
U.S. Geological Survey
USGS Fact Sheet FS-022-02
Online version 1.0
This document is also available in pdf format: fs-022-02.pdf (1.0 MB)
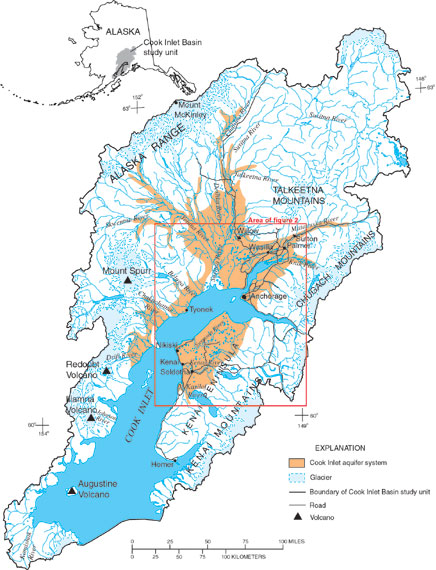
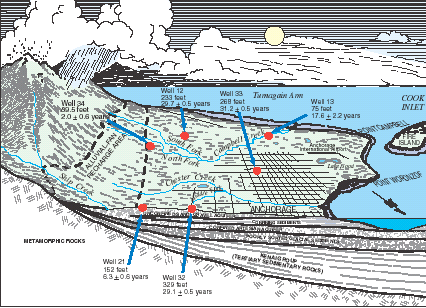
The Cook Inlet Basin (Fig. 1) encompasses 39,325 square miles in south-central Alaska. Approximately 350,000 people, more than half of Alaska’s population, reside in the basin, mostly in the Anchorage area. However, rapid growth is occurring in the Matanuska–Susitna and Kenai Peninsula Boroughs to the north and south of Anchorage. Ground-water resources provide about one-third of the water used for domestic, commercial and industrial purposes in the Anchorage metropolitan area and are the sole sources of water for industries and residents outside Anchorage. In 1997, a study of the Cook Inlet Basin was begun as part of the U.S. Geological Survey's National Water-Quality Assessment Program. Samples of ground water were collected from 35 existing wells (Fig. 2) in unconsolidated glacial and alluvial aquifers during 1999 to determine the regional quality of ground water beneath about 790 mi2 of developed land and to gain a better understanding of the natural and human factors that affect the water quality (Glass, 2001). Of the 35 wells sampled, 31 had water analyzed for atmospherically derived substances to determine the ground water’s travel time from its point of recharge to its point of use or discharge—also known as ground-water age.
|
Ground water is the source of almost all water used in homes and businesses outside the metropolitan Anchorage area and makes up about one-third of the Anchorage public-water supply. |
 |
| Figure 1. Location and major geographic features, Cook Inlet Basin, Alaska. |
Ground water moves slowly from its point of recharge to its point of use or discharge. This water starts as rain and melting snow that soak into the ground as recharge. In the Matanuska–Susitna, Anchorage, and Kenai Peninsula areas, ground water generally moves from near the mountain fronts toward Cook Inlet or the major rivers. Much of the water pumped by domestic and publicsupply wells may have traveled less than 10 miles, and the trip may have taken as short a time as a few days or as long as several decades. This ground water is vulnerable to contamination from the land surface, and many contaminants in the water would follow the same paths and have similar travel times from recharge areas to points of use as the chemical substances analyzed in this study. The effects of contamination may not be seen for several years after a contaminant is introduced into the groundwater system. Many contaminants could make the water unsuitable for drinking for many years, even in concentrations too low to detect without expensive chemical tests. The travel time of a chemically conservative substance depends primarily on the velocity of ground water through the aquifer, which in turn depends on the hydrologic characteristics of the aquifer system.
