S
Effects of Atmospheric Deposition on the Quality of Ground Water and Surface Water
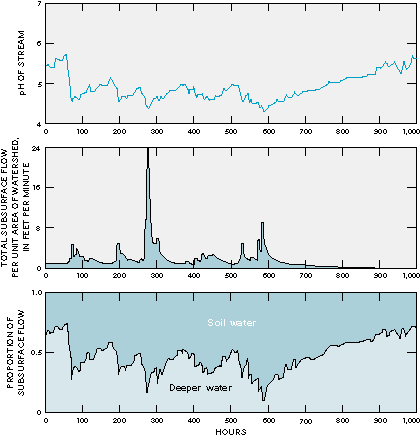
In areas where soils have little capacity to buffer acids in water, acidic precipitation can be a problem because the infiltrating acidic water can increase the solubility of metals, which results in the flushing of high concentrations of dissolved metals into surface water. Increased concentrations of naturally occurring metals such as aluminum may be toxic to aquatic organisms. Studies of watersheds have indicated that the length of subsurface flow paths has an effect on the degree to which acidic water is buffered by flow through the subsurface. For example, studies of watersheds in England have indicated that acidity was higher in streams during storms when more of the sub- surface flow moved through the soil rather than through the deeper flow paths (Figure S-1). Moreover, in a study of the effects of acid precipitation on lakes in the Adirondack Mountains of New York, the length of time that water was in contact with deep subsurface materials was the most important factor affecting acidity because contact time determined the amount of buffering that could take place (Figure S-2).
Figure S-1: Acidity is higher (pH is lower) in streams when most of the flow is contributed by shallow soil water because the water has had less time to be neutralized by contact with minerals compared to water that has traversed deeper flow paths. (Modified from Robson, A., Beven, K.J., and Neal, C., 1992, Towards identifying sources of subsurface flow-A comparison of components identified by a physically based runoff model and those determined by chemical mixing techniques: Hydrological Processes, v. 6, p. 199-214.) (Reprinted with permission from John Wiley & Sons Limited.)
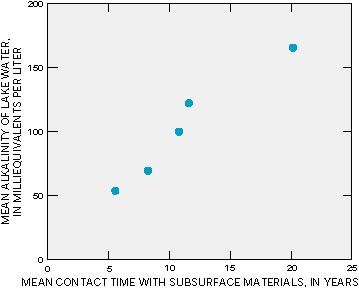 |
Figure S-2: The longer water is in contact with deep subsurface materials in a watershed, the higher the alkalinity in lakes receiving that water. (Modified from Wolock, D.M., Hornberger, G.M., Beven, K.J., and Campbell, W.G., 1989, The relationship of catchment topography and soil hydraulic characteristics to lake alkalinity in the northeastern United States: Water Resources Research, v. 25, p. 829-837.)