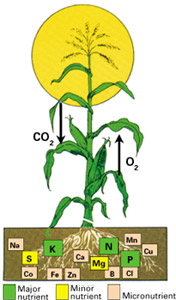
Figure 1. Sketch showing how plants require many different elements for optimum growth. Another essential requirement, which is not shown here, is water. (Graphic courtesy of the International Fertilizer Industry Association.)


Figure 1. Sketch showing how plants require many different elements for optimum growth. Another essential requirement, which is not shown here, is water. (Graphic courtesy of the International Fertilizer Industry Association.) |
A fertilizer is a substance applied to soil to enhance its ability to produce plentiful, healthy plants. Fertilizers are natural and manufactured chemicals containing nutrients known to improve the fertility of soils. Nitrogen, phosphorus, and potassium are the three most important nutrients for crop growth; some plant scientists think sulfur is also a major nutrient because of its benefit to plant health and growth. These and other nutrients (fig. 1) are found naturally in soils. Soils used for agriculture, however, become depleted in these nutrients and frequently require fertilizing before the soils can be used successfully again. The most efficient way to produce fertilizer is through mining or industrial processes.
Fertilizers are increasingly important to improve crop yields needed to feed a growing world population. The United Nations estimates that the world population will reach 7.7 billion by 2020, an increase of 35 percent from 5.7 billion in 1995. Much of the population increase will be in developing countries, where food supply and malnutrition are already serious problems (Pinstrup-Andersen and Cohen, 1998).
Although demand for food will increase as population increases, the area of cultivated land will not increase significantly. For this reason, methods for improving crop production must be found to satisfy the nutritional requirements of the expanding population. The use of fertilizers is one way to increase food supplies.
Planted acreage varies little from year to year in the United States. Agricultural production is found in every State but is concentrated in the Midwest (fig. 2). Many different crops are grown in the United States, but more than 80 percent of crop land is planted in corn, soybeans, and wheat (fig. 3). Because efforts to improve crop yields have intensified, increased quantities of mineral fertilizers are applied to replace nutrients depleted from the soil. This is one of the easiest and quickest ways to improve soil fertility. Research has helped determine nutrient requirements for specific crops (fig. 4).
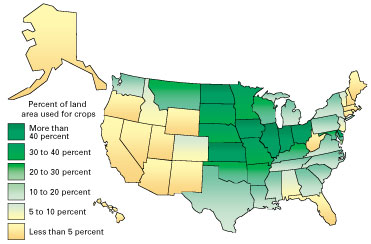
Figure 2. Map showing percent of land area used for crops. Major domestic growing areas are concentrated in the Midwest. The darker green shades depict the States where the largest percentages of their land area are used for crop land. Acreage data published by the U.S. Department of Agriculture. |
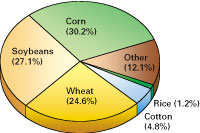
Figure 3. Diagram showing major crops in the United States. A tremendous variety of crops are grown in the United States. A few types, however, dominate the total area planted -- corn, soybeans, and wheat occupy more than 80 percent of planted crop land. Acreage data published by the U.S. Department of Agriculture. |
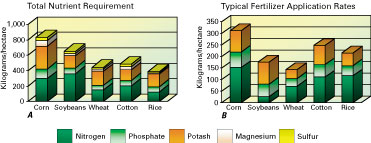
|
Figure 4. (Left) A, Chart showing total nutrient requirements for major crops. B, Chart showing typical fertilizer application rates for major crops. As seen in A, different crops require different nutrients. Chart B shows the average application rates for typical good crop yields. Note the small nitrogen application rate for soybeans relative to the other crops. Soybeans and other legumes are able to obtain their nitrogen requirements from air. Nutrient requirement data published by the Potash and Phosphate Institute and application rate data from Mississippi Chemical Corp. |
An average of 85 percent of the ammonia produced in the United States is used in fertilizers. About 11.5 million metric tons per year (Mt/yr) of nitrogen in all forms is used in fertilizers in the United States. Ammonia represents about 32 percent of the total fertilizer nitrogen used; urea and urea-ammonium nitrate solutions together represent 37 percent; ammonium nitrate, 5 percent; and ammonium sulfate, 2 percent. The remainder is supplied by multiple-nutrient fertilizers that contain varying quantities of nitrogen, phosphorus, and potassium.