|
Ground-water age refers to the time elapsed since the water entered the saturated zone and was isolated (through additional recharge) from the atmosphere. The age actually applies to the date of introduction of the chemical substance and not the age of the water. |
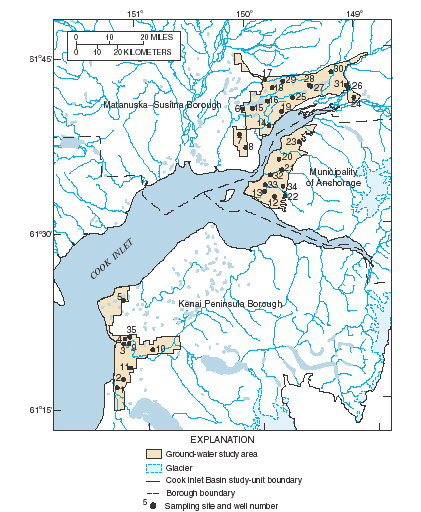 |
| Figure 2. Selected ground-water sampling sites, Cook Inlet Basin, Alaska, 1999. For characteristics of selected wells, see Table 1. |
Over the last 50 years, human activities have introduced a large number of substances into the air and water. Compounds that move readily through the environment relatively unchanged physically or chemically can help determine when the water was recharged, the areas of water recharge, the flow path, and the rate of water movement through an aquifer system. Tritium and chlorofluorocarbons (CFCs) are present in very small concentrations in the atmosphere and were used as tracer substances in this study. In the atmosphere, tritium, a radioactive isotope of hydrogen, becomes part of a water molecule in water vapor whereas CFCs dissolve in water vapor. Precipitation containing tritium and CFCs falls to the Earth and becomes incorporated into the hydrologic cycle. Water that has been exposed to the atmosphere within the past 50 years contains small amounts of these substances and is considered to be “young” or “modern.”
Dating a ground-water sample by using a radioactive isotope and its stable decay product is possible because the age of the sample is proportional to the amount of the product resulting from radioactive decay relative to the amount of the parent product. Dating ground water by using CFCs is possible because their amounts in the atmosphere over the past 50 years have been reconstructed. Age is determined by relating the measured concentrations of these substances in ground water back to known historical atmospheric concentrations and(or) to calculated concentrations expected in water in equilibrium with air.
For these or any other environmental tracer, age applies to the date of introduction of the chemical substance into the water and not to the water itself. The accuracy of a determined age depends in part on the estimate of the initial concentrations at the time of recharge and how perfectly the tracer substance moves with water. The ages or travel times calculated assume no dilution by mixing or delay by interaction with aquifer materials, which are common processes for contaminants moving with ground water. Wells that have long screened intervals may produce a mixture of water from several parts of an aquifer system that have different recharge areas and flow paths, which would yield different ground-water ages. The concentrations of all substances moving with water through the unsaturated zone and beneath the water table are affected, to some extent, by transport and chemical processes, including dispersion, mixing, degradation, and sorption. In Alaska, the presence of most manmade substances in a ground-water sample indicates some recent recharge (within about the last 50 years).
Tritium (3H or hydrogen-3), a radioactive isotope of hydrogen that has two neutrons and one proton in its nucleus, is produced naturally in the upper atmosphere from the bombardment of nitrogen by neutrons in cosmic radiation. It combines with oxygen and hydrogen and is incorporated directly into a water molecule, which moves with other water molecules through the hydrologic cycle. Tritium, which has a half-life of 12.4 years, decays to the stable isotope helium-3 (3He) by beta release. Prior to the initiation of atmospheric testing of nuclear weapons in 1953, the natural tritium content in precipitation was low, less than 20 tritium units (TU) and possibly as low as about 5 TU. (A tritium unit is equivalent to 1 tritium atom in 1018 atoms of hydrogen.) The explosions of nuclear devices resulted in peak tritium concentrations in precipitation during 1963–64 and a large input of tritium to the ground-water system. Concentrations in precipitation in Anchorage— measured in monthly composite samples and reported by the International Atomic Energy Agency/World Meteorological Organization 2001)—reached a peak of 6,100 TU in July 1963 declined to approximately the natural background range during the 1990s (Fig. 3). The decline of tritium in precipitation since 1963 is radioactive decay (about 5.5 percent per year) attenuation by the oceans and ground water. Much of the scatter in the tritium concentrations precipitation is due to circulation of upper-level masses and the movement of the jet stream.
