USGS Fact Sheet FS218–96
October 1996
To answer these questions, the NAWQA Program combines the best historical information with the new data to arrive at the most comprehensive assessment of nutrients available.
Nutrient pollution is not a new problem, but it is among the most persistent. Consequently, the status of nutrients is one of the first water– quality issues evaluated by the U.S. Geological Survey (USGS) National Water–Quality Assessment (NAWQA) program.
Public concern about water pollution from nutrients prompted passage of the Clean Water Act in 1972. Yet, more than 20 years later, the U.S. Environmental Protection Agency (USEPA) reports that nutrient are still a water–quality problem—the leading cause of degradation in lakes and estuaries, the third leading cause of impairments in rivers, and the main reason why public supply wells fil to met USEPA drinking–water standards.
All living things need nitrogen and phosphorus, two of eh most important nutrients. However, an access of these nutrients can cause tow main te s of water–quality problems: degraded aquatic habitat and polluted drinking water. In addition, economic and recreational problems, such as clogged intake pies and water that are unfit for swimming, can results.
Scientific guidelines rather than regulatory standards are used to determine when too much nitrate, ammonia or phosphate exists in aquatic habitats. These guidelines reflect the fact that "too much" of any nutrient depends on more than just the amount; water temperature and acidity (pH) are also factors. For example, " maximum exposure criteria" for ammonia, which can be very toxic to fish, range from 0.1 milligram per litter (mg/L) under some conditions to 2 mg/L under others.
The USEPA enforces a drinking–water standard or Maximum Contaminated Level (MCL), for nitrate under the Safe Water Drinking Act. If drinking water contains nitre concretions in excess of 10mg?L, it is considered unsafe for human consumption. Indeed, too much nitrate can cuse "blue baby syndrome" or methemoglobienmia. This condition can cause death, particularly in infants.

Too much nitrate in ground water can render it undrinkable. Near Greenly, Colorado, residents purchase bottled water.

