
Environmental Processes that Affect Mineral Deposits in the Eastern United States
Introduction
A thorough understanding of the environmental processes that affect mineral deposits and mine wastes has become increasingly important as the Nation wrestles with how to meet our current demand for metals without compromising the environment and how to mitigate the damage caused by the mining practices of previous generations. Regulatory requirements are dominated by empirical approaches to environmental problems associated with mining, but mitigation and reclamation can be enhanced greatly by a theoretical and conceptual understanding of the processes that affect the availability, transport, and fixation of metals and the generation of acidic waters.
U.S. Geological Survey (USGS) research efforts in the Eastern United States are concentrating on environmental processes that affect a class of mineral deposits known as massive sulfide deposits. These occurrences were valued historically for their sulfur content and recently for their metals. This deposit type is a research priority because of its economic significance and high potential for adverse environmental impact due to its high sulfide content and the low acid-buffering capacity of host rocks. Numerous examples of these deposits are found in the East, including reclaimed mine sites, abandoned mines, active mines, and sites currently in the permitting process for future production.
 Published studies of mine drainage chemistry from the Iron Mountain massive sulfide deposit in California have documented extreme conditions of very low pH and high heavy-metal concentrations. These extreme conditions are attributed to the unique hydrologic and climatic settings of the deposit and probably are independent of the mineral deposit type.
Published studies of mine drainage chemistry from the Iron Mountain massive sulfide deposit in California have documented extreme conditions of very low pH and high heavy-metal concentrations. These extreme conditions are attributed to the unique hydrologic and climatic settings of the deposit and probably are independent of the mineral deposit type.
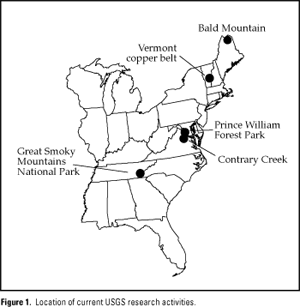
Areas currently under study include Bald Mountain, Maine, the Great Smoky Mountains National Park, the Vermont copper belt, Contrary Creek, Virginia, and Prince William Forest Park, Virginia (fig.1). Goals of the research are (1) to give land-use planners and the mining industry a better empirical framework from which to assess potential environmental impacts of mining, particularly under eastern climatic conditions, and (2) to provide a better theoretical and conceptual framework from which to design more effective and cost efficient mitigation and reclamation programs.
Research Strategies
The general strategy of current research is to use an integrated, multidisciplinary approach to understand the geologic, geochemical, and biogeochemical processes that affect the environmental behavior of these deposits and their mining wastes. Field studies emphasize the interaction of water and rock, as mediated by biological activity. Water sampling involves U.S. Environmental Protection Agency-approved protocols. Onsite measurements are made of pH, specific conductance (a measure of the total dissolved solids), temperature, flow rate, and numerous unstable species such as dissolved oxygen, dissolved iron, phosphate, and nitrate. Solid sampling includes country rocks, ores, mine drainage precipitates, solid mine wastes, soils, stream sediments, and biota. Site studies also include soil pH and geoelectrical surveys. Laboratory analysis includes a variety of techniques to determine mineralogical and chemical compositions of solids, dissolved constituents of waters, and stable isotope compositions of solids and waters. Low-temperature experimental capabilities include calorimetric and solubility studies.
Bald Mountain Massive Sulfide Deposit, Maine
The Bald Mountain copper, zinc, gold, and silver deposit in northern Maine presents an unparalleled opportunity to understand the environmental behavior of this deposit type because Bald Mountain was discovered in an area that has been untouched by mining. Thus, all of the present-day geochemical anomalies in the soil, stream sediments, and surface and ground waters surrounding the deposit are the result of natural processes, unrelated to human activity. The Black Hawk Mining Company is seeking a permit to mine a gold-rich portion of the deposit. Black Hawk and its predecessors have characterized the geochemical and biological environment around the deposit, as an aid to setting responsible standards for post-mining reclamation.
The USGS wants to determine how much of the natural groundand surface-water baseline of heavy metals, dissolved sulfate, and acid is derived from the natural weathering of the deposit and how much is derived from anthropogenic sources such as acid rain. This task is being accomplished by two approaches. First, mineralogical and geochemical characterization of transects of weathered ore (known as gossan or oxidized ore), the zone of secondary enrichment (known as supergene ore), and the primary unweathered ore will be used to determine the long-term weathering behavior of the deposit. Second, stable oxygen, hydrogen, and sulfur isotope data for surface and ground waters from the area will be used to fingerprint sources of water and sulfate in the hydrologic system surrounding Bald Mountain. Preliminary results indicate a significant atmospheric contribution of sulfate in the lakes around Bald Mountain.
Great Smoky Mountains National Park
Within the Great Smoky Mountains National Park (GSMNP) in southwestern North Carolina, two significant abandoned copper mines, Fontana and Hazel Creek, are point sources of acid mine drainage (pH as low as 3.1 and total heavy-metal contents as high as 30 mg/L). Additional complications arise because these mineral deposits are hosted by a regionally extensive pyritic schist that is itself a source of natural (and human-induced) acid rock drainage. Watersheds underlain by the schist have pH values less than 5.0, and the acid conditions are known to adversely affect fish populations. As with Contrary Creek, Virginia, drainage from the Fontana mine flows a short distance and empties into a popular recreational lake -- in this case, Tennessee Valley Authority Fontana Lake. The USGS is also interested in assessing the impact of acid drainage on the lake.