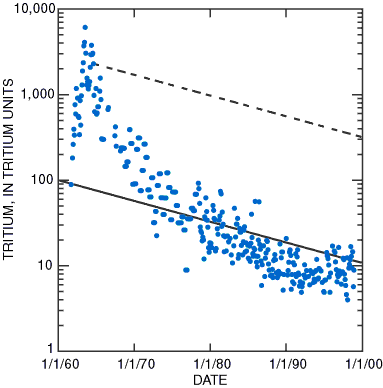 |
| Figure 3. Concentrations of tritium in composite monthly precipitation samples collected at Anchorage, Alaska, reached a peak of 6,100 tritium units (TU) in 1963 and declined to a range of 4–17 TU during 1998 (data from International Atomic Energy Agency/World Meteorological Organization, 2001). Diagonal lines represent the tritium concentration due to radioactive decay after the precipitation has infiltrated the ground. A sample collected in 1999 that contained about 12 TU could have been recharged in about 1961 or any time during 1972–99 (solid line). |
Tritium’s concentration decreases 50 percent every 12.4 years. This relatively rapid natural decay makes tritium an excellent indicator of recent ground-water recharge because water samples containing no detectable concentrations of tritium indicate that post-1953 water is not present in significant amounts. Water samples containing tritium at concentrations greater than 1 TU indicate some active ground-water recharge since 1953. Concentrations greater than 30 TU indicate that a considerable amount of the water was recharged during the 1960s or 1970s. A concentration greater than 300 TU would indicate water recharged in about 1963–64.
Ground-water age estimates can be improved by measuring the concentrations of the radioactive parent (tritium) and its stable daughter product (helium-3) in a ground-water sample. If all the helium-3 was derived from tritium decay and from air, a sample’s age can be calculated from the following formula:
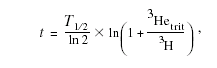
T1/2=12.4, half life of tritium, in years;
3Hetrit is amount of tritiogenic helium-3 (3He
derived from radioactive decay of tritium in
water), in tritium units; and
3H is sample’s tritium concentration, in
tritium units.
However, helium-3 in ground water also can be derived from the Earth’s crust and mantle and from nuclear fission of lithium. Helium-3 also can be lost from ground water through diffusion of gases across the water table to the unsaturated zone. Measurements of the concentrations of helium-4 and neon and the relative abundances of helium isotopes (3He/4He) and noble gases (He/Ne) in the sample can be used to estimate these amounts and to adjust the apparent ages, if needed.
Tritium/helium-3 analyses of ground water collected from 30 wells were performed at the Noble Gas Laboratory of the Lamont–Doherty Earth Observatory of Columbia University in Palisades, N.Y. In the laboratory, each water sample was degassed by using a vacuum-extraction system. After purification and separation of the helium and neon gases by means of a cryogenic cold trap, the 4He concentration was measured and the 3He/4He and He/Ne ratios determined. Tritium concentrations ranged from less than 1 to 34.6 TU (Table 1). The median value was 12.9 TU, and the median analytical precision was +0.3 TU. Of the 30 samples, 28 had tritium concentrations greater than 1 TU, indicating that these samples contained some modern ground water. Apparent ground-water ages, as estimated by Peter Schlosser and others at the Lamont–Doherty Earth Observatory, ranged from less than 1 to greater than 50 years, with a median age of about 18 years. About 30 percent of the wells (9 out of 30) yielded water that may have been recharged within the last 10 years.
Chlorofluorocarbons are a group of stable manmade compounds containing chlorine and fluorine that have been used in numerous domestic and industrial applications since the 1930s. CFCs were used as coolants in air conditioning; propellants in aerosol cans; blowing agents in open- and closedcell foams, insulation, and packing materials; and as solvents. However, the manufacturing of most CFC products ended in 1996 due to concerns about CFCs depleting ozone in the stratosphere. Because atmospheric CFCs dissolve in precipitation and enter the ground-water flow systems with recharge, they provide excellent tracers and are very useful for dating young water. Because CFCs do not occur naturally in the environment, ground water containing any amount of CFC must have a component of modern recharge water. Concentrations of three types of CFCs were determined: CFC-11 (CCl3F, trichlorofluoromethane or freon-11), CFC-12 (CCl2F2, dichlorodifluoromethane), and CFC-113 (C2Cl3F3; 1,1,2-trichlorotrifluoroethane).
Concentrations of CFCs in the air are measured the National Oceanic and Atmospheric Administration Climate Monitoring and Diagnostics Laboratory. CFC measurements recorded since 1976 at Niwot Ridge meteorological station about 20 miles west of Boulder, Colo., are shown in Figure 4. Concentrations prior to 1976 were reconstructed using chemical-production data. Atmospheric CFCs monitored at Barrow, Alaska, are nearly equal to those in Colorado, which shows that CFCs well mixed in the atmosphere at scales greater than a few hundred miles. Even though the concentration of CFC-12 in the air is nearly twice that of CFC-11, the solubilities in water are reversed. The solubility of CFC-11 in water in equilibrium with 1999 air is approximately 1,300 picograms per kilogram (pg/kg), compared to about 520 pg/kg for CFC-12 (assuming a recharge temperature of 2°C). (A picogram per kilogram is equivalent to one part per quadrillion.)
