Sustainability of Ground-Water Resources--Circular 1186
Previous chapters have discussed quantities of water recharging, flowing through, and discharging from the ground-water system and quantities of water stored in the system. This brief discussion of ground-water quality adds a further dimension to ground-water resource sustainability; namely, the question of the suitability of ground water for different uses. Various measures of water quality such as taste and odor, microbial content, and dissolved concentrations of naturally occurring and manufactured chemical constituents define the suitability of water for different uses.
The availability of ground water and the suitability of its quality for different uses are inextricably intertwined. To take an extreme example, salt brines having very high dissolved-solids concentrations occur adjacent to fresh ground water almost everywhere. Although brines represent huge volumes of ground water in storage, these brines are not included in most inventories of available ground water because of their inherent unsuitability for almost all uses. Ground waters having somewhat lower dissolved-solids concentrations may be suitable for some uses but not for others. For example, some cattle can tolerate a higher dissolved-solids concentration in their drinking water than humans.
A key consideration in managing a ground-water resource is its vulnerability to sources of contamination that are located primarily at and near the land surface. Because of generally low ground-water velocities, once contaminants have reached the water table, their movement to nearby surface-water discharge areas or to deeper parts of the ground-water-flow system is slow. For the same reason, once parts of an aquifer are contaminated, the time required for a return to better water-quality conditions as a result of natural processes is long, even after the original sources of contamination are no longer active. Ground-water-quality remediation projects generally are very expensive and commonly are only partly successful. In some settings, steep gradients caused by ground-water pumping can greatly increase the rate at which contaminants move to deeper ground water. For these reasons, State and Federal environmental agencies seek to protect the ground-water resource by stressing regulatory efforts to prevent ground-water contamination.
The availability of ground water and the suitability of its quality for different uses are inextricably intertwined.
Contamination of ground water is not always a result of the introduction of contaminants by human activities. Possible natural contaminants include trace elements such as arsenic and selenium, radionuclides such as radon, and high concentrations of commonly occurring dissolved constituents.
The first two subsections below involve two of the most significant linkages in hydrology--the land-surface/water-table connection and the ground-water/surface-water connection. The third subsection, saltwater intrusion, involves movement of naturally occurring, highly saline ground water into parts of adjacent aquifers that contain less saline water. Pumping of the less saline (commonly potable) ground water generally causes this movement.
In principle, virtually any human activity at and near the land surface can be a source of contaminants to ground water as long as water and possibly other fluids move from the land surface to the water table. Sources of chemicals introduced to ground water in this way include fertilizers, manure, and pesticides applied to agricultural lands; landfills; industrial-discharge lagoons; leaking gasoline storage tanks; cesspools and septic tanks; and domestically used chemicals. These sources commonly are classified as "point" or "nonpoint" sources. For example, industrial lagoons, leaking storage tanks, and landfills are considered to be point sources. A considerable number of these sources and associated contaminant plumes have undergone intensive studies followed by a remediation program. Many of the chemicals associated with point sources--for example, gasoline and other manufactured organic chemicals--even at very low concentrations, render the contaminated ground water highly undesirable or useless as a source of domestic or public supply.
Croplands are a primary nonpoint source of contamination because of their large areal extent and significant applications per unit area of possible contaminants (fertilizers and pesticides) to ground water. Irrigated agriculture also has noteworthy effects on ground-water (and surface-water) quality. Increased areal recharge from excess irrigation-water applications results in the potential for increased transport of contaminants from the land surface to ground water. Also, a marked increase in dissolved-solids concentrations in soil water and shallow ground water may result from evaporation of irrigation water during delivery of the water to the crops and from transpiration of the applied water by the crops. In addition to cropland, agricultural activities include numerous point sources such as animal feedlots, waste lagoons, and storage sheds for agricultural chemicals.
Although the area occupied by urban land is small compared to the total area of the Nation, the diverse activities in urban areas provide innumerable point sources of contamination that can affect the quality of shallow ground water. From a regional perspective, urban land can be considered as a nonpoint source that exhibits a wide range in water quality. These effects on ground-water quality are particularly important from a water-management viewpoint if the water-table aquifer beneath urban land is used or could be used as a source of water supply.
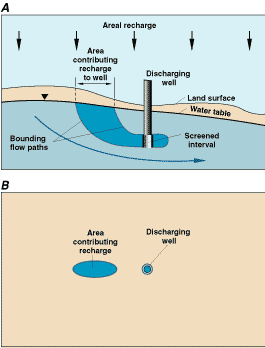
A noteworthy effort to protect ground-water quality and the sustainability of the local ground-water resource, specifically to protect the quality of ground water that is pumped from public-supply wells, is the wellhead protection programs undertaken by the U.S. Environmental Protection Agency and the States. The approach of these programs is to estimate areas at the water table that contribute recharge to public-supply wells (Figure 23) and then to implement ground-water protection practices on the overlying land surface. Because many uncertainties exist in estimating areas contributing recharge to pumping wells (particularly for well-screen placements at some distance below the water table), and because areas contributing recharge may be located a considerable distance from the pumped wells, implementing ground-water protection practices at the land surface often poses considerable challenges.

