
U.S. Geological
Survey
Open-File Report 00-369
ONLINE ONLY
Version 1.0
Hands-on Experiments To Test for Acid Mine Drainage
Dr. Eleanora I. Robbins
U.S. Geological Survey
956 National Center
Reston, VA 20192
EERSC Team
|
Sandra L. McSurdy
Department of Energy
Pittsburgh Research Center
P.O. Box 10940
Pittsburgh, PA 15236
SANDRA.MCSURDY@netl.doe.gov
|

|
Timothy Craddock
West Virginia Division of
Environmental Protection
Office of Waste Management
1356 Hansford Street
Charleston, WV 25301
tcraddock@mail.dep.state.wv.us
|

|
This report is preliminary and has not been reviewed for conformity with U.S. Geological Survey editorial standards or with the North American Stratigraphic Code. Any use of trade, product, or firm names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Science Experiments for Kids Living
Where Creeks are Orange, Yellow, or Red

Contents
You may browse the entire
document by using the scrolling arrows to the right of the page, or you
may go directly to the experiment you are interested in by clicking on
the links below. Please read the safety tips below
before conducting any of these experiments in or around your stream.
We recommend that you read the Introduction
before choosing an experiment.
-
Introductions
by Eleanora "Norrie" Robbins
-
Make
your own litmus paper
-
What
is acid and how do you know it?
-
Clear
water -- Is it clean? - Aquatic
macroinvertebrates
-
Why
does acid mine drainage form?
-
What
plants love acid water?
-
Who
is small and living in your creek?
-
What
is in your water?
-
How
many colors does iron have?
-
What
is the black on the rocks?
-
Is the
groundwater acid also?
-
What
is the white stuff in the water?
-
How
can acid mine drainage be fixed with natural things?
-
Using
stinky bacteria to treat acid mine drainage
-
Designing
your own experiment
Resources
for more information
Internet
links
"Thoughts
on Pollution": Tell us what you think by adding
your comments, ideas, and suggestions.
The
experiments in this document should always be done with the proper supervision
from a teacher, parent, or other adult. Always use caution and the proper
protective equipment when you are in or around a stream (recommendations: waders or boots, gloves, safety
glasses). Additional safety tips are listed
below. Always make sure you have permission before entering private property,
and wear shoes with rubber soles and good traction when in and around a stream.
Safety Tips: Always
use the buddy system, and have an adult check the stream flow and the stream
bottom for sharp objects before entering a stream. Do not enter the stream
if you cannot see the bottom, and do not enter after a hard rain. Wait for
several days of dry weather before conducting any outside experiment in
or around a stream. Do not go stream collecting without the presense of
a responsible adult!
956 National Center
Reston, VA 20192
Dear
Kids, Parents, Guardians, Teachers, and all Concerned Citizens,
This is a prototype for a
new book we want to write for kids. This is the first draft, and we want
kids to help write it. If they do, their names and comments will appear
in the book. The deadline for submissions is spring 2001. Right now, we
would love to hear your questions. What questions do you have about acid
mine drainage, colors in the water, critters in the water, or any other
water quality questions? Our addresses are on the front page.
My sister and I wrote a science
book for kids in 1992. (It is now out of print by the federal government
and only available from the Colorado School of Mines.) It is called "What's
Under Your Feet?" We talked to many scientists while we were writing the
book. One fascinating finding was that most scientists find their vocation
by age 8 or 9. This means that when they were children, they were making
observations about their environment. These observations were so powerful
that they formed the basis for understanding of how the world works. Now,
not everyone is going to become a scientist. But everyone is going to
enter the job market. It is my opinion that environmental cleanup is going
to provide many jobs in the future. I think that if we get the kids out
and looking and getting dirty now, they will have a body of observational
knowledge needed to compete in that future job market.
The kinds of experiments
that are laid out here are some of the very things that scientists do when
they are trying to understand the natural environment and to help clean
up problems left from past activities. The observations that kids will
be making will also be helping present day scientists.
These are mine and others'
thoughts on why we started putting together these experiments. We are outdoors
people, so most of these experiments are for other outdoor lovers. However,
many can also be done indoors. Please remember that our objectives are
to help you learn about the effects of acid mine drainage on our environment
through experimentation and observation. As you try these experiments
and invent new ones at school and at home, record your observations, keep
in mind the principals of scientific experimentation, and, most importantly,
have fun.
Sincerely yours,
Dr. Eleanora (Norrie) Robbins
To download your copy of the entire document
of "Hands-on Experiments To Test for Acid Mine Drainage" in Adobe PDF format, simply click here:
Open-File Report 00-369 PDF [217 KB]
Making your own litmus
paper

The pH is a measurement (color
change or number) of how acidic or basic (alkaline) a chemical substance
may be. As you can see in the above picture, the pH scale ranges from 0
to 14, with 7 being the middle or neutral point. A substance with
a pH of less than 7 is acidic, and if it is greater than 7 the substance
is basic or alkaline. Each time the pH changes 1 unit there is
a tenfold increase or decrease in the strength of the acid or base being
measured. Litmus paper is one way to measure the strength of an acid
or base. Other ways include electronic pH meters and chemical tests kits
that measure the pH by comparing results to a color scale. For more
information and definitions, check the websites below.