Vermont Copper Belt
Copper mining has been historically important to the economy of Vermont, but today, drainage from abandoned mines is negatively affecting the environment. The USGS is cooperating with a local citizens group, the Elizabeth Mine Study Group, to evaluate acid mine drainage around the Elizabeth mine near South Strafford; the evaluation could serve as the basis for future reclamation of the site. Mining began at the Elizabeth in the late 18th century and continued intermittently until 1958. Similar, but smaller, mines were also in operation north of South Strafford near West Fairlee and Corinth. Today, waters draining the Elizabeth mine workings and waste piles are acidic and laden with toxic heavy metals.
USGS studies are focused on understanding the chemical and hydrological processes by which the rocks and waste piles weather to produce acid mine drainage. Cooperative efforts with the Elizabeth Mine Study Group are concentrating on assessing toxic heavymetal loads from the upper part of the watershed. To date, the USGS has helped train the group in water sampling protocols and is providing analytical support and assistance in data interpretation. Reconnaissance studies are underway at other mines in the Vermont copper belt. In addition to the Elizabeth Mine Study Group, USGS efforts are being coordinated with a variety of State and Federal agencies, including the Vermont Department of Environmental Conservation, U.S. Department of Energy, U.S. Army Corps of Engineers, U.S. Environmental Protection Agency, and Dartmouth College.
Contrary Creek, Virginia
Contrary Creek drains the Mineral District of central Virginia and empties into Lake Anna. The mines in the area are abandoned and have received minimal reclamation attention. Waters issuing from the mine sites have pH values as low as 2.7, heavy-metal concentrations of up to 300 mg/L, and sulfate concentrations of up to 2,300 mg/L. In spite of this poor water quality, a diverse algal and bacterial population is thriving in the acidic waters.
The surface waters fall into two categories, those with surface discharge and ponded waters without surface discharge. Research to date demonstrates that the acidity and heavy-metal content of these waters are the result of a combination of processes, including oxidation of pyrite and other sulfide minerals, precipitation of iron oxide/hydroxide minerals, dissolution of previously precipitated secondary heavy-metal salts, evaporation, mixing with dilute precipitation and near-neutral surface runoff, and algal and bacterial interactions. Future work will concentrate on the impact of acid mine drainage on Lake Anna.
Prince William Forest Park
Volcanic rocks that host the Mineral District extend into northern Virginia where they contain several massive sulfide deposits. One of these deposits, at the Cabin Branch pyrite mine on the banks of Quantico Creek, is in Prince William Forest Park, part of the National Park system. In 1995, the National Park Service reclaimed the site and installed ground-water monitoring wells. Prior to reclamation, surface water pH values as low as 2.6 were reported, with heavy-metal concentrations in excess of 25 mg/L. The poor water quality was attributed to the weathering of mine tailings (fine-grained pyritic waste) piled on the surface. In cooperation with the National Park Service, the USGS is assessing the reclamation effort through an integrated characterization of groundand surface-water chemistry in the vicinity of the mine. In addition to water chemistry, the site studies will include mineralogical and geochemical char acterization of the mine wastes to identify the dominant processes of pyrite oxidation, acid generation, and heavy-metal mobilization. Related work in collaboration with the National Park Service is investigating the relationship of water chemistry in seasonal ponds in drainage trenches at the reclamation site to the restoration of the indigenous amphibian population.
Experimental Geochemistry
One of the greatest limiting factors in predicting the geochemical behavior of mine drainage water is the poor understanding of the stabilities of minerals and other compounds that interact with ground and surface waters around mineral deposits. The need for a more thorough knowledge of the stabilities of these compounds is essential (1) for understanding the conditions of formation of acid mine drainage and predicting how climatic variations might change the severity of the problem and (2) for designing effective remediation strategies.
Stability data for a class of compounds known as efflorescent salts are notably limited. Efflorescent salts are highly soluble, hydrated, heavy-metal sulfate compounds that have been shown to be very important for storing heavy metals and acid during dry periods in acid mine drainage settings. Dur ing rain storms or spring melting of snow packs, these salts readily dissolve and release large amounts of heavy metal and acid to the watershed. Mineralogic studies by the USGS have identified many of these minerals at study sites in the East.
Compilations of existing data demonstrate that the stabilities of some heavy-metal salts vary systematically and predictably. This observation is being used to estimate stability parameters for compounds for which there is no information. New experimental studies are aimed at verifying the estimation procedures and at measuring stability parameters for compounds that are not amenable to the estimation procedures. Ultimately, the new experimental data will be used to model the chemistry of acid mine drainage waters and their interactions with surrounding rocks and watersheds.
|
For more information, please contact:
Robert R. "Bob" Seal II
U.S. Geological Survey
954 National Center
Reston, VA 20192
Telephone: (703) 648-6290
Fax: (703) 648-6383
E-mail: rseal@usgs.gov
|
This page is https://pubs.usgs.gov/info-handout/seal1/
Maintained by Eastern Publications Group Web Team
Last revised 12-21-99
 Published studies of mine drainage chemistry from the Iron Mountain massive sulfide deposit in California have documented extreme conditions of very low pH and high heavy-metal concentrations. These extreme conditions are attributed to the unique hydrologic and climatic settings of the deposit and probably are independent of the mineral deposit type.
Published studies of mine drainage chemistry from the Iron Mountain massive sulfide deposit in California have documented extreme conditions of very low pH and high heavy-metal concentrations. These extreme conditions are attributed to the unique hydrologic and climatic settings of the deposit and probably are independent of the mineral deposit type.