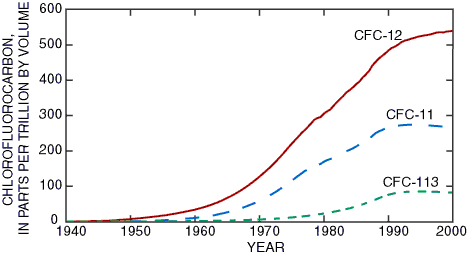 |
| Figure 4. Concentrations of chlorofluorocarbons CFC-11, CFC-12, and CFC-113 in North American air. Data from Plummer and Friedman (1999) and from the National Oceanic and Atmospheric Administration Climate Monitoring and Diagnostics Lab- oratory. |
Chlorofluorocarbon content was determined for ground water from 30 of the 35 wells sampled in 1999 that penetrate unconsolidated aquifers in the Cook Inlet Basin (Table 1). Because of the presence of atmospheric CFCs, a special water-sampling apparatus that prevents contact with air was used. Generally, five samples were collected at each site and were sent to the U.S. Geological Survey Chlorofluorocarbon Laboratory in Reston, Va., for analysis. Samples were analyzed by techniques that allow detection at extremely low levels—about 0.3 pg/kg. Almost all wells yielded water that contained detectable concentrations of CFCs, indicating some recent ground-water recharge. Some ground-water samples contained concentrations of CFC-11 and CFC-12 that were higher than expected for equilibrium with air. This finding suggests that CFC-enriched water, possibly from septic systems, may be reaching these wells. However, all CFC concentrations were less than 0.1 part per billion and present no risk to human health. Most measured concentrations of CFCs were less than those estimated for equilibrium with air for ages determined by the tritium/helium-3 techniques. This suggests that CFCs were not transported through the unsaturated or saturated zones at the same rate as tritium or that the CFCs were not as chemically conservative as tritium in the Cook Inlet aquifer system’s hydrogeologic environment. Some of the CFCs may have been absorbed by peat or other organic matter, degraded by microbes, or lost during sampling.
| Table 1. Comparison
of 1972 peak flows with other peak flows for selected current (2002) U.S.
Geological Survey streamflow-gaging stations [ft3/s; cubic feet per second; --, no record] |
|||||||||||||||
|
|
|||||||||||||||
|
Chemical
analyses and age computations1 |
|||||||||||||||
| Well number |
Sample date (mm/dd) |
Well characteristics |
Tritium (tritium units) |
Tritogenic helium-3 (tritium units) |
Computed apparent age (years) |
Chlorfluorocarbons
(picograms per kilogram) |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Depth (feet below land surface) |
Open interval (feet below land surface) |
CFC-11 |
CFC-12 |
CFC-113 |
|||||||||||
|
|
|||||||||||||||
|
1
|
08/27 | 95 |
90-95
|
14.7±0.3
|
103.0±0.2 | 20.2±0.3 | 5 |
100
|
5
|
||||||
|
2
|
08/10 | 177 |