Hint: Lemon juice
and vinegar are acids and should turn the paper pink; baking soda is a
base and should turn the paper green. If there are no color changes with
your test substances, this means that they fall somewhere close to the
middle of the pH scale.
Tools and Things you
will need
| Red cabbage |
Lemon juice |
Blender |
| 5 x 8 white card |
Vinegar |
Strainer |
| Plastic sheet to contain
the mess |
Baking soda |
Eye dropper |
What to do.
-
Pull off the cabbage leaves.
-
Press them down hard on the
white card until the card turns purple (6th grader Peter Cable
discovered that the purple in the leaves works the best).
-
When you have turned the whole
card purple, put a drop of each test substance on the card.
More things to do (You'll
need a blender and adult supervision).
-
Cut cabbage into chunks.
-
Blend the cabbage chunks into
a blender until liquid.
-
Strain the contents of the blended
cabbage with the strainer.
-
Mix a drop of this liquid with
the substance being tested.
http://www.encyclopedia.com/articles/00085.html
http:www.chem4kids.com/reations/acidbase.html
What is
acid and how do you know it?
Robert Angus Smith, an English
chemist, was the first to use the phrase "acid rain" in 1852. He
noticed that the bricks in many of the cities buildings were falling apart,
and through scientific experimentation he made a connection between London's
polluted skies and the pH of its rainfall. Most scientist agree that
normal rainfall is slightly acid with a pH of about 5.6. The rain
in the atmosphere reacts with carbon dioxide to form a weak carbonic acid
that gives the rain its lower pH. Scientist define acid rain as any
form of wet precipitation with a pH of less than 5.6. The rain
becomes more acid when the water molecules react with different gases in
the air such as sulfur dioxide and various nitrogen oxides. These
gases occur in the atmosphere naturally. However, their amounts have been
increasing for many years due to our many types of industrial processes
and the buring of fossil fuels.
Tools and Things you
will need
| Gloves |
Bowl |
| Measuring cup |
Litmus paper |
| Stirring rod |
Miscellaneous substances
* |
* vinegar, lemon juice, baking
soda, chalk, milk of magnesia, cola, coffee, etc. - use only a few drops
of each.
What to do.
-
Get a cup of water out of your
local creek and pour it into a bowl.
-
Wash and dry the measuring cup.
-
Test the water with litmus paper
(acids turn the paper pink).
-
Measure out one cup of baking
soda.
-
If the water is acid, slowly
stir baking soda into the bowl.
-
Test with new litmus paper periodically
until the paper turns blue. (This happens when the acid is neutralized.)
-
Repeat with a variety of acids
(lemon juice, cola, and coffee) and a variety of bases (milk, milk of magnesia,
and chalk).
What did you see?
-
How much baking soda was needed
to neutralize your creek water, how about cola or lemon juice?
-
What happened when you combined
two different acids?
-
What else did you see?
What do you think?
-
Which substance had the highest
pH (was the most basic), and which had the lowest pH (the most acidic)?
-
What else can you conclude?
Write down what you think.
http://antoine.frotburg.edu/chem/senese/101/
 If your
creek water is clear, is it clean?
If your
creek water is clear, is it clean?
Studying aquatic organisms
Each creek has a different
chemistry. Some are polluted, others are not. One way to learn your creek's
chemistry is to study the aquatic life (macroinvertebrates)
that live in the creek. Scientists and others have collected these organisms
from many places, and they have found that their presence can help to determine
what kind of chemical elements (pollutants)
are present in the creek and can give you a good idea of the health of the
creek.
Tools and Things you
will need
| Gloves |
Rubber boots |
Netting (fine mesh net) |
| Tweezers (forceps) |
Jar to collect organisms |
Litmus paper |
| Reference materials* |
|
|
* Reference sheets are available
from the web or from your state dvision of environmental protection.
The next section also shows some common aquatic organisms that may be found
in your creek.
What to do.
-
Test the pH of the creek water
with litmus paper. Write it down.
-
Find a stretch of your creek
that has a riffle. A riffle is a shallow rapid where water flows swiftly
over small rocks (cobblestones).
-
Stretch a net downstream from
the riffle.
-
Kick and rub the cobblestones
to get the organisms to fall off the rocks. Do this for 2 or 3 minutes.
(Make sure you rub the rocks upstream from the net.)
-
Collect the aquatic organisms
caught in the net and place them in a jar.
-
Lay out the aquatic organisms
and compare them to the pictures found in your reference material.
