O
Effects of Nitrogen Use on the Quality of Ground Water and Surface Water
Nitrate contamination of ground water and surface water in the United States is widespread because nitrate is very mobile in the environment. Nitrate concentrations are increasing in much of the Nation's water, but they are particularly high in ground water in the midcontinent region of the United States. Two principal chemical reactions are important to the fate of nitrogen in water: (1) fertilizer ammonium can be nitrified to form nitrate, which is very mobile as a dissolved constituent in shallow ground water, and (2) nitrate can be denitrified to produce nitrogen gas in the presence of chemically reducing conditions if a source of dissolved organic carbon is available.
 |
High concentrations of nitrate can contribute to excessive growth of aquatic plants, depletion of oxygen, fishkills, and general degradation of aquatic habitats. For example, a study of Waquoit Bay in Massachusetts linked the decline in eelgrass beds since 1950 to a progressive increase in nitrate input due to expansion of domestic septic-field developments in the drainage basin (Figure O-1). Loss of eelgrass is a concern because this aquatic plant stabilizes sediment and provides ideal habitat for juvenile fish and other fauna in coastal bays and estuaries. Larger nitrate concentrations supported algal growth that caused turbidity and shading, which contributed to the decline of eelgrass.
Significant denitrification has been found to take place at locations where oxygen is absent or present at very low concentrations and where suitable electron-donor compounds, such as organic carbon, are available. Such locations include the interface of aquifers with silt and clay confining beds and along riparian zones adjacent to streams. For example, in a study on the eastern shore of Maryland, nitrogen isotopes and other environmental tracers were used to show that the degree of denitrification that took place depended on the extent of interaction between ground-water and the chemically reducing sediments near or below the bottom of the Aquia Formation. Two drainage basins were studied: Morgan Creek and Chesterville Branch (Figure O-2). Ground-water discharging beneath both streams had similar nitrate concentration when recharged. Significant denitrification took place in the Morgan Creek basin where a large fraction of local ground-water flow passed through the reducing sediments, which are present at shallow depths (3 to 10 feet) in this area. Evidence for the denitrification included decreases in nitrate concentrations along the flow path to Morgan Creek and enrichment of the 15N isotope. Much less denitrification took place in the Chesterville Branch basin because the top of the reducing sediments are deeper (10 to 20 feet) in this area and a smaller fraction of ground-water flow passed through those sediments.
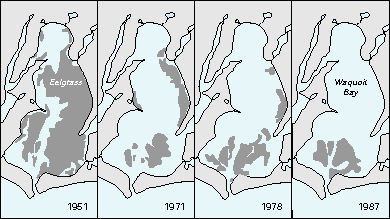 |
Figure O-1. The areal extent of eelgrass in Waquoit Bay, Massachusetts, decreased markedly between 1951 and 1987 because of increased inputs of nitrogen related to domestic septic-field developments. (Modified from Valiela, I., Foreman, K., LaMontagne, M., Hersh, D., Costa, J., Peckol, P., DeMeo-Andeson, B., D'Avanzo, C., Babione, M., Sham, C.H., Brawley, J., and Lajtha, K., 1992, Couplings of watersheds and coastal waters-Sources and consequences of nutrient enrichment in Waquoit Bay, Massachusetts: Estuaries, v. 15, no. 4, p. 433-457.) (Reprinted by permission of the Estuarine Research Federation.)
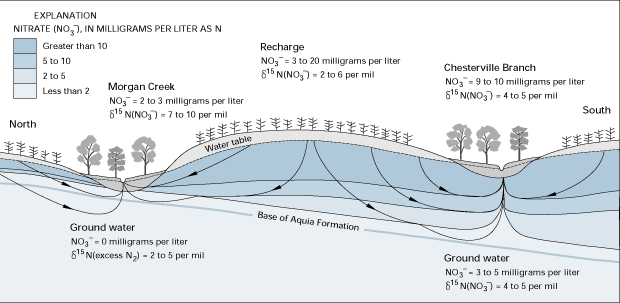 |
Figure O-2. Denitrification had a greater effect on ground water discharging to Morgan Creek than to Chesterville Branch in Maryland because a larger fraction of the local flow system discharging to Morgan Creek penetrated the reduced calcareous sediments near or below the bottom of the Aquia Formation than the flow system associated with the Chesterville Branch. (Modified from Bolke, J.K., and Denver, J.M., 1995, Combined use of ground-water dating, chemical, and isotopic analyses to resolve the history and fate of nitrate contamination in two agricultural watersheds, Atlantic coastal plain, Maryland: Water Resources Research, v. 31, no. 9, p. 2319-2337.)