 |
 |
A Laboratory Manual for X-Ray Powder Diffraction
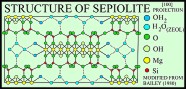
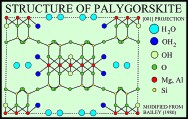
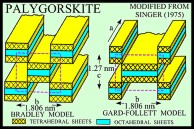
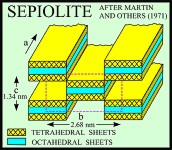 Sepiolite and palygorskite have similar fibrous or lath-like morphologies, but palygorskite exhibits more structural diversity and, although both minerals are Mg silicates, has less Mg and more Al than sepiolite (Caillere and Henin, 1961; Moore and Reynolds, 1997). The structures of both minerals are similar in that tetrahedra pointing in the same direction form 2:1 ribbons that extend in the direction of the a-axis and have an average b-axis width of three linked tetrahedral chains in sepiolite and two linked chains in palygorskite (Singer, 1989). Rectangular channels, which contain some exchangeable Ca and Mg cations and zeolitic water, lie between the ribbons, and molecules of bound water lie at the edges of the ribbons.
Sepiolite and palygorskite have similar fibrous or lath-like morphologies, but palygorskite exhibits more structural diversity and, although both minerals are Mg silicates, has less Mg and more Al than sepiolite (Caillere and Henin, 1961; Moore and Reynolds, 1997). The structures of both minerals are similar in that tetrahedra pointing in the same direction form 2:1 ribbons that extend in the direction of the a-axis and have an average b-axis width of three linked tetrahedral chains in sepiolite and two linked chains in palygorskite (Singer, 1989). Rectangular channels, which contain some exchangeable Ca and Mg cations and zeolitic water, lie between the ribbons, and molecules of bound water lie at the edges of the ribbons.
Both sepiolite and palygorskite require alkaline conditions and high Si and Mg activities for stability (Singer,
 1989). They are also loosely associated with low latitudes and semi-arid climates. Most major deposits were originally formed in shallow seas and lakes as chemical sediments or by the reconstitution of smectites, in open oceans by the hydrothermal alteration of volcanic materials, and in calcareous soils by direct crystallization (Callen, 1984). Owing to their absorptive properties, sepiolite and palygorskite are used commercially as carriers, fillers, clarifying agents, and in used lubricant reclamation.
1989). They are also loosely associated with low latitudes and semi-arid climates. Most major deposits were originally formed in shallow seas and lakes as chemical sediments or by the reconstitution of smectites, in open oceans by the hydrothermal alteration of volcanic materials, and in calcareous soils by direct crystallization (Callen, 1984). Owing to their absorptive properties, sepiolite and palygorskite are used commercially as carriers, fillers, clarifying agents, and in used lubricant reclamation.
 The fibrous nature of sepiolite and palygorskite precludes the production of oriented aggregate mounts to enhance the 001 reflection for X-ray powder diffraction (Wilson, 1987). However, strong reflections from the 011 planes yield intense peaks at 12.2 angstroms in sepiolite and at 10.5 angstroms in palygorskite. These peaks are unaffected by solvation with ethylene glycol, but change during heat treatments. After heating to 400 C, the 001 peaks of both minerals are reduced in intensity and new palygorskite peaks occur at 9.2 and 4.7 angstroms (Singer, 1989). After heating to 550 C, the original 011 palygorskite and sepiolite peaks are completely destroyed, but now new peaks for sepiolite occur at 10.4 and 8.2 angstroms.
The fibrous nature of sepiolite and palygorskite precludes the production of oriented aggregate mounts to enhance the 001 reflection for X-ray powder diffraction (Wilson, 1987). However, strong reflections from the 011 planes yield intense peaks at 12.2 angstroms in sepiolite and at 10.5 angstroms in palygorskite. These peaks are unaffected by solvation with ethylene glycol, but change during heat treatments. After heating to 400 C, the 001 peaks of both minerals are reduced in intensity and new palygorskite peaks occur at 9.2 and 4.7 angstroms (Singer, 1989). After heating to 550 C, the original 011 palygorskite and sepiolite peaks are completely destroyed, but now new peaks for sepiolite occur at 10.4 and 8.2 angstroms.
| X-ray powder diffraction patterns of oriented-aggregate mounts showing the effects of standard treatments: | |