Figure 23. Area contributing recharge to a single discharging well in a simplified hypothetical ground-water system: (A) cross-sectional view, and (B) map view. (Modified from Reilly and Pollock, 1993.)
The area contributing recharge to a pumping well can be defined as the surface area at the water table where water entering the ground-water system eventually flows to the well. If the system is at equilibrium, this area must provide an amount of recharge that balances the amount of water being discharged from the well. Thus, lower areal recharge rates result in larger contributing areas of wells. If a nearby surface-water body also contributes water to the discharging well, the area contributing recharge is reduced and is a function not only of the areal recharge rate but also of the amount of water obtained from the surface-water body. Depending on factors that describe the three-dimensional flow system and the placement of the well, the area contributing recharge to a well does not necessarily have to include the location of the well itself.
The movement of water in both directions between ground-water systems and surface-water bodies has been discussed previously in this report. Chemical constituents are transported along with the moving water. Thus, contaminants in surface water can be transported into adjacent ground-water systems, and contaminants in ground water can be transported into adjacent surface-water bodies.
Because ground water commonly is a major component of streamflow, the quality of discharging ground water potentially can affect the quality of the receiving stream in many hydrologic settings (Figure 24). Because the proportion of streamflow contributed by ground water can vary greatly throughout the year, seasonal variations in the effects of ground-water quality on stream-water quality can occur.
Reductions in the quantity of ground water discharged to a stream as a result of pumping may have significant consequences where this discharge significantly dilutes the concentration of contaminants introduced to streams from point sources and surface runoff. In such situations, streamflow capture by pumping wells may reduce the contaminant-dilution capacity of the stream during periods of low flow below the dilution capacity assumed in setting discharge permits for the stream.
Contributing areas to wells often include surface-water bodies, and increasing attention is being placed on surface water as a potential source of contamination to wells. Possible contamination by induced infiltration of surface water adds several dimensions to the protection of ground water. These include consideration of the upstream drainage basin as part of the "contributing area" to the well and greater consideration of microbial contamination. Contaminated surface water may have a significant effect on the sustainable development of ground water near streams or on the need for treatment of ground water prior to use. Among the settings of greatest concern for contamination of ground water by streams are karst terrains where aquifers are hydraulically connected by sinkholes or other conduits that can channel river water directly into an aquifer with little or no filtration (see Box E).
In many aquifers, large changes in chemical oxidation conditions, organic-matter content, and microbial activity occur within a relatively thin (a few feet or even inches) zone or interface between ground water and surface water. Thus, conditions near the interface between ground water and surface water can significantly affect the transport and fate of nutrients, metals, organic compounds, and other contaminants between the two resources. Reactions at this interface commonly decrease the concentrations that might be transported between surface water and ground water (Winter and others, 1998).
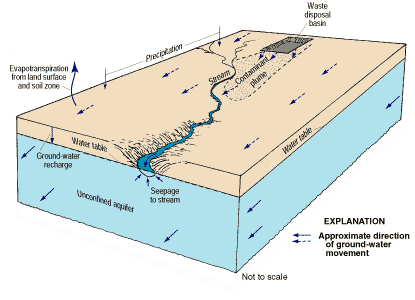
Figure 24. Simplified representation of a contaminant plume in ground water.
In this hypothetical example, sufficient time has elapsed for part of the plume of contaminated ground water to reach and discharge into a nearby stream. As shown, the stream intercepts the plume as it reaches the stream. In some situations, depending upon the geometry of the ground-water-flow system and the location of the plume in the flow system, part or all of the plume may flow under the stream and contaminate ground water on the other side of the stream.
The fresh ground-water resource of the United States is surrounded laterally and below by saline water. This is most evident along coastal areas where the fresh ground-water system comes into contact with the oceans, but it is also true in much of the interior of the country where deep saline water underlies the freshwater. The fresh ground-water resource being surrounded by saltwater is significant because, under some circumstances, the saltwater can move (or intrude) into the fresh ground-water system, making the water unpotable.