What did you see?
-
How many aquatic organisms came
off the cobblestones?
-
What kinds of organisms were there?
-
What were some other things
collected in the net?
-
What else did you see?
What do you think?
-
What information can you interpret
from the different types of aquatic macroinvertebrates present in the creek?
-
What are your conclusions? Write
down what you think.
This
section contains information to help you identify some of the more common
aquatic insects found in streams. For more information visit EPA's Biological
Indicators of Watershed Health website at http://www.epa.gov/ceis/atlas/bioindicators/
or Virginia's Save Our Streams website at http://www.sosva.com/.
Some common aquatic organisms (macroinvertebrates)
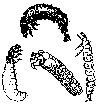
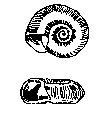
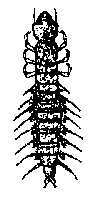
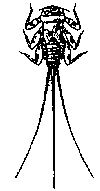


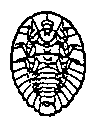
The aquatic organisms above
are generally pollution intolerant, which means they cannot live
in streams that are polluted, even in small amounts. Their presence in
large numbers is usually an indication of good water quality.



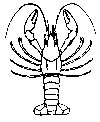

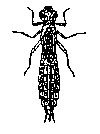


The aquatic insects above
are pollution sensitive, which means they can tolerate small amounts of pollutants. Their presence generally indicates moderately good
water quality conditions.
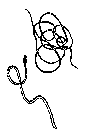




The aquatic insects above
are generally pollution tolerant, which means they can live in streams
that have high amounts of pollutants. Their presence in large numbers is
usually an indication of poor water quality.
Note: The macroinvertebrates
shown above are not to scale, which means they are not rhe actual size
as they will be if you find them in your creek.
Why does acid
mine drainage form?
The weathering process
In many areas of the country,
acid mine drainage forms naturally when certain materials come into contact
with water, air, and bacteria through a process called
weathering.
The weathering of rocks slowly releases acids, metals, and sulfates into
rivers, lakes, streams, wetlands and groundwater. The process may
be speeded up and the acid amounts increased (the environment contains
more acid than it can clean by natural processes) when industry does not
take the proper precautions to protect the environment. When too
much of these acids and minerals is released into creeks, creeks can
become polluted and will no longer support animals.
Tools and Things you
will need
| Limestone |
Tap water |
Small piece of coal |
| Litmus paper |
Metal ores * |
Bottles |
| Other types of rocks or
solid materials |
* such as iron, aluminum, or
magnesium available at local science and nature stores: http://www.worldofscience.com
What to do.
-
Collect solid materials that
are found where you live.
-
Add tap water to bottles and
measure the pH with litmus paper.
-
Add one type of solid material
to each of the water bottles. To get a good result, break the solid materials
into very small (crushed) pieces.
-
Put water but no solids in one
bottle. (This is called the control.)
-
Measure the pH of each bottle
over time and write down what you see.
What did you see?
-
What materials lowered the pH
of the water?
-
What materials raised the pH
of the water?
-
How long did it take for
acid to form?
-
What else did you see?
What do you think?
-
What materials in your areas
creek do you think can cause acid mine drainage?
-
How could you prevent acid mine
drainage from forming?
-
What are your conclusions? Write
down what you think.
http://cotf.edu/ete/modules/waterq/wqacidmine.html
 What
type of plants love acid water?
What
type of plants love acid water?
Very few plants
like acid conditions in creeks. But some
plants such as cattails can help change the chemistry by
cleaning up some substances that are carried into the water. These plants
work in cooperation with the bacteria in the soil to improve the coditions
in the water. Many scientists are testing these plants to learn how
they are able to do this. Scientists are also creating new environments,
such as wetlands,
that can help clean up the acid water (more about this in later experiments).
Tools and Things you
will need
| Small shovel or dowel |
Litmus paper |
| Rubber boots |
Gloves* |
| Magnifying lens |
Camera or drawing pad with
pencils |
* Caution: Some plants
have spines or sticky substances that can annoy the skin.
What to do.
-
Test the pH of the water with
litmus paper. (Is your creek acidic?)
-
Collect, draw, or take
photos of the plants growing along your creek. If you collect the
plants, only take one. If you find only one kind of a plant by your
creek, do not collect iit. We would prefer that you draw or take
pictures of the plants instead.
-
Take any plants that you collect
and press them in a phone book between sheets of newspaper.
-
Identify and make a list of
these plants.
-
Dig out one or two of the plants
to the root, or find their seeds to see if you can get them to grow somewhere
else (do not take any plants without permission).
-
Take a field trip to a wetland
area.
What did you see?
-
What types of plants grow along
the creek?
-
What kind and color of soil
did they grow in (hard, soft, squishy, wet, orange, black, or gray)?
