable if recovery of As and Sb is ±20% at five times the LOD and the calculated percent RSD of duplicate samples is no greater than 20%.
Method 10. Selenium. Selenium is determined by weighing 0.25 g of sample into a test tube, adding a mixture of nitric, hydrofluoric and perchloric acids and heating. After the solution is cooled hydrochloric and nitric acid are added, heated again and cooled. The samples are diluted and analyzed using hydride generation atomic absorption spectrometry. Data for selenium will be deemed acceptable if recovery of that element is ±20% at five times the LOD and the calculated percent RSD of duplicate samples is no greater than 20%.
This report is available online in Portable Document Format (PDF). If you do not have the Adobe Acrobat PDF Reader, it is available for free download from Adobe Systems Incorporated.
Download the report (PDF, 1.1 MB)
Document Accessibility: Adobe Systems Incorporated has information about PDFs and the visually impaired. This information provides tools to help make PDF files accessible. These tools convert Adobe PDF documents into HTML or ASCII text, which then can be read by a number of common screen-reading programs that synthesize text as audible speech. In addition, an accessible version of Acrobat Reader 5.0 for Windows (English only), which contains support for screen readers, is available. These tools and the accessible reader may be obtained free from Adobe at Adobe Access.
| AccessibilityFOIAPrivacyPolicies and Notices | |
 |
|