

|
Roger N. Clark1,
Gregg A. Swayze1,
Richard Wise1,
K. Eric Livo2,
Todd M. Hoefen1,
Raymond F. Kokaly1,
and
Steve J. Sutley2
U. S. Geological Survey
1 Crustal Imaging and Characterization Team
2 Minerals Team
U.S. Geological Survey
Box 25046 Federal Center
Denver, CO 80225
U.S. Geological Survey, Open File Report 03-395
Director's Approval 9/17/2003
(Any use of trade names is for descriptive purposes only and does not imply endorsement by the U.S. Geological Survey.)
Introduction
We have assembled a digital reflectance spectral library of spectra that covers wavelengths from the ultraviolet to near-infrared along with sample documentation. The library includes samples of minerals, rocks, soils, physically constructed as well as mathematically computed mixtures, vegetation, microorganisms, and man-made materials. The samples and spectra collected were assembled for the purpose of using spectral features for the remote detection of these and similar materials.
Analysis of spectroscopic data from the laboratory, from aircraft, and from spacecraft requires a knowledge base. The spectral library discussed here forms a knowledge base for the spectroscopy of minerals and related materials of importance to a variety of research programs being conducted at the U. S. Geological Survey. Much of this library grew out of the need for spectra to support imaging spectroscopy studies of the Earth and Planets. Imaging spectrometers, such as the Airborne Visible/Infra-Red Imaging Spectrometer (AVIRIS), or the Cassini Visual and Infrared Mapping Spectrometer (VIMS) on its way to orbit Saturn in 2004, have narrow band widths in many contiguous spectral channels that permit accurate definition of absorption features from a variety of materials. Identification of materials from such data requires a comprehensive spectral library of minerals, vegetation, man-made materials, and other subjects in the scene.
Our research involves the use of the spectral library to identify the components in a spectrum of an unknown. Therefore, the quality of the library must be very good. However, the quality required in a spectral library to successfully perform an investigation depends on the scientific questions to be answered and the type of algorithms to be used. For example, to map a mineral using imaging spectroscopy and the Clark et al. (1990a; 2003b) mapping algorithm, one simply needs a diagnostic absorption band. The mapping system uses continuum removed reference spectral features fitted to features in observed spectra. Spectral features for such algorithms can be obtained from a spectrum of a sample containing large amounts of contaminants, including those that add other spectral features, as long as the shape of diagnostic feature of interest is not modified. If, however, the data are needed for radiative transfer models to derive mineral abundances from reflectance spectra, then completely uncontaminated spectra are required. This library contains spectra that span a range of quality, with purity indicators to flag spectra for (or against) particular uses.
Acquiring spectral measurements and sample characterizations for this library has taken about 12 person-years of effort. Software to manage the library and provide scientific analysis capability is also provided (Clark, 1980, 1993). A personal computer (PC) reader for the library is also available (Livo et al., 1993). The program reads specpr (Clark 1980; 1993) binary files and plots spectra.
In our view, an ideal spectral library consists of samples covering a very wide range of materials, has large wavelength range with very high precision, and has enough sample analysis and documentation to establish the quality of the spectra. Budgets, time, and available equipment limit what can be achieved.
Ideally, for minerals, the sample analysis would include X-ray diffraction (XRD), electron microprobe (EM) or X-ray Fluorescence (XRF), and petrographic microscopic analyses. For some minerals, like iron oxides, additional analyses, such as Mossbauer, would be helpful. We have found that to make the basic spectral measurements, provide XRD, EM or XRF, microscopic analysis, document the results, and complete an entry of one spectral library sample, takes about one person-week. Additional spectra of the same sample (e.g. a grain size series) increases the time, but usually not an additional week per spectrum, but more like 0.5 day per spectrum (mostly sample preparation). We had hoped as our experience increased this time would decrease, but it has not in our experience of over 15 years of developing this and previous spectral databases.
Our budgets and time commitment have not allowed the level of effort described above, so this release of the library does not have all samples completely characterized. The characterization of samples will continue as our budgets allow, and results will be added in future releases of the database.
This report describes spectral library 05: splib05a. Libraries 01, 02, and 03 were unpublished experimental exercises that investigated what to include in a spectral library. Library 04, splib04a, Clark et al., 1993, has been our main working library until this release. The splib05a library include all spectra from the splib04a library with additional documentation derived since that study.
Types of Materials
Minerals from borate, carbonate, chloride, element, halide, hydroxide, nitrate, oxide, phosphate, sulfate, sulfide, sulfosalt, and the silicate (cyclosilicate, inosilicate, nesosilicate, phyllosilicate, sorosilicate, and tectosilicate) classes are represented. X-Ray and chemical analyses are tabulated for many of the entries, and all samples have been evaluated for spectral purity. The library also contains end and intermediate members for the olivine, garnet, scapolite, montmorillonite, muscovite, jarosite, and alunite solid-solution series. We have included representative spectra of H2O ice, kerogen, ammonium-bearing minerals, rare-earth oxides, desert varnish coatings, kaolinite crystallinity series, kaolinite-smectite series, zeolite series, and an extensive evaporite series. Because of