-
What equipment did you need
to keep the plants alive?
-
What else did you see?
What do you think?
-
What information did the plants
give you?
-
What are your conclusions? Write
down what you think.
http://psybergate.com/wetfix/WetlandFix/WetlandFix4/Part4.htm
Who is very
small and living in your creek?
Looking at bacteria and algae
Many plants and animals may
not like acid conditions, but certain types of bacteria and algae do.
These small one-celled life forms collectively know as microorganims can
show a wide variety of colors. The colors are a result of the many
different types of chemical processes of which these life forms are capable.
For example, iron-oxidizing
bacteria are able to "remove" the dissolved iron in the water and form minerals
that look like rust. Many types of bacteria and algae use energy
from sunlight as a food source (similar to higher plants) in a process
called photosynthesis.
They can produce brilliant colors such as green, blue, purple, red, yellow,
or brown.
Tools and Things you
will need
| Baby food jar |
Eye dropper |
Gloves |
| Magnifying lens |
Rubber boots |
Litmus paper |
| Microscope (if possible) |
|
|
What to do.
-
With an eyedropper, collect
in baby food jars the different red, yellow, orange, or brown flocculates
in the water. Try filling some of the jars all the way to the top, and
leave an air space in some of the other jars.
-
Test each jar with litmus paper
to check its pH, and write down the results.
-
Put the jars on a windowsill.
(Put some jars on a south-facing windowsill - gets direct sunlight. Put
other jars on a north-facing windowsill - doesn't ever get sunlight). Certain
algae will probably develop in the jars receiving the sunlight, and certain
bacteria will develop in the jars that do not receive sunlight.
-
Observe over 2 or more weeks
and take the pH of the water regularly. Take notes of what you see
and be sure to include the dates and times of your observations.
What did you see?
-
Observe the water sample every
other day or so (if possible). Did swimming protozoans
hatch out?
-
Did any of the iron bacteria
colonize (coat) the jar?
-
Did any of the iron bacteria
form a reddish, oily looking film at the surface between the air and the
water?
-
Did any of the iron bacteria
form a brown ring at the top of the water?
-
What else did you see?
What do you think?
-
Did you collect acid loving
iron oxidizing bacteria or neutral iron bacteria? How can you tell the
difference?
-
What are your onclusions? Write
down what you think.
http://www.uga.edu/srel/Fact_Sheets/microbial_ecology.htm
What is
in your creek water?
A creek can carry an amazing
amount of chemicals. This is because the activities on the land
surrounding the creek (watershed) can affect what is in the creek. Some
of these chemicals may change the pH in a creek because they are so abundant.
When these chemicals are removed or fall from the water, such as when the
water passes through a wetland or over rocks, the water loses some of these
chemicals to the sediments. This may cause stains and different colors on
the bottom or on the rocks. When these chemicals fall into the sediments
by the actions
of rocks, bacteria, and plants, many times the result is a change in the creek's
pH.
Tools and Things you
will need
| Gloves |
Rubber boots |
| Litmus paper |
Access refrigerator and
stove |
| Small jar* |
hydrogen peroxide |
* Baby food jars, pill bottles,
etc.
What to do.
-
Fill a small jar or pill bottle
full with water from your creek.
-
Test the water for its pH with
litmus paper. Write it down.
-
Pour in one capful of hydrogen
peroxide.
-
Let the red flocculates
settle.
-
Test the water again for its
pH. Write it down.
-
Repeat the experiment with water
that has been kept in a refrigerator for 1 hour.
-
Repeat the experiment with water
left in a warm stove for 1 hour.
-
Repeat the experiment with baking
soda.
What did you see?
-
What happened when you put hydrogen
peroxide in the water? What about baking soda?
-
What happened to the pH?
-
Which reaction (refrigerator,
warm stove) occurred faster?
-
What else did you see?
What do you think?
-
What kind of chemical reaction
did you perform?
-
Does heat or cold speed up a
reaction?
-
What are your conclusions? Write
down what you think.
http://www.partstap.com/ground-water-and-drinking-water-an-expert-guide-for-kids.aspx
How many colors
does iron have?
 Iron
and the oxidation-reduction process
Iron
and the oxidation-reduction process
A common element in Appalachian
creeks is iron (Fe). Iron has many different forms and colors, and
each tells its own story about the chemistry of the creek where it is found.
Iron that occurs naturally in the creeks does not normally cause a problem,
but it can be increased by human activities to a point where it becomes
harmful to the life in a creek. The color of the iron in a creek
will show you what type of chemical reaction is occurring. If the
iron is red it is being oxidized, if it is black it is being reduced.
Tools and Things you
will need
| Gloves |
magic marker |
Rubber boots |
| Baby food jars with lids |
Shovel |
Litmus paper |
| Eyedropper |
What to do.