172-177
|
26.9±0.5
|
84.1±0.3 | 25.6±0.3 | <1 |
5
|
<1
|
||||||
|
4
|
09/01 | 36 |
31-36
|
8.1±0.2
|
21.4±0.1 | 23.9±0.4 | 500 |
300
|
50
|
||||||
|
5
|
09/21 | 32 |
32
|
5.8±0.2
|
19.3±0.1 | 25.9±0.5 | 10 |
100
|
4
|
||||||
|
6
|
07/28 | 219 | 219 | 0.1±0.2 | 1.5±0.1 | (2) | 1 | 1 | <1 | ||||||
|
7
|
08/03 | 60 | 60 | 11.8±0.4 | 19.0±0.1 | 17.0±0.6 | 300 | 33,000 | 50 | ||||||
|
8
|
08/03 | 67.3 | 56.3-67.3 | 8.5±0.2 | 4.3±0.1 | 7.3±0.4 | 400 | 400 | 70 | ||||||
|
10
|
08/09 | 68 | 90 | 18.1±0.5 | 27.0±0.0 | 15.7±0.6 | 100 | 300 | 10 | ||||||
|
11
|
08/10 | 110 | 105-110 | 13.2±0.3 | 30.5±0.1 | 21.0±0.3 | 1 | 14 | <1 | ||||||
|
12
|
07/27 | 233 | 233 | 15.2±0.3 | 65.4±0.4 | 29.7±0.5 | 3 | 1 | <1 | ||||||
|
13
|
07/26 | 75 | 70-75 | 2.0±0.2 | 0.1±0.0 | 17.6±2.2 | 10 | 500 | 7 | ||||||
|
14
|
08/04 | 73.6 | 61-73.6 | 34.6±0.7 | 113.2±0.7 | 25.8±0.4 | 39,000 | 30 | 3 | ||||||
| 15 | 08/06 | 24 | 24 | 10.3±0.4 | 0.3±0.1 | 1.8±0.6 | 900 | 300 | 60 | ||||||
| 16 | 09/13 | 190 | 190 | 0.1±0.2 | 2.3±0.1 | (2) | 1 | 1 | 4 | ||||||
| 18 | 08/06 | 121 | 121 | 13.5±0.3 | 61.8±0.5 | 30.1±0.4 | 1 | 20 | 6 | ||||||
| 19 | 08/04 | 100 | 100 | 31.6±0.6 | 45.4±0.3 | 15.2±0.3 | 3 | 20 | <1 | ||||||
| 20 | 08/02 | 85 | 75–85 | 12.1±0.2 | 2.4±0.1 | 3.2±0.1 | 400 | 320,000 | 70 | ||||||
| 21 | 07/30 | 152 | 132–152 | 12.5±0.3 | 5.0±0.2 | 6.3±0.6 | 90 | 300 | 20 | ||||||
| 23 | 07/27 | 129 | 119–129 | 24.6±0.5 | 34.8±0.2 | 16.0±0.4 | 4 | 50 | <1 | ||||||
| 24 | 09/29 | 50 | 50 | 9.1±0.2 | 4.2±0.1 | 6.5±0.4 | 320,000 | 320,000 | 80 | ||||||
| 25 | 09/15 | 213 | 213 | 19.7±0.4 | 10.2±0.2 | 7.1±0.2 | 20 | 70 | 8 | ||||||
| 26 | 08/31 | 81 | 81 | 8.9±0.2 | 0.0±0.1 | 0.8±0.5 | 800 | 500 | 90 | ||||||
| 27 | 08/30 | 60 | 60 | 21.6±0.4 | 22.3±0.2 | 13.4±0.3 | 300 | 500 | 5 | ||||||
| 28 | 08/16 | 141 | 141 | 21.7±0.4 | 73.7±0.3 | 26.9±0.4 | <1 | 6 | 1 | ||||||
| 29 | 08/23 | 80 | 80 | 9.1±0.2 | 15.2±0.1 | 19.1±0.4 | 80 | 330,000 | 4 | ||||||
| 30 | 10/20 | 61 | 50–55 | 13.8±0.3 | 1.9±0.2 | 23.4±0.4 | — | — | — | ||||||
| 31 | 08/24 | 186 | 151–171 | 12.0±0.4 | 0.1±0.1 | 0.3±0.5 | 32,000 | 500 | 100 | ||||||
| 32 | 07/26 | 329 | 270–322 | 20.6±0.4 | 76.3±0.5 | 29.1±0.5 | 9 | 60 | 3 | ||||||
| 33 | 07/21 | 268 | 232–268 | 12.5±0.3 | 49.5±0.3 | 31.2±0.5 | 20 | 5 | <1 | ||||||
| 34 | 07/23 | 89.5 | 70–89 | 14.0±0.5 | 1.1±0.1 | 2.0±0.6 | 300 | 300 | 60 | ||||||
| 35 | 08/11 | 420 | 348–420 | — | — | — | <1 | <1 | <1 | ||||||
| 1Tritium
and helium-3 analyses and apparent-age computations were performed by Peter
Schlosser and others at Lamont–Doherty Earth Observatory, Columbia University.