The effects of eutrophication on rivers, streams and lakes may be quite visible. Here alage form a green scum on the Snake River near Buhl, Idaho.
Ammonia, a form of nitrogen, promotes eutrophication. It dissolves easily in water and can be toxic to fish. It can change into nitrate ane ammonia gas.
Nitrate, a form of nitrogen, can harm human health and promote eutrophication. Because it dissolves easily but does not degrade quickly, nitrate can be flushed down to ground water or carried long distances in streams and rivers.
Phosphates, a form of phosphorus, in moderately water–solutable and not very mobile because they adhere to soil particles. Eroded soil carries phosphates into streams and lakes where they can cause eutrophication.
Natural and anthropogenic (human) source of nutrients are ubiquitous. In fact, human sources increase as population increase.
Natural source of nutrient includes soils and decaying plant materials such as fallen leaves and grass. Most nutrients introduced by humans are associated with agriculture and urban settings, and originate in:
Agriculture covers nearly one–half of the continental United States. This creates wide–spread opportunities for nutrients that originate on farms to migrate into streams and ground water. Indeed, more nitrogen found in water nationwide comes from agricultural activities than from any other source (fg. 1).
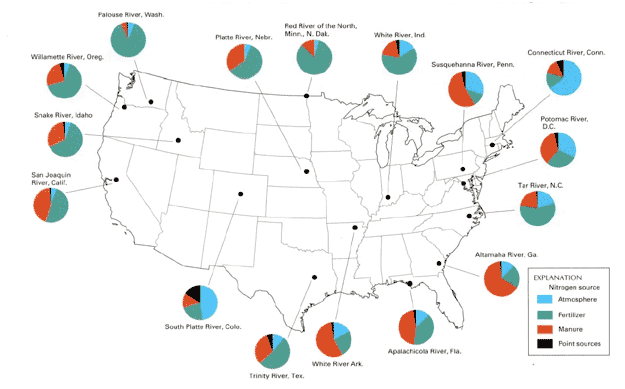
Figure 1. Most nutrients loads are found in streams — 92 percent of the nitrogen and 76 percent of the phoshours — come from nonpoint sources. By contrast 8 percent of nitrogen and 24 percent phosphorus stream load are generated from point sources (d).
"Load" refers to the amount of nutrient carried by a stream over a period of time (for example, tons of nitrogen per year).
"Point–source" pollution can be traced to specific point of discharge like pipes from waste–treatment plants and factories, and confined animal feedlots or combined sewers.
In urban areas, which occupy less than 5 percent of the land base, human sewage, emissions from power generators and automobiles, lawn fertilizers, and pet waste contain nutrient that can get into water.
The atmosphere is often overlooked as a source of nutrient pollution in water. However, 54 percent of the nitrogen emitted by the burning of fossil fuels (about 3.2 million tons of nitrogen) falls into U.S. watersheds every year.
From watershed to watershed, the source of nutrients differ, depending ok how much land is dedicated to urban, agriculture, forest, or other uses. Because some nutrients can move fairly easily through the rivers, on air currents, and in rain, they affect the quality of water not only where they originate, but where they end up in the water system. For example, evidence suggests that nutrients in fertilizers use in Minnesota travel through the Mississippi River as far south as the Gulf of Mexico.
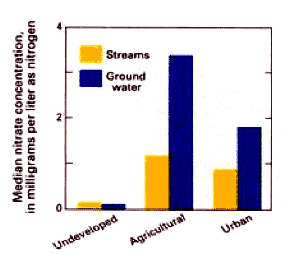
Nitrate concentrations are highest in shallow ground water in agricultural areas (d).
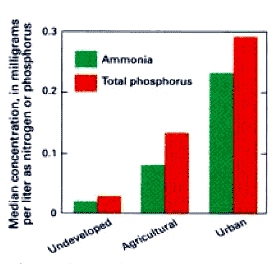
Ammonia and phosphorus concentrations are highest down stream from urban areas (d).
. . . in domestic wells that are located in agriculture areas. Fully 12 percent of such sample wells exceeded the MCL for nitrate. This exceedance rate is twice that of domestic wells in other land use settings, and more than sis time higher than the rate of exceedance in public supply wells..
. . . not in public supply wells. Only one percent of public wells exceed the nitrate MCL. Why? Because public supply wells tend to be deep an are less likely to be placed close to cropland and septic systems that increase the rick of nitrate commatimation. Also under the Safe Drinking Water Act, regular monitoring and prompt corrective action is public supplies exceeds an MCL..
Risk factors for contamination can be identified regionally, and, in some cases, locally. Targeted monitoring in watersheds is underway to measure actual concentrations and trends. These methods can also be used for delineation and assessments by States.
. . . in rivers and streams downstream from agricultural and urban areas. Ammonia and phosphates concentrations are higher downstream from urban areas due by accumulated nutrients due to sewage affluent and second highest downstream from agricultural areas, owing to nonpoint runoff. Aquatic ecosystems are agricultural area appears to offset the very sensitive to nutrients, eve at concentrations well below human drinking water standard. About 10 percent of samples takes from urban rivers and streams have ammonia levels greater than "chronic exposure criteria" for aquatic life. Phosphorus concentrations exceed in–stream limits recommenced bu USEPA in75 percent of urban streams sampled and25 percent of agricultural streams sampled..
"Concentration " refers to the amount of a nutrient contained in a tertian volume of water (for example, milligrams of nitrate per litter).
. . . in the first few weeks of every growing season. Seasonal highs and lows exceed in nitrate concentrations reflect the timing of fertilizer use and the amount of rainfall. For example, nitrate levels in Midwestern streams peak after spring fertilizer applications and heavy rains.
. . . in watersheds in bottom of regional drainage basins. Nitrogen and phosphorus move across State boundaries downstream in the currents of large rivers. Coastal estuaries and other terminal points in drainage basins may be especially affected from urban areas due by accumulated nutrients.
. . . not in the Southeast, where the large amount of woodland within the agricultural area appears to be offset the presence of risk factors for nitrate commination (fig 2).
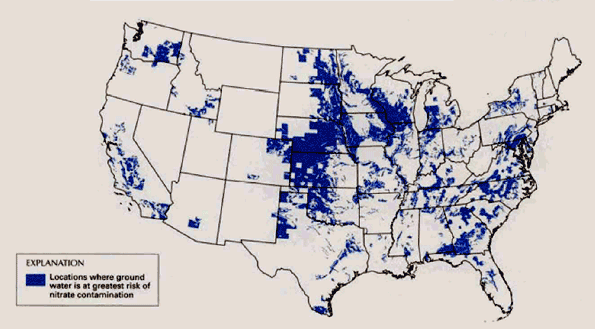
Figure 2. Ground water nitrate levels are likely to be highest where (1) the water table is less tan 100 feet (2) soils are well–drained (3) nitrogen inputs (like fertilizer or manure) are high, (4) the population density is high, and (5) the ratio of woodland to cropland is low (d).
Yes. A pattern of declining phosphorus loads in rivers downstream from several urban areas can be explained by restricted use of phosphate in detergents and more effective treatment of waste water. In one case, the amount of phosphorus contained in treated sewage effluent coming from Atlanta, Georgia, dropped by 83 percent in 4 years (1998—93). USGS determined that the amount of phorphours in the Chattanagoooa River fell 53 percent as a results (fig 3).

