
Radiogenic Isotopes and the Eastern Mineral Resources Program of the U.S. Geological Survey
Radiogenic isotope geology relies on a small group of naturally occurring but unstable elements that naturally produce energy and disintegrate or decay to more stable atoms of different elements. Because this phenomenon occurs at a known rate, is unaffected by most physical and chemical processes, and takes place over periods of time comparable to the age of the Earth, these elements can be used in radiometric dating or geochronology.
Radioactive Decay and Age Determinations
An atom of an element is composed of a nucleus, which contains virtually all the mass in an atom, and electrons (negatively charged particles) orbiting around the nucleus (Faure, 1986; Dickin, 1995). The nucleus consists of two kinds of atomic particles: neutrons (electrically neutral particles) and protons (positively charged particles).
Differences in the number of protons in the nucleus are used to distinguish elements so that the total number of protons represents the element's unique atomic number. The sum of the masses of protons and neutrons in the nucleus represents the atomic weight of an atom. Although all atoms of a given element have the same atomic number, atoms of a given element may contain different numbers of neutrons in the nucleus; such atoms of a given element that differ only as a function of the number of neutrons in the nucleus are known as isotopes. Lead (symbol Pb; atomic number 82) has four naturally occurring isotopes of different masses (204Pb, 206Pb, 207Pb, 208Pb). Only the isotope of Pb with mass 204Pb is stable; 206Pb and 207Pb, on the other hand, are daughter products resulting from the radioactive decay of uranium (U), and 208Pb is a product from thorium (Th) decay. As a result of decay, the heavier isotopes of lead will increase in abundance compared to 204Pb.
The half-lives of these decays -- the time necessary for the original number of atoms of a radioactive element in a rock to be reduced by half -- are relatively well known and range from hundreds of millions to billions of years. Small amounts of radio- active elements (for example, potassium, K; rubidium, Rb; samarium, Sm; lutetium, Lu; rhenium, Re; uranium, U; and thorium; Th) used for age determinations are found in common minerals, rocks, soils, sediments, and fluids. Geologists are able to read Nature's radioactive clocks as follows:
- by extracting and purifying the radioactive parent and daughter from the relevant rock or mineral,
- by precisely measuring variations in the masses of the parent and daughter isotopes using machines known as mass spectrometers (figs. 1, 2), and
- by combining the isotope abundances with the known rates of decay to calculate an age.
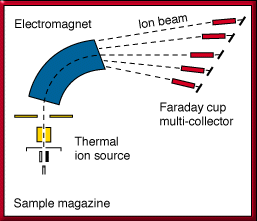
Figure 1. Features of a mass spectrometer. A mass analysis is obtained by volatilizing the element placed in the thermal ion source of the machine, which is kept at high vacuum. The vapor is then ionized and accelerated through a magnetic field produced by an electromagnet. The ion beam travels through the analyzer tube and then goes to a collector, where it is measured.
|

Figure 2. Automated, nine-collector Finnigan-MAT 262 mass spectrometer equipped with ion-counting capabilities. This spectrometer is used by the U.S. Geological Survey in Reston, Va., for research. Photograph by David Usher.
|
As the physical laws governing radioactive decay are universally obeyed, radiogenic isotope dating techniques represent a quantitative method to reliably establish the age of materials in Nature, applicable even to the oldest objects on the Earth, the Moon, meteorites, and asteroids and, potentially, in the rest of the universe.
Isotopes used for age determinations include radiogenic Sr (strontium produced by decay of radioactive rubidium), Nd (neodymium from decay of samarium), and Os (osmium from decay of rhenium); application of all radiogenic systems requires knowledge of the appropriate half-lives and chemical analyses to detect the relative abundances of parents and daughters. Isotopic dating is useful for all types of rocks having a closed system -- geologists assume that the age is an estimate of the time elapsed since the radioactive parent became part of the rock or mineral and that no parent elements or daughter products have been added or lost.
Isotopic Tracer Geology
An increasingly important application of radiogenic isotopes is in their use as tracers of otherwise undetectable contributors to composite, complex, and heterogeneous environments. Radiogenic isotope tracer applications using Pb, Sr, and Nd, among others, rely on the fact that these are heavy isotopes -- thus, in contrast to lighter isotopes such as hydrogen (H), oxygen (O), and sulfur (S), they remain unaffected by the following conditions:
- changes in temperature and pressure during transport and accumulation,
- different rates of chemical reactions, and
- the presence of different chemical species available in the environment.
The behavior of heavy isotopes means, for example, that chemical reactions and processes involving the element Pb will not discriminate among the naturally occurring isotopes of this element on the basis of atomic mass differences (204Pb, 206Pb, 207Pb, 208Pb). As a result of these special attributes, radiogenic isotopes are routinely used to study the architecture and evolution of the continents and oceanic rocks, to learn about unexposed material in the interior of the Earth that feeds volcanoes, and to establish the evolution of seawater through geological time.