Ammonia is produced in 21 States, but more than one-half of total U.S. ammonia production capacity is in Louisiana, Oklahoma, and Texas. The region has large reserves of natural gas used in ammonia preparation. The United States is the world's second largest ammonia producer and consumer following China. An average of 13 Mt/yr of nitrogen as ammonia is produced in the United States. Because this does not supply all its domestic demand, the United States imports significant quantities of ammonia -- between 3 and 4 Mt/yr -- primarily from Canada, Trinidad and Tobago, and Russia. The United States exports less than 1 Mt/yr of ammonia.
Phosphate rock, when used in an untreated form, is not very soluble and provides little available phosphorus to plants, except in some moist acidic soils. Treating phosphate rock with sulfuric acid makes phosphoric acid, the basic material for producing most phosphatic fertilizers. Phosphatic fertilizers include diammonium phosphate (DAP) and monoammonium phosphate (MAP), which are produced by reacting phosphoric acid with ammonia, and triple superphosphate, produced by treating phosphate rock with phosphoric acid. More than 90 percent of the phosphate rock mined in the United States is used to produce about 12 Mt/yr of phosphoric acid. Domestic consumption of phosphate in fertilizers has averaged 4.5 Mt/yr since 1994.
The United States supplies most of the phosphate fertilizers in the world. Overall, more than 50 percent of the phosphoric acid produced in the United States is exported as finished fertilizers or commercial acid. The United States accounts for more than 50 percent of global interregional trade in phosphates; 90 percent in MAP; and 75 percent in DAP. The United States also imports some phosphate rock for processing -- about 1.8 Mt/yr.

Figure 5. Photograph showing a soybean test plot demonstrating the improved growth obtained with the addition of potash. Photograph courtesy of the Potash and Phosphate Institute. |
Potassium is found in potash, a term that includes various mined and manufactured salts; all contain potassium in a water-soluble form. Potash is produced at underground mines, from solution-mining operations, and through the evaporation of lake and subsurface brines. Minerals mined for potash include potassium chloride [KCl or muriate of potash (MOP)], potassium-magnesium sulfate [K2SO4·MgSO4 or sulfate of potash magnesia (SOPM)], or mixed sodium-potassium nitrate (NaNO3+KNO3 or Chilean saltpeter). Manufactured compounds are potassium sulfate [K2SO4 or sulfate of potash (SOP)] and potassium nitrate (KNO3 or saltpeter).
The United States produces about 3 Mt/yr of potash, mostly in New Mexico. About 1 Mt of that production is exported. About 8 Mt is imported by the United States every year, primarily from Canada, the largest potash producer in the world. The United States consumes about 11 Mt/yr tons of potash of all types and grades. About 95 percent of this is used for agricultural purposes.
Nearly 60 percent of all sulfur consumption is in the production of phosphate fertilizers. Nearly 10 percent of additional consumption is used in other agricultural applications, including the production of nitrogenous fertilizers and plant nutrient sulfur.
The largest sources of elemental sulfur are petroleum refining and natural gas processing at numerous facilities throughout the United States. Elemental sulfur is mined at a few locations worldwide. Smaller quantities of sulfur are recovered as sulfuric acid at nonferrous metal smelters, and minor amounts are recovered at coking operations. Between 11 and 12 Mt/yr of domestic sulfur in all forms are produced. The United States imports about 3 Mt/yr of sulfur as elemental sulfur and sulfuric acid. Exports total less than 1 Mt/yr. The majority of U.S. imports come from Canada, the largest sulfur exporter in the world. Annual apparent consumption is almost 14 Mt.
The CRP, the Federal Government's largest environmental improvement program, is a voluntary program designed to decrease topsoil erosion, increase wildlife habitat, and protect ground and surface waters by reducing runoff and sedimentation. The program provides incentives to encourage farmers to plant permanent covers of grass and trees where vegetation can prevent erosion, improve water quality, and provide food and habitat for wildlife. The goal is to remove the most vulnerable acreage from agricultural production, limiting planted acres and thus fertilizer consumption in these areas (Farm Service Agency Online, April 26, 1999, Conservation Reserve Plan, accessed April 27, 1999, at URL http://www.fsa.usda.gov/dafp/cepd/crp/ pubs.htm).
Genetic research has developed crop varieties that increase yields without requiring comparable increases in fertilizer requirements. New strains of crops also are being developed that are resistant to insects and specific herbicides and have increased nutritional value for people or animals.