Figure 3. Phosphorus loads in the Chatthagoona Rive downstream form Atlanta, Georgia, dropped bu more than 50 percent afer water quality improvement efforts began (d).
Yes. In Dallas, Texas, and nine other urban areas where enhancements in sewage treatment were begun in the 1970’s and 1980’s downstream ammonia concentrations have decreased. These enhancements, which involve transforming ammonia into nitrate, make effluent less toxic to fish, but may not resole problems of eutrophication. Why? Because the total amount of nitrogen in the water remains the same, despite of the change in form.
Nationwide, nutrient levels in streams, rivers and ground water changed little between 1980 and 1989—phosphorus levels declined slightly and nitrate levels remained steady. Local trends, which may be more variable, are difficult to access because historical data are often missing.
Maybe. In several agriculture area (see inset, right), nitrate levels, in streams and rivers generally increase following increased in fertilizer use and decrease following deceases in fertilizer use. Erosion control, reduced tillage pratices, nutrient mangement, and other best management practices (BMPs) can potentially effect water quality. However, it is not cleat whether BMPs have overall lead to improved water quality.
Can’t tell yet. Information collected by the USEPA indicates that nitrate commination causes many "exceedances" (failures to meet a drinking wat MCL) in public supply wells. Does this mean ground–water management efforts are not working? Not necessarily. These deep wells may be tapping into "old" water—as much as 50 years old. The benefits of reducing nitrogen use today may not become evident for many years.
Nitrate concentrations in the lower San Joaquin River have increased since the 1950’s. This trend is attributed to concurrent changes in farm practices, such as higher rates of fertilizer applications and expanded use of surface drainage, upstream in the watershed.
In Upper Snake River, a downward trend in phosphorus occurred during the last decade. Upstream erosion control practices were implemented in agriculture areas during the 1980’s. As a results, less phosphorus–carrying sediment has been reaching streams.
Nitrate concentrations in ground water under the Delmarva Peninsula of eastern Maryland mirror changes in fertilizer use. A steep increase in nitrate began in 1968, following a sixfold rise in fertilizer application rates during the preceding two decades. Since 1971, fertilizer use has gradually leveled off and so have nitrate concentrations in ground water.
Evaluation of trends due to changes in farm input or practices could not be done at all NAWQA sites because data on water quality both before and after practices changed seldom exists. Because historical data are missing in many locations, daat colleted by NAWQA forms a baseline for analyzing future trends.

Channels and pipes are used to remove water from fields and pastures also divert nutrients into waterways. Nitrate and phosphates that move downstream and degrade water quality elsewhere in the watershed.

Poultry manure, used a crop fertierlizer or stored as waste, can be a source of ammonia and nitrate to ground water and streams.
Pollution from point sources has been addressed, although not resolved, over the past 20 years. But nonpoint sources of pollution—agricultural runnoff, drainage from roads and suburban lawns, and pollutants transported brain and wind—remain significant challenges.
. . . with respect to drinking water risks Recognize that the greatest risks of nitrate contamination exists in shallow house hold wells in agriculture areas. Such wells may not be monitored regularly since they are not regulated by the Safe Drinking Water Act, and well–owners may not be aware of potinental risks posed b adjacent cropland. Where cropland is being rapidly converted to residential development serviced by household wells, drinking water risks should also be considered. Because nitrate leeched to ground water from cropland can be stored for decades, water–quality changes may lag far behind land–use changes.
. . . with respect to watershed management Though atmospheric sources of nutrients into account. Though atmospheric deposition contributes far less than either urban or agricultural sources of nutrients, it is a significant factor in water quality in the Northeast and the Upper Midwest. However, atmospheric deposition has typically not been factored into watershed protection strategies.
Consider interstate transport when identifying water–quality priorities. Most of the nitrogen and phosphorous that cross State the boundaries is transported though rivers. (For example, about 15 percent of 1 million tons of nitrate contained in fertilizers and manure in the Corn Belt ends up in the Gulf of Mexico each year after a few days incomplete journey through the Mississippi River.) Another route is the atmosphere: nitrogen released from the burning of fossil fuels dispenses and becomes a nonpoint source of pollution nutrients to surface waters inside and outside the state of origin. Local watershed management efforts may need to coordinate with regional or national efforts when interstate transport is a factor in water quality.