In the Eastern Mineral Resources Program of the U.S. Geological Survey (USGS), radiogenic isotope tracers have been used traditionally as follows:
- to establish the identity of reservoirs supplying metals of economic interest,
- to provide information on the origin of rocks hosting ore deposits in different geologic environments,
- to provide insights into the genesis and evolution of mineralizing systems in order to explain a region's metal endowment,
- to recognize fluids involved with the formation of ore deposits, and
- to catalog economic ore deposits having similar isotopic compositions to facilitate recognition of differences between economic and subeconomic deposits in the same region.
New avenues of USGS research have also been initiated that will use radiogenic isotopic tracers to evaluate processes leading to the availability of metals from mineral deposits to the surface environment and to gage the effect and toxicity of mine areas on their immediate surroundings.
Innovative applications of heavy-isotope tracers have flourished in fields as varied as public health and environmental research. A health application is to identify from among possible reservoirs the one responsible for lead pollution in children. Environmental research can trace transport pathways and assess the relative environmental impact of human activities; for example, by determining the origins of industrial wastes contributed to complex natural environments.
Analytical Capabilities in Reston, Va.
The Reston, Va., isotope laboratory of the USGS is currently capable of analyzing most geological materials, including rocks, silicate minerals, sulfides, and sediments, in an ultraclean, lead-free environment for both geochronology and isotope tracer applications. Three radiogenic systems are routinely used: U-Th-Pb, Nd-Sm, and Rb-Sr. Research projects use an automated, nine-collector Finnigan-MAT 262 mass spectrometer equipped with ion-counting capabilities (fig. 2). The mass spectrometer is presently configured for automated analysis of lead, strontium, neodymium, uranium, thorium, rubidium, samarium, and boron.
Carolina Slate Belt Gold Deposits
The main objective of the current USGS studies of the Carolina slate belt in Virginia, North Carolina, South Carolina, and Georgia is to explain the origin of the gold accumulations. In order to do this, scientists are attempting to establish the age of the hydrothermal events, identify the nature of the mineralizing fluids responsible for mobilization, transport, and deposition of metals in the magmatic and hydrothermal stages, and ultimately explain the impact of toxic metal fluxes as a result of surface processes occurring at ambient conditions (Ayuso, 1998).
U-Th-Pb, Nd-Sm, and Rb-Sr dating is being done on sulfides, silicate minerals, and rocks in our Reston labs, and U-Th-Pb zircon dating is being done as part of a collaborative study between the USGS and VPI (Virginia Polytechnic Institute and State University). New isotopic data have shown that the gold deposits are Early to Middle Cambrian (about 560 million years old) and have different ranges in Pb, Sr, and Nd isotopic compositions. All of the major gold deposits (Haile, Brewer, Ridgeway, and Barite Hill) in the slate belt show evidence of gold mobilization after the original formation of the deposits. Another fundamental observation as a result of the radiogenic isotope studies, critical for successful exploration for gold deposits in the Eastern United States, is that the gold mines have distinctive isotopic signatures that uniquely identify the rocks involved with their genesis.
Pb and Nd isotopic compositions demonstrate that the gold deposits are underlain by root rocks (basements) that are not found west of the Carolina slate belt but that occur only as part of a trend that stretches to the northeast and southwest for hundreds of miles. This presence of basement rocks is a feature of considerable importance that will lead to more precise regional exploration and assessment of the endowment of precious metals in the Carolina slate belt and adjacent regions. Ongoing research will improve the understanding of the factors controlling the distribution of the gold deposits in the Eastern United States by documenting their characteristic geologic, geochemical, and geophysical features and will facilitate comparisons of the slate belt with deposits in adjacent regions.
Base- and Precious-Metal Deposits in New England
Current applications of the properties of radioactive elements are also being used to provide better resolution on the chronology of formation of the New England massive sulfides, to explain their regional distribution as well as their size and number, and to identify large regional tracts most likely to contain additional economic deposits.
The USGS work is currently focused on the Bald Mountain massive sulfide deposit in northern Maine and consists of U-Th-Pb zircon dating of key units, in addition to Pb-Nd-Sr isotopic studies of volcanic rocks hosting the ore deposits. Isotopic tracer studies on sulfide minerals seek to establish constraints on the possible sources of the metals and on the contributions of different types of mineralizing fluids.
Cooperative Studies, International Studies, and Other Outreach Activities
A pilot cooperative study was initiated recently between the Mineral Resources Program and the Water Resources Division of the USGS; the study is focused on the application of radiogenic isotopes to improve our understanding of the effects of human activities on water quality. Radiogenic isotope tracers can be powerful tools to look for environmental and geochemical pathways, to establish the relative timing of chemical contributions to urban water reservoirs, and to discriminate between results of natural processes and results of human activities in the near-surface environment. In the White Rock Lake reservoir, Texas, recently acquired lead isotopic data document large isotopic shifts in the compositions of sediments. The isotopic compositions are significantly different in sediments deposited since 1976 and in sediments deposited before 1952. The results parallel the documented chemical variations in response to changes in land use and, particularly, the introduction of unleaded gasoline in the 1970's.