-
Fill jars with red, yellow,
or orange flocculates
and water from a creek. These can be found in many different creeks
but are more common in those affected by acid mine drainage.
-
Dig with a shovel in different
places along the creek bed until you find the change from red to black.
-
Add some of the black sediment to some of the jars and fill
all the way to the top to keep out oxygen.
-
Label the jars with the dates
of collection.
-
Put covered jars on windowsills and observe them over several weeks.
What did you see?
-
Make observations as to what
happened to the colors. Write down everything you observe.
-
At the end of the experiment,
uncover the jars and smell them. Write down what you smell.
-
What else did you see?
What do you think?
-
Which of these colors are from
iron?
-
Which iron was oxidized and
which was reduced?
-
What are your conclusions? Write
down what you think.
https://pubs.usgs.gov/gip/microbes/
What is
that black stuff on the rocks?
The manganese cycle
Finding metals that coat
the rocks is a very old profession. The prospectors of years ago
used to scrape off the coatings from rocks and send them to the laboratory
for analysis. The information would help them decide if they should
look upstream for such metals as gold and silver. The coatings and
the colors on the rocks can usually tell you what is the most abundant
mineral in the creek. Manganese is almost always the darkest mineral.
Tools and Things you
will need
| Rubber boots |
Gloves |
Magic marker |
| hand lens |
Litmus paper |
Glass slides |
| String or small rope |
Microscope (if possible) |
Miscellaneuos materials* |
* cans, bottles, tile, styrofoam,
paper, plastic, etc.
What to do.
-
This experiment requires that
your creek has cobblestones in it that are coated black.
-
Tie a string or small rope across
your creek. Attach some of the materials listed above to the string, allowing
them to dangle into the creek. (You may also attach them to tree roots and wooden stakes.)
-
Fill a large jar with creek
water collected near the riffles; drop a microscope slide into the jar.
Examine this slide at least once a week. Write down what you see and how changes occurred over time.
-
Write down what day you started
the experiment.
-
Examine the items dangling in
the creek at least once a week. Write down what you see and how changes
occurred over time.
-
After about 6 weeks, end the
experiment (write down the ending date) and make your final observations
and comments.
What did you see?
-
What materials became coated
with manganese?
-
What did you see on the microscope
slides?
-
Why do you think manganese sticks
to many things?
-
What else did you see?
What do you think?
-
Which materials do manganese
oxidizing bacteria like best?
-
What are your conclusions? Write
down what you think.
http://energy.er.usgs.gov/products/papers/wvsmdtf/index.htm
Is the groundwater
acid also?
 The surface water, mostly
from rain, runs off from the ground into creeks, rivers, lakes and wetlands.
A small amount of this surface water does not run off, but instead seeps
underground. This underground water is called groundwater.
Groundwater fills the spaces that are found in the soils and rocks and
eventually flows downhill, just like the creeks do. The place that
the groundwater is first observed is called the water
table. If you live near creeks that are polluted with acid mine
drainage, there is a good chance that the groundwater may also be polluted.
The surface water, mostly
from rain, runs off from the ground into creeks, rivers, lakes and wetlands.
A small amount of this surface water does not run off, but instead seeps
underground. This underground water is called groundwater.
Groundwater fills the spaces that are found in the soils and rocks and
eventually flows downhill, just like the creeks do. The place that
the groundwater is first observed is called the water
table. If you live near creeks that are polluted with acid mine
drainage, there is a good chance that the groundwater may also be polluted.
Tools and Things you
will need
| Shovel |
Gloves |
Rubber boots |
Hollow pipe* |
* Short length of PVC, steel,
or aluminum pipe.
What to do.
-
Test your creek's pH with litmus
paper.
-
Move away from the stream in
a straight-line (transect) and push a pipe down into the sediment, or dig
a hole with a shovel.
-
Collect the water from the hole
and test its pH with litmus paper.
-
Test another distance further
away from your creek.
What did you see?
-
How deep did you have to dig
to find water as you moved farther away from the creek? What does
this tell you about the water
table?
-
Was the underground water the
same chemistry as the creek?
-
What else did you see?
What do you think?
-
Would acids leak into the groundwater
from the creek or from other underground sources?
-
What are your conclusions? Write
down what you think.
http://www.groundwater.org/
What is the
white stuff in the water?
Both natural processes and pollution cause white colors in creeks. If the white is foam, foam is "a gas (usually oxygen) mixed in a liquid containing some impurity." If the white is aluminum, the pH is slightly acidic. If the white is sulfur, it is from surfur-oxidizing bacteria that live above sulfur-reducing bacteria. Sulfur-reducing bacteria give off hydrogen sulfide, which is the smell of rotten eggs.
Tools and Things you
will need
| Rubber boots |
Gloves |
| Microscope (if possible) |
Litmus paper |
| Eyedropper |
Shovel or stick |
| Baby food jars with lids |
What to do.