One possible effect of too much fertilizer entering bodies of water is hypoxia. Hypoxia affects water near the bottom of the Gulf of Mexico along the Louisiana-Texas coast where dissolved oxygen can be less than 2 parts per million. Hypoxia can cause stress or death in bottom-dwelling organisms that cannot move out of the hypoxic zone. The amount of fertilizer entering the Gulf can be determined by examining data from the USGS streamflow and water-quality monitoring stations throughout the Mississippi River basin. USGS scientists, as members of the Committee on Environment and Natural Resources of the National Science and Technology Council, are analyzing current and historic data from these stations to understand better the causes and consequences of hypoxia in the Gulf. Specifically, the USGS, through its Toxics and National Stream Quality Accounting Network programs, is addressing two issues: (1) the loads and sources of nutrients delivered to the Gulf of Mexico, and (2) the relative importance of specific human activities such as agriculture, atmospheric deposition, and point-source discharges in contributing these nutrients (U.S. Geological Survey, December 18, 1998, Hypoxia in the Gulf of Mexico, accessed May 4, 1999 at URL http://www.rcolka.cr.usgs.gov/ midconherb/hypoxia.html).
The USGS, in cooperation with the Maryland Department of the Environment, the Metropolitan Washington Council of Governments, and the Virginia Department of Environmental Quality, is also studying the amount of nutrient pollution that enters the Chesapeake Bay each year from its major tributaries. Results of the study are used to determine whether steps taken to reduce the amount of pollution entering the bay are working (Zynjuk, 1995).
Excessive phosphorus runoff has long been recognized as associated with eutrophication of lakes and other nonflowing bodies of water. Eutrophication is the response of a body of water to enrichment by nutrients. The results include heavy growth of aquatic plants and algal mats and deoxygenation. Although industrial and sewage discharges of phosphate have been reduced greatly during the past 25 years, eutrophication remains a major environmental issue. Most phosphorus enters bodies of water through soil erosion from agricultural lands. Improved planting methods, fertilizer management, and soil conservation techniques are being used to reduce phosphorus runoff (Potash and Phosphate Institute, 1999).
Fertilizer production also is an environmental concern. For every ton of phosphoric acid produced, five tons of phosphogypsum are generated. Phosphogypsum is a solid material that results from the reaction of phosphate rock with sulfuric acid. Although it is nearly identical to natural gypsum, it may contain small amounts of sand, phosphate, fluorine, radium, and other elements present in phosphate ore. Federal regulations restrict both use and research involving phosphogypsum because of its radium content and require phosphogypsum to be stacked on the ground. A limited amount of phosphogypsum, with a minimal radium content, is used as an agricultural soil amendment. During the past 50 years, more than 700 Mt have accumulated in Florida alone. These enormous stacks, some covering an area of more than 300 hectares and up to 60 meters high, have settling ponds on top that contain highly acidic water that can overflow into waterways. New regulations have been enacted to guard against potential spills (Johnson and Traub, 1996).
Nelson, F.J., and Schertz, L.P., eds., 1996, Provisions of the Federal Agriculture Improvement Act of 1996: U.S. Department of Agriculture Bulletin 729, 147 p.
Pinstrup-Andersen, Per, and Cohen, M.J., 1998, The role of fertilizer in future world security, in Fertilizer Industry Round Table, 48th, Annapolis, Md., October 26-28, 1998, Proceedings: Glen Arm, Md., The Fertilizer Industry Round Table, p. 1-19.
Potash and Phosphate Institute, 1999, Phosphorus and the environment: Better Crops with Plant Food, v. 83, no. 1, p. 37-39.
Zynjuk, L.D., 1995, Chesapeake Bay -- Measuring nutrient pollution: U.S. Geological Survey Fact Sheet FS-055-95, 2 p.
-- Stephen M. Jasinski, Deborah A. Kramer, Joyce A. Ober, James P. Searls
|
For more information, please contact:
Joyce A. Ober |
|||
|
More information on fertilizer materials can be found on the Internet at:
http://minerals.usgs.gov/minerals/ or by fax from MINES FAXBACK at (703) 648-4999 |

|
||
|
U.S. Department of the Interior U.S. Geological Survey |
USGS Fact Sheet 155-99 September 1999 |
This page is https://pubs.usgs.gov/factsheet/fs155-99/fs155-99.html
Maintained by Eastern Publications Group Web Team
Last revised 11-30-99 (krw)