
![]()
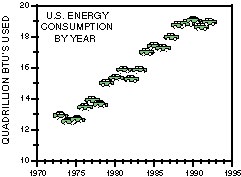 The United States energy industry faces the challenge of satisfying
our expanding thirst for energy while protecting the environment. This challenge
is magnified by the increasing volumes of saline water produced with oil
and gas in the Nation's aging petroleum fields. Ultimately, energy-producing
companies are responsible for disposing of these waters. USGS research provides
basic information, for use by regulators, industry, and the public, about
the chemistry of co-produced waters and environmentally acceptable ways
of handling them.
The United States energy industry faces the challenge of satisfying
our expanding thirst for energy while protecting the environment. This challenge
is magnified by the increasing volumes of saline water produced with oil
and gas in the Nation's aging petroleum fields. Ultimately, energy-producing
companies are responsible for disposing of these waters. USGS research provides
basic information, for use by regulators, industry, and the public, about
the chemistry of co-produced waters and environmentally acceptable ways
of handling them.
 Great volumes of saline water are produced as a byproduct of the
extraction of energy resources. Presently, for each barrel of crude oil
produced, approximately 10 barrels of water also are produced. The issue
of produced waters is becoming more important in the U.S. and abroad as
proportionally more water is produced as each field gets older (shown in
the figure at the right). Collection, handling, treatment, and disposal
of these waters in a producing field are major undertakings that, if done
poorly, can have a lasting negative environmental impact. However, when
handled and disposed of properly, these fluids should present no threat
to the environment. This Fact Sheet describes the quantity and quality of
the waters produced and highlights research being done by USGS scientists.
Great volumes of saline water are produced as a byproduct of the
extraction of energy resources. Presently, for each barrel of crude oil
produced, approximately 10 barrels of water also are produced. The issue
of produced waters is becoming more important in the U.S. and abroad as
proportionally more water is produced as each field gets older (shown in
the figure at the right). Collection, handling, treatment, and disposal
of these waters in a producing field are major undertakings that, if done
poorly, can have a lasting negative environmental impact. However, when
handled and disposed of properly, these fluids should present no threat
to the environment. This Fact Sheet describes the quantity and quality of
the waters produced and highlights research being done by USGS scientists.
HOW MUCH WATER IS PRODUCED AND WHAT IS DONE WITH IT?
In 1993, the U.S. produced about 2.5 billion bbl of crude oil and roughly 25 billion bbl of water. About 0.3 billion bbl of water were produced with natural gas; lesser amounts were produced with coal and coalbed methane and very small amounts were produced with geothermal resources. The total volume of water produced in 1993 from all sources, slightly less than 26 billion bbl or roughly a trillion gallons, would fill a tank the size of a football field and 530 miles (850 km) deep. This volume of water is equivalent to the average flow over Niagara Falls for 9 full days.
Prior to the institution of Federal regulations in the 1970's, large volumes of these waters were discharged into rivers, streams, and unlined evaporation ponds, contaminating surface and ground waters and soils in energy-producing States. Currently, about 65% of this water is beneficially reinjected into the same geologic formation from which it was originally produced to maintain production pressures. About 30% is pumped into deep injection wells with indigenous fluids which are already saline. The remaining 5% of the water (about 50 billion gallons per year) is discharged into surface waters such as rivers, natural and artificial lakes, or to the ocean.
WHAT IS THE QUALITY OF THIS WATER?
The water produced with hydrocarbon resources is generally unsuitable for most domestic or agricultural purposes because of its high salinity, or because of the presence of toxic or radioactive compounds. Total salinities range from about 1,000 milligrams per liter (mg/L; 1,000 milligrams equals 1 gram) to more than 400,000 mg/L. For comparison, the salinity of sea water is 35,000 mg/L and the U.S. EPA's recommended safe drinking-water limit is 500 mg/L.
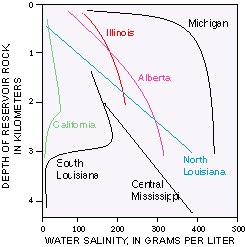 The salt content of these waters is inherited from original water
deposited with sediments in oceans and lakes. The original composition is
modified by mixing with waters of different origins; dissolution and precipitation
of minerals in the rocks at the high pressures and temperatures found in
oil and gas reservoirs; dissolution of salt minerals, such as halite (NaCl);
and other chemical reactions that occur between water and rocks in the subsurface.
The salt content of these waters is inherited from original water
deposited with sediments in oceans and lakes. The original composition is
modified by mixing with waters of different origins; dissolution and precipitation
of minerals in the rocks at the high pressures and temperatures found in
oil and gas reservoirs; dissolution of salt minerals, such as halite (NaCl);
and other chemical reactions that occur between water and rocks in the subsurface.
In most oil and gas fields, the salinity of formation water is greater when the hydrocarbons are produced from deeper in the ground. However, in some cases where the rocks in the basin consist mainly of shales or siltstones, fresher water is found at greater depths. The figure below shows the relationship between water salinity and depth for selected oil and gas basins in North America.
HOW DOES USGS STUDY THESE WATERS?
USGS research focuses on understanding the chemical reactions between the water and rocks that impact the quality of the water and also modify the mineral composition and other rock properties that influence oil and gas production. This work involves sampling the co-produced waters and characterizing their inorganic, organic, and isotopic compositions, their interactions with rocks as they migrate in the ground prior to production, and their behavior if released into the near-surface environment. For example, studies of petroleum- or gas-bearing rocks yield clues to the origin of chemical constituents found in the waters, and may provide geologic information necessary to anticipate the quality of waters produced with oil or gas in new production areas. Studies of the chemistry of some co-produced fluids have suggested environmentally safe handling methods such as evaporative concentration, which can yield commercially valuable solid byproducts. USGS also has conducted studies of the migration of brines in shallow aquifers and their potential impacts on water supplies. The recognition of naturally occurring radioactive materials in co-produced fluids studied by USGS has led to suggestions for safer ways to handle such fluids and other waste products. USGS research is continuing on this topic and results of specific projects will be forthcoming.
Selected References
USGS Home page on the Internet: http://www.usgs.gov
The Future of Energy Gases: U.S. Geological Survey Circular 1115, 1993.
Energy and the Environment- Application of Geosciences to Decision-Making:
U.S. Geological Survey Circular 1108, 1995.
1995 Assessment of Oil and Gas Resources: U.S. Geological Survey DDS-30,
Release 2.
Naftz, D.L., Spangler, L.E., and Peterman, Z.E., 1995, Using geochemical
data to identify sources of salinity to the freshwater Navajo Aquifer in
southeastern Utah: U.S. Geological Survey Fact Sheet 95-095.
For more information contact:
Richard B. Wanty
U.S. Geological Survey
Denver Federal Center
M.S. 973, P.O. Box 25046
Denver, Colorado 80225
(303) 236-1819
e-mail: rwanty@usgs.gov