-
How can you tell if an impurity
is present in the environment?
-
Find out if your creek has white
foam.
-
Collect the white flocculate
in a baby food jar with an eyedropper.
-
Smell the mud under the white
flocculate. What do you smell? A sulfur smell may indicate
the presence of sulfur-reducing bacteria.
What did you see?
-
Look at the flocculates under
a microscope.
-
Do you see changes over time?
-
What else did you see?
What do you think?
-
What types of substances are
present in your creek? How can you find out what is in your creek?
-
What are your conclusions? Write
down what you think.
http://www.science.murdoch.edu.au/teaching/m234/recycle01.htm
How
can acid mine drainage be fixed with natural things?
"Passive
Treatment Methods" - The
next two experiments provide information about passive treatments.
Acid mine drainage sometimes
forms when minerals are exposed during mining. These minerals weather and
release the contents into nearby creeks which sometimes causes them to
become acidic (lower the pH). Acid drainage effects thousands of miles
of streams throughout the U.S. It can affect animals, plants, and small
organisms living in or near the stream. At the present time, many industries
use chemicals to treat the contaminated waters. However, less expensive
methods are also available to help clean up acid mine drainage.
Tools and Things you
will need
| Compost |
Litmus paper |
Limestone |
| Leaves |
Pine needles |
Cobblestones |
| Bottles with caps* |
Acid water from creek |
* You will need enough bottles,
and large enough bottles, for each object that you plan to test.
What to do.
-
Find an acid creek and use litmus
paper to measure the pH.
-
Collect the water in bottles.
-
Add one natural material (leaves,
pine needles, cobblestones) to each bottle. Measure the initial pH and
write it down.
-
Every few days for about a 2-week period, measure the pH to see what happens in each bottle. Write down
your results.
What did you see?
-
What materials decreased the
pH of the water?
-
What materials increased the
pH of the water?
-
What materials caused solids
to form at the bottom of the bottles?
-
What colors were the solids?
-
Did one of your experiments
change acidic water to neutral?
What do you think?
-
What natural materials may be
used to treat acid mine drainage?
-
Can you create a treatment system
using these natural materials?
-
What are your conclusions? Write
down what you think.
http://www.fetc.doe.gov/coolscience/teacher/lesson-plans/lesson10.html
Using
stinky bacteria to treat acid mine drainage
The sulfate-reduction process
Passive Treatment -- continued
As mentioned in the previous
experiment, there are many expensive ways to treat acid mine drainage
problems. Another alternative is a more passive treatment of
constructed and natural wetlands. In the wetland, a combination of
the plants, the holding capacity (how long the water will stay in the wetland),
the soil, and the bacteria are responsible for treating the acid mine
drainage. Many times these areas have a "rotten egg" smell, which
is hydrogen sulfide gas being released through biological reactions that
take place in the wetlands. This odor's presence is one way to tell if the wetland
is helping to treat acid mine drainage.
Tools and Things you
will need
| Acid water from creek |
Collection boxes* |
9 bottles with caps** |
| Litmus paper |
Wetland mud |
Shovel |
| Yeast |
* The plastic collection boxes
should be large enough for one small shovel full of mud.
** The bottles should be
large enough for at least a tablespoon of mud and a half of a tablespoon
of yeast.
What to do.
-
Collect acid mine drainage in
9 bottles.
-
Collect mud that
smells like rotten eggs from a wetland.
-
Add mud to 6 of the bottles
and yeast to 3 of the bottles.
-
Measure the pH in the bottles
over time, label each bottle, write down its pH, and watch for different
colors to form.
-
Smell each bottle and write
down what you smell (rotten eggs, a yeast smell, or something else).
What did you see?
-
What happened in the bottles
having no yeast?
-
What happened in the bottles
having yeast?
-
Which bottles really smelled
like rotten eggs?
-
Which bottles had bacteria activity?
-
How did the bacteria affect
the smell of the water?
-
What color was associated with
the strong smell?
-
What else did you smell?
What do you think?
-
What might affect the ability
of the bacteria to treat the water?
-
Why do people use compost in
constructed wetlands?
-
What are your conclusions? Write
down what you think.
http://www.dep.state.pa.us/dep/deputate/minres/bamr/amd/science_of_amd.htm
Designing your own experiments
To use the Scientific Method
you should design an experiment to test your hypothesis. A hypothesis
is a question that has been reworded into a form that can be tested
by an experiment. Your hypothesis should be based on the background information
you gathered. Make a numbered, step-by-step list of what you will do to
answer your question. This list is called an "experimental procedure."
Your procedure should be detailed enough that someone else could do your
experiment without needing to talk to you about it. This procedure should
include:
-
Any amounts or measures
you will be using
-
Your one variable and how you
will change it
-
Your control
-
How you are going to measure
the change you observe
-
Diagrams or drawings so your
ideas are clear
Your experiments must be repeated to guarantee what you observe is accurate and to obtain an average
result. This process of repeating the same experiment many times is called
"repeated trials." Experiments can be more or less complex, depending on
how they are set up or designed.