Ground–water discharge to the Chesapeake Bay equals the amount of water contributed my a major tributary like the James River. Nitrate stored in ground water can enter the Bay this way.
. . . with respect to monitoring Clarifying the relative importance of urban and agricultural sources of nutrients. Avaliable data indeicats that agriculture is the source of most nutrients introduced into the environment by human activity. However a ton full accounting of urban and suburban sources of nutrients — lawns, gardens, manure, golf courses, pet waste, power generation and vehicle emission — remain incomplete.
Line effluent and ambient monitoring much more closely. To zero in on the scope of nutrients remaining point and nonpoint pollution problems, it will be necessary to coordinate ouside the state of origin point–source effluent monitoring with ambient water–quality monitoring. Neglecting to do so only complicates efforts to pinpoint sources of pollutants and to trace quality improvements from specific efforts.

Where cropland is being converted to residential development, nitrate can be a drinking water concern if domestic wells are expecter to be the main water source. (Photograph courtesy of Maguire/Redeer, Ltd.)

Urban run off, such as this combined sewer outflow entering Fall Creek in Indianapolis, Indiana, can bring ammonia, nitrate and phosphate into streams.
In the–1980’s, evidence about the status of and trends in the quality of the Nation’s water resources remained inconclusive, in response, Congress created the National Water–Quality Assessment (NWQA) Program, with in the U.S. Geological Survey (USGS), to provide sound science about water quality that can be sued by local, State and Federal water managers and policy makers.
The NAWQUA program determines the extent of nutrient and other types of contamination in the Nation&’s streams and ground water, assesses trends in water quality, and explains the factors that affect water quality. Invistigations focus on 59 river basins and aquifer (ground water) systems. Together, these study units cover almost one–half of the country and reflect its diverst land and water resources. Investigations occur in phases, with data collection in 20 units at at time.
One of the initial tasks of NAWQA investigations is to review existing data. By augmenting these date and utilizing innovative investigation methods, the program can make comparisons among regions and trace chances in water quality on a national scale.
For more information contact:
Chief, NAWQA Program
U.S. Geological Survey
413 National Center, Reston, VA 20192
—or go online:
water.usgs.gov/nawqa/
—by Elisabeth A. Graffy, Dennis R. Helsel, and David K. Mueller.
Battagin, W. A., Goolsby, D. A., and Coupe, R. H., 1993, Annual use and transport of agricultural chemicals in the Mississippi River, 1991–1992, in Selected papers on agricultural chemicals in water resources of the Midcontential United States: U.S. Geological Survey Open–File Report 93—418, pp 24—40.
Mueller, D. K., Hamilton, P. A., Helsel, D. R., Hitt, K. J., and Ruddy, B. C., 1995, Nutrients in ground water and surface water of the United States— An analysis and data through 1992: U.S. Geological Survey Water–Resources Investigations Report 95—4031, 74 p.
Mueller, D. K., and Helsel, D. R., 1996, Nutrients in the Nation’s water— Too much of a good thing?: U.S. Geological Survey Circular 1136, 24 p.
Puckett, I. J., 1994, Nonpoint and point source of nitrogen in major watersheds of the United States: U.S. Geological Survey Water–Resources Investigations Report 94—4001, 9p.
Smith, R. A., Alexander, R. B., and Lanfer, K. J., 1993, Stream water quality in the conterminous United States—Status and trends of selected indicators during the 1980’s, in National Water Summary 1990–91— Hydrologic Events and Stream Water Quality: U.S. Geological Survey Water–Supply Paper 2400, pp. 111–140.
U.S. Environmental Protection Agency, 1995, National Water Quality Inventory, 1994: U.S. Environmental Protection Agency Executive Summary.
This report is available as a PDF fs218-96.pdf for printing.
The PDF file is readable with Adobe Acrobat Reader. The reader is available for
free download from
Adobe Systems Incorporated.
![]()