Freshwater is less dense than saline water and tends to flow on top of the surrounding or underlying saline ground water. Under natural conditions, the boundary between freshwater and saltwater maintains a stable equilibrium, as shown in Figure 25A. The boundary typically is not sharp and distinct as shown in Figure 25A, but rather is a gradation from fresh to saline water known as the zone of diffusion, zone of dispersion, or the transition zone. When water is pumped from an aquifer that contains or is near saline ground water, the saltwater/freshwater boundary will move in response to this pumping. That is, any pumpage will cause some movement in the boundary between the freshwater and the surrounding saltwater. If the boundary moves far enough, some wells become saline, thus contaminating the water supply. The location and magnitude of the ground-water withdrawals with respect to the location of the saltwater determines how quickly and by how much the saltwater moves. Even if the lateral regional movement of saltwater is negligible, individual wells located near the saltwater/freshwater boundary can become saline as a result of significant local drawdowns that cause underlying saltwater to "upcone" into the well (Figure 25B).
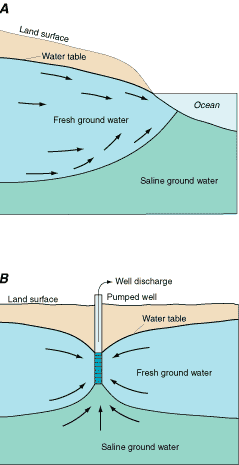
Figure 25. Relation of fresh and saline ground water. (Modified from U.S. Geological Survey, 1984.)
(A) In coastal areas, fresh ground water discharges to the surrounding saline surface-water bodies by flowing over the denser saline ground water. (B) In both coastal and inland areas, large drawdowns in an individual well can cause underlying saline water to migrate upward into the well and cause contamination of the water being discharged.
In 1969, the Task Committee on Saltwater Intrusion of the American Society of Civil Engineers (1969) indicated that saltwater intrusion of some type is an existing problem in nearly every State. Examples of saltwater intrusion are especially numerous along the coasts (U.S. Geological Survey, 1984). Some prominent examples follow.
Los Angeles and Orange Counties in California operate artificial-recharge programs to control saltwater intrusion caused by ground-water withdrawals. In Hawaii, several aquifers susceptible to saltwater intrusion underlie the island of Oahu. In Florida, saltwater intrusion occurs in the Jacksonville, Tampa, and Miami areas. Farther north on the Atlantic Coast, saltwater intrusion is occurring near Brunswick and Savannah, Georgia, and on Hilton Head Island, South Carolina. In New Jersey, aquifers underlying parts of Atlantic, Gloucester, Monmouth, Cape May, Ocean, and Salem Counties are being affected by saltwater intrusion. The threat of saltwater intrusion is always present on Long Island, New York, and Cape Cod, Massachusetts, because saltwater bodies surround both localities. A specific example of saltwater intrusion into the Old Bridge aquifer of New Jersey (Schaefer and Walker, 1981) is shown in Figure 26.
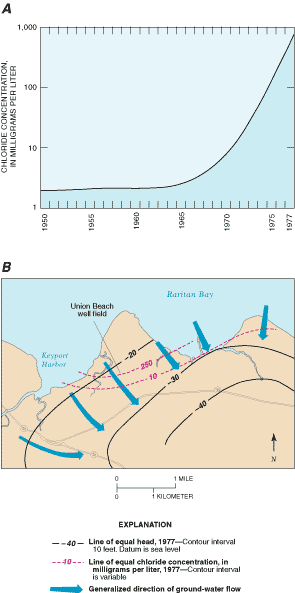
Figure 26. Saltwater intrusion into the Old Bridge aquifer, New Jersey. (Modified from Schaefer and Walker, 1981.)
(A) A composite graph of chloride concentration in water samples from wells screened at about the same depth in the Union Beach Borough well field. Chloride concentration in water samples from the Union Beach well field increased significantly above background levels beginning in about 1970 and increased steadily after that time. (B) As pumping in the area caused water levels to decline below sea level, saline ground water moved landward and caused the increase in chloride (and dissolved solids) in wells near the shore. Because of the increasing chloride and dissolved solids, pumpage was curtailed in the 1980's, and the well field was abandoned in the early 1990's and replaced by wells farther inland.
An inland area where saltwater intrusion is an important issue is the Mississippi River alluvial plain in Arkansas. For example, ground-water withdrawals from the alluvial aquifer for irrigation near Brinkley, Arkansas, have caused upward movement of saline water from the underlying Sparta aquifer into the alluvial aquifer (Morris and Bush, 1986). A confining unit separating the aquifers is discontinuous, and the intrusion appears to occur mainly where the confining unit is absent.
Many of the deeper aquifers in the central part of the United States contain saline water. Withdrawals from the overlying aquifers in these areas increase the potential for saltwater intrusion from below.
In summary, the intrusion of saltwater or mixing of fresh ground water with the surrounding saltwater, caused by withdrawals of freshwater from the ground-water system, can make the resource unsuitable for use. Thus, ground-water development plans should take into account potential changes in water quality that might occur because of saltwater intrusion.