Conduct Experiment, Gather
Data, and Record Observations: As you experiment, record all numerical
measurements made. Data can be amounts of chemicals used, how long something
is, the time something took, etc. If you are not making any measurements,
you probably are not doing an experimental science project. Observations
can be written descriptions of what you noticed during an experiment or
problems encountered. You should be looking for differences between your
control group and your experimental group(s).
Two things to be aware of
while doing your experiment and making observations.
-
First, if you did not observe
any differences between the control and experimental groups, then the variable
you changed may not affect your experiment.
-
Second, if you did not observe
a consistent, reproducible trend in your experimental runs, there may be
experimental errors affecting your results or something else that you might
not have thought about.
If you suspect experimental
errors, the first thing to check is how you are making your measurements.
Is the measurement method questionable or unreliable? Maybe you are reading
a scale incorrectly, or maybe the measuring instrument is not working.
If measurements do not seem to be a problem, check to make sure you are
following the rest of your procedure carefully from one run to the next.
If you determine that experimental errors are influencing your results,
carefully rethink the design of your experiments. Review each step of the
procedure to find sources of potential errors. If possible, have a teacher
review the procedure with you. Sometimes the designer of an experiment
can miss something obvious.
Always keep careful notes
of everything you do and everything that happens. Observations are valuable
when drawing conclusions and useful for locating experimental errors.
For more information on science fair projects, designing experiments, and other closely related information, visit the website
http://ipl.lub.lu.se/youth/projectguide/
Send us a letter or
e-mail with your name, address, city, state, and zip code. Describe your experiments, let us know what you think, tell
us what you have discovered about our environment, and become an important
part of the writing of our book on acid mine drainage. The e-mail
and mailing addresses can be found at the beginning of this document.
"Thoughts on Pollution"
from Tim Craddock: The
quality of a stream is the result of what happens along its banks and in
its watershed. A watershed is "all the land surrounding the stream that
drains into the stream." In a watershed that has been transformed into
shopping centers, factories, mines, homes, highways, and farms, many potential
sources of pollution exist. If the sources are not identified and corrected,
the stream will no longer be able to provide habitat for all the living
things that depend on the stream, including fish, wildlife, and even humans.
The goals of these experiments are to help you become more familiar with
one of the pollution problems, acid mine drainage. However, the ultimate
goals are to encourage you to get outside and develop a better understanding
about your environment through observation and experimentation and to
help you become more aware of the many sources of pollution that exist
in our world today, even in our own backyards.
http://www.epa.gov/owow/nps/
Resources for
more Information
-
AIMS Foundation, 1988, Water,
precious water: A collection of elementary water activities for grades
2 through 6: PO Box 8120, Fresno, CA
-
Costen, J., and Hornberger, M.,
1995, Water wizardry: A teachers guide to classroom activities and demonstrations
about water pollution and remediation: U.S. Geological Survey, Menlo Park,
CA (To order contact: Michelle I. Hornberger, 345 Middlefield Rd., MS 465,
Menlo Park, CA 94025.)
-
U.S. EPA, 1990, Acid rain: A students
first source book: U.S. Environmental Protection Agency (RD-682), 401 M Street,
SW. Washington, DC 20460 (Also available on the Internet at: http://www.epa.gov/acidrain/student/student2.html)
-
Gartrell, J.E., and others, 1992,
Earth - The water planet: National Science Teachers Association. (To order contact:
U.S. Geological Survey, MS 950, Reston, VA 20192.)
-
McGee, E., 1995, Acid rain and
our Nations Capitol: A guide to effects on buildings and monuments: U.S.
Geological Survey, Reston, VA (To order contact: U.S. Geological Survey, MS 950,
Reston, VA 20192.) (Also available on the Internet at: https://pubs.usgs.gov/gip/acidrain/)
-
Robbins, E.I., and Hayes, M.,
1997, What's the red in the water? What's the black on the rocks? What's the oil
on the surface?: (Available on the Internet at: https://pubs.usgs.gov/gip/microbes/)
-
Schrock, J.R., 1993, Surface
mining of coal: The Kansas School Naturalist, v. 4, no. 1, Emporia State
University, 1200 Commercial St., Emporia, KS 66801-5087
-
Sly, C., 1990, Water wisdom:
A curriculum for grades 4 - 8: Publication of the Alameda County Office
of Education. (To order contact: U.S. Geological Survey Library, MS 950, Reston,
VA 20192.)
-
Zielinski, E.J., 1995, Acid mine
drainage in Pennsylvania, K-12 Awareness Activities: Pennsylvania Department
of Environmental Resources, Bureau of Land and Water Conservation.