Chlorofluorocarbons analyses were performed by U.S. Geological Survey. 2Unknown but probably old (likely beyond dating range of method). 3Concentration greater than possible for equilbrium with air. |
|||||||||||||||
|
Chlorofluorocarbons were not as reliable for estimating ground-water age as tritium because their concentrations in ground water appear to have been reduced by sorption and degradation or increased by septic-tank effluent. |
The presence of tritium and CFCs in most ground-water samples in the unconsolidated-aquifer system in Cook Inlet Basin supports the conceptual model that ground water is primarily from the infiltration of local precipitation and water from streams, rivers, lakes, and wetlands rather than older water from deeper aquifers (Fig. 5). Most wells used to supply water to individual homes in the study area are completed near the top of saturated glacial till, outwash, or alluvial deposits and are expected to yield water that was recharged within the last 25 years. Public-supply wells generally yield water from the middle or deeper zones in the unconsolidated aquifer. However, the finding that water from several of these wells also contained measurable quantities of tritium and CFCs suggests that their high yields may capture water from above or that the horizontal rate of ground-water movement from areas of recharge is fairly fast, in the several-feet-per-day range rather than several-feet-per-year range.
The wells yielding the oldest water were in the western part of the study area in the Matanuska–Susitna Borough. Well 6 (depth, 219 feet), and well 16 (depth, 190 feet) yielded water containing very low concentrations of tritium (<1 TU) and CFCs (<5 pg/kg), indicating water more than 50 years old. Three wells (numbers 24, 26, and 31) in Palmer near the Matanuska River yielded very young water and had chemical contents similar to the river; this indicates that the aquifer system that these wells tap is in hydraulic connection with the river. In Anchorage, the ages of water from sampled wells generally increased from the mountain front to the lowlands. Water samples from wells 34 and 21 near the mountain front were estimated to have been recharged within the last 10 years, whereas water from well 32 (depth, 329 feet) in the Mountain View area and well 33 (depth, 268 feet) in the midtown area yielded water recharged about 30 years ago.
 |
| Figure 5. Concentrations of tritium and CFCs were measured in eight wells in Anchorage, Alaska, during July 1999. (Six wells are represented in this figure.) Ground-water dating shows that water samples from wells near the mountain front were the most recently recharged. This suggests a general movement of water from the mountains toward Cook Inlet. However, wells in all parts of the city may receive some water from the infiltration of precipitation or from local streams, lakes and wetlands. Depth of well, in feet, and computed ground-water age or travel time, in years, are shown for each well. (Well locations are also plotted in Fig. 2. Sampling date and well characteristics are given in Table 1.) |
|
The presence of any tritium or CFCs in ground water from sampled wells indicates that some of the well water was recharged within the last 50 years and that the aquifers tapped by those wells are susceptible to contamination. Common types of contaminants include bacteria, viruses, and nitrates from septic systems; organic compounds from gasolines and fuel oils; cleaning solvents; and pesticides. Removing contaminants from soils, aquifer materials, and ground water is difficult and much more expensive than preventing contamination in the first place. Septic tanks need to be completed in acceptable soils and an adequate distance above the water table. Fuels need to be stored in secure, corrosion-free tanks and their distribution pipes kept from leaking. Chemicals should be used and disposed of properly when they are no longer needed. Pesticides used on gardens, lawns, and crops and herbicides used on right-of-ways should be applied properly to avoid contaminating the aquifer.
—Roy L. Glass
Glass, R.L., 2001, Ground-water quality, Cook Inlet Basin, Alaska, 1999: U.S. Geological Survey Water-Resources Investigations Report 01–4208, 58 p.d
International Atomic Energy Agency/World Meteorological Organization, 2001, Global network for isotopes in precipitation—The GNIP database: URL http://isohis.iaea.org (accessed Aug. 9, 2001).
Lamont–Doherty Earth Observatory, 2001, Environmental Tracer Group, Lamont–Doherty Earth Observatory of Columbia University: URL http://www .ldeo.columbia.edu/~etg/ (accessed Dec. 4, 2001).
Plummer, L.N., and Friedman, L.C., 1999, Tracing and dating young ground water: U.S. Geological Survey Fact Sheet 134–99, 4 p. [URL: http://water .usgs.gov/pubs/FS/FS-134-99/ (accessed Sept. 19, 2001)].
U.S. Geological Survey, 2001, The Reston Chloro- fluorocarbon Laboratory: URL http://water.usgs .gov/lab/cfc/ (accessed Dec. 4, 2001)]
|
For more information, please contact:U.S. Geological Survey 4230 University Drive, Suite 201 Anchorage, AK 99508-4664
Telephone 907-786-7100 or vist website URL: http://ak.water.usgs.gov
|
U.S. Department of the Interior
U.S. Geological Survey Fact Sheet FS–022–02
April 2002
| AccessibilityFOIAPrivacyPolicies and Notices | |
 |
|