Water Quality in the Western Lake Michigan Drainages, Wisconsin and Michigan, 1992-95
Table of contents || Previous section || Next section || Glossary
Many naturally occurring chemicals are present in streams and ground water. It is important for water and land managers to understand the natural water chemistry in order to assess the effects of human activities on water quality.
 (100,063 bytes)
(100,063 bytes)
The chemical composition of natural water is primarily derived from the dissolution of minerals in surficial deposits and bedrock.
Dissolved constituents in rivers and ground water include the major cations (calcium, magnesium, sodium, potassium), major anions (bicarbonate, chloride, and sulfate), nutrients, trace elements (including iron and manganese), and radionuclides (uranium, radium, and radon).
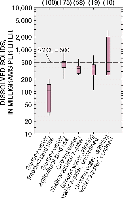
The most common ions in most waters in the study area are calcium, magnesium, and bicarbonate. These ions provide many of the dissolved solids in study-area waters. Median dissolved-solids concentrations in surface waters range from about 50 milligrams per liter (mg/L) in forested parts of the study area to about 500 mg/L in agricultural basins. Median concentrations of dissolved solids in ground water range from about 250 mg/L in shallow alluvial aquifers to more than 2,500 mg/L in the drinking-water aquifer where it is confined by the Sinnipee confining layer (Richards and others, in press; Saad, 1996 and 1997).
Concentrations of dissolved solids exceeded the USEPA secondary maximum contaminant level (SMCL) of 500 mg/L in 22 percent of surficial aquifer wells and 17 percent of deep drinking water wells sampled (Saad, 1996 and 1997).
 (78,071 bytes)
(78,071 bytes)
Runoff in agricultural areas may contribute significant suspended solids to stream water, resulting in streams with a muddy appearance.
 (6,964 bytes)
(6,964 bytes)
Dissolved solids concentrations are variable in the study area. Highest concentrations are found in the confined part of the drinking water aquifer, which may increase treatment costs. SMCL is secondary maximum contaminant level.
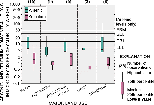
Concentrations of arsenic and selenium in streambed sediment are variable in the study area. Median arsenic concentrations are highest in the forested land use area and are correlated with the igneous/metamorphic bedrock. Arsenic concentrations exceeded the concentrations that 95 percent of the benthic biota can tolerate, the Lowest Effect Level (LEL), at 48 percent of sites sampled.
 (10,865 bytes)
(10,865 bytes)
Forested land use was related to high concentrations of arsenic and selenium in streambed sediment (Scudder and others, 1997). The igneous/metamorphic bedrock and sand and gravel surficial deposits that are found in most of the forested areas may be a source for these elements in the study area. Ontario Ministry of Environment and Energy values (Persaud and others, 1993) are shown for the Lowest Effect Level (LEL) and Severe Effect Level (SEL). Sediment effect criteria from Ingersoll and others (1996) for Effects Range Low (ERL) and Effects Range Median (ERM) are for arsenic only.
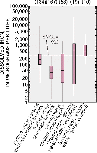
Concentrations of iron in ground water varied from near zero to 23,000 micrograms per liter (µg/L). Concentrations of dissolved iron exceeded the USEPA SMCL of 0.3 mg/L in 38 percent of samples collected from drinking-water wells and 10 percent of shallow alluvial wells (Saad, 1996 and 1997). The source of the iron in the drinking water aquifer is probably the minerals in the Cambrian/Ordovician aquifer geologic material. The source of iron in the shallow alluvial aquifers may be the minerals in the alluvial materials derived from the igneous and metamorphic bedrock or possibly from supplemental iron that is in some fertilizers.
 (8,479 bytes)
(8,479 bytes)
Dissolved iron concentrations are variable in the study area. SMCL is secondary maximum contaminant level.
Radon, which can cause lung cancer, is a radioactive, odorless, and chemically inert gas that occurs naturally in the air, soil, and ground water. Radon is a decay product of radium, which in turn is a decay product of uranium. Rocks break down mechanically and chemically to form sediments that contain various amounts of uranium, depending on the source rocks.
Radon in ground water affects radon concentrations in indoor air in homes because it escapes to the indoor air as people use water. Open water-distribution systems allow ground water to aerate and radon to escape. In small, closed water-distribution systems with short transit times, radon cannot escape from the system; therefore it escapes into the indoor air. Research suggests that ingestion of water with high radon concentrations also may pose risks, although these risks are believed to be much lower than those from inhalation of radon.
 (3,704 bytes)
(3,704 bytes)
Radon, a known carcinogen, exceeds the previously proposed USEPA standard of 300 picocuries per liter in many deep wells.
Until late 1996, the USEPA had proposed an MCL of 300 picocuries per liter (pCi/L) for radon in drinking water. However, this proposed maximum contaminant level (MCL) was withdrawn by USEPA for further evaluation; thus, no proposed MCL currently exists. Radon concentrations measured during this study were larger than the previously proposed MCL in 66 percent of the drinking-water aquifer wells (Saad, 1996).
 (13,393 bytes)
(13,393 bytes)
Radon, a known carcinogen, exceeds the previously proposed USEPA standard of 300 picocuries per liter in many deep wells.
Radon concentrations greater than 300 pCi/L were detected in a variety of geohydrologic environments throughout most of the study area, with the exception of the southwest (Saad, 1996). Elevated radon concentrations do not appear to be related to a particular geologic formation or land use.
U.S. Geological Survey Circular 1156
Table of contents || Previous section || Next section || Glossary
Suggested citation:
C. A. Peters, D. M. Robertson, D. A. Saad, D. J. Sullivan, B. C. Scudder, F. A. Fitzpatrick, K. D. Richards, J. S. Stewart, S. A. Fitzgerald, and B. N. Lenz, 1998, U.S. Geological Survey Circular 1156, on line at <URL: https://water.usgs.gov/pubs/circ1156>, updated June 11, 1998