Internet Links
A Community Water Quality Monitoring Manual: http://www.vic.waterwatch.org.au/manual/
Acid Mine Drainage, the unseen enemy: http://www.valdosta.edu/~tmanning/hon399/wally.htm
American Rivers: http://www.amrivers.org/mines.html
Aquatic Macroinvertebrate ID: http://www.net1plus.com/users/tdriskell/macroinvertebrates.html
Bacteria in Groundwater: http://www.ce.vt.edu/enviro2/gwprimer/bacteria/bacteria.html
Biological Time clock Experiments: http://www.cbt.virginia.edu/Olh/
Environmental Education: Internet Resources: http://www.wcupa.edu/library.fhg/recommnd/Environ.htm
Environmental Links: http://www.innovative-solutions.net/links.htm
Explore EE Links on the Internet: http://www.uwsp.edu/acad/wcee/links.htm
Hydrogeology and the Water Cycle: http://www.arch.cuhk.edu.hk/~patrick/slope/background_information/water_cycle.htm
Learn about Wetlands: http://athena.wednet.edu/curric/land/wetland/
Micro-organisms in Acid Rock Drainage: http://www.enviromine.com/ard/Microorganisms/roleof.htm
Mine Net: http://www.microserve.net/~doug/aciddra.html
NACD Links to Internet Resources: http://www.nacdnet.org/resources/links.htm#EviEd
National Watershed Focus: http://www.ctic.purdue.edu/KYW/Focus/Nov96.html
National Wildlife Federation: http://www.nwf.org/nwf/kids/cool/water2.html
Native American uses for Cattails: http://www.nativetech.org/cattail/cattail.htm
Natural Resource Conservation Service: http://www.nrcs.usda.gov/
Passive Treatment Technologies for Treating AMD: http://www.wvu.edu/~agexten/landrec/passtrt/passtrt.htm
Recycling and Solid Waste Management Resources: http://wayne-health.org/wc_recycling_info.html
Restoration of a Stream Degraded by AMD: http://wwwpah2o.er.usgs.gov/projects/amd/restoration.html
Save-Our-Streams, Stream Doctor Program: http://www.iwla.org/SOS/streamdo.html
Science Made Simple: http://www.sciencemadesimple.com/
The Environmental Education Network: http://www.envirolink.org/enviroed/content.html
The Water Page: http://www.thewaterpage.com/water-conservation.htm
The Young Scientist Introduction to Wetlands: http://www.wes.army.mil/el/wetlands/ysi.html
U.S. Environmental Protection Agency: http://www.epa.gov/
U.S. EPA Region 3: Mountain top Mining: http://www.epa.gov/region3/mtntop/
U.S. Fish and Wildlife Service: http://www.fws.gov/
U.S. Geological Survey: Educational Resources: http://water.usgs.gov/education.html
U.S. Geological Survey: Programs in West Virginia: http://water.usgs.gov/wid/html/wv.html
USDA Backyard Conservation: http://www.fb-net.org/Backyard.htm
USDA: Watersheds and Wetlands Division: http://www.ftw.nrcs.usda.gov/programs.html
USGS National Wetlands Research Center: http://www.nwrc.nbs.gov/
USGS: New Techniques to Treat AMD: http://www.usgs.gov/tech-transfer/factsheets/FS-212-96.html
Volunteer Stream Monitoring: http://www.epa.gov/owow/monitoring/volunteer/stream/
Water Quality Modules: http://www.cotf.edu/ete/modules/waterq/wqacidmine.html
Watershed Education Links: http://www.adopt-a-watershed.org/aawlinks.htm
Watershed Education: http://stopnpp.com/educate/educate.htm
West Virginia Geology: http://129.71.2.20/www/geology/geology.htm
West Virginia Wetlands: http://www.geocities.com/Athens/Aegean/8003/wvwet.html
Wetlands and People: http://psybergate.com/wetfix/ShareNet/Sharenet1/Share.htm
WV Division of Environmental Protection: http://www.dep.state.wv.us/
WV Division of Natural Resources: http://www.dnr.state.wv.us/default.htm
WV K-12 Rural Net Project: http://www.wvu.edu/~ruralnet/monitor/monitor.html
WV Nongame and Natural Heritage Program: http://www.dnr.state.wv.us/wvwildlife/nongame/default.htm
WVU: Acid Mine Treatment: http://www.wvu.edu/~research/techbriefs/acidminetechbrief.html
U.S. Department of Interior, U.S. Geological Survey
URL of this page:
https://pubs.usgs.gov/of/2000/of00-369/
Contact: EERSC Team
Last modified: 01.04.12 (jmw)




 If your
creek water is clear, is it clean?
If your
creek water is clear, is it clean?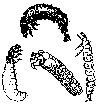


















 Iron
and the oxidation-reduction process
Iron
and the oxidation-reduction process