The radiogenic isotope laboratory in Reston, Va., is also involved with outreach, technology transfer efforts, and training of guest investigators from within the USGS and from Europe and Latin America. Current collaborative work includes studies on the metallogeny of alkalic rocks from Italy and on precious-metal deposits in Mexico.
Metallogeny and Basement Terranes
Radiogenic isotope studies have made important contributions to the understanding of mountain building events and provided key observations explaining the origin and evolution of major blocks of continental crust in the Eastern United States. Detailed isotopic information is vital for assessing the metal endowment of rocks in a given region because crustal blocks commonly have contrasting isotopic compositions.
Isotopic variations of Pb, Nd, and Sr have been used to determine the internal variations within large rock masses in New England and to identify zones associated with anomalously high contents of economically important elements (tin, molybdenum, and tungsten). Isotopic and geochemical studies indicated that rocks associated with copper-molybdenum mineralization in New England originated from crustal blocks and sources that were fundamentally distinct from sources of rocks containing tin-tungsten and rare-earth-elements-uranium mineralization. More importantly, Pb and Nd isotopic compositions have been used in recent studies to uniquely characterize the types of root rocks (basements) underlying the crustal blocks that make up the Appalachian Mountains. Recognition of such key isotopic differences in unexposed rocks from deep within the Earth and associated with the mechanisms of mountain building provides powerful insights into the evolution of continental land masses.
References on Radiogenic Isotope Research
General References
Dickin, A.P., 1995, Radiogenic isotope geology: Cambridge, U.K., Cambridge University Press, 452 p.
Faure, Gunter, 1986, Principles of isotope geology: New York, John Wiley and Sons, 589 p.
Koppel, V., and Grunenfelder, M., 1979, Isotope geochemistry of lead, in Jager, E., and Hunziker, J.C., eds., Lectures in isotope geology; Berlin, Springer-Verlag, p. 132-153.
Selected Articles of Interest
Ayuso, R.A., 1998, Carolina slate belt gold deposits in Virginia, North Carolina, South Carolina, and Georgia: Reston, Va., U.S. Geological Survey Information Handout, 2 p.
Ayuso, R.A., and Bevier, M.L., 1991, Regional differences in lead isotopic compositions of feldspars in plutonic rocks of the northern Appalachian Mountains, U.S.A. and Canada: A geochemical method of terrane correlation: Tectonics, v. 10, p. 191-212.
Ayuso, R.A., DeVivo, B., Rolandi, G., Seal, R., and Paone, A., 1998, Geochemistry, metallogenesis, and isotopic (Nd-Sr-Pb-O) variations bearing on the genesis of volcanic rocks from Vesuvius, Italy: Journal of Volcanology and Geothermal Research, v. 82, nos. 1-4, p. 53-78.
Ayuso, R.A., and Foley, N.K., 1993, Rb-Sr isotopic redistribution and character of hydrothermal fluids in the Catheart Mountain Cu-Mo deposit, Maine, in Scott, R.W., Jr., Detra, P.S., and Berger, B.R., eds., Advances related to United States and international mineral resources: Developing frameworks and exploration technologies: U.S. Geological Survey Bulletin 2039, p. 45-57.
Ayuso, R.A., and Smith, R.L., 1994, Pb isotope compositions of the Bandelier Tuff, Valles Caldera, Jemez Mountains, New Mexico: An active geothermal system associated with Mo mineralization, in Berger, B.R., ed., Advances in research on mineral resources: U.S. Geological Survey Bulletin 2081, p. 3-11.
Foley, N.K., and Ayuso, R.A., 1994, Paragenetic constraints on the Pb isotopic evolution of the North Amethyst Au-Ag vein, Creede mining district, San Juan volcanic field, Colorado: Economic Geology, v. 89, p. 1842-1859.
Van Metre, P.C., and Callender, E., 1996, Water-quality trends in White Rock Creek Basin from 1912-1994 identified using sediment cores from White Rock Lake reservoir, Dallas, Texas: Journal of Paleolimnology, v. 10, p. 1-11.
|
For more information, please contact:
Robert A. Ayuso
U.S. Geological Survey
|
Mail Stop 954, National Center
Reston, VA 20192
|
Telephone: (703) 648-6347
E-mail: rayuso@usgs.gov
|
U.S. Department of the Interior
U.S. Geological Survey
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
USGS Information Handout
October 1998
|
Any use of trade, product, or firm names in this handout
is for descriptive purposes only and does not imply endorsement
by the U.S. Government.
|
This page is https://pubs.usgs.gov/info-handout/ayuso2/
Maintained by Eastern Publications Group Web Team
Last revised 12-4-98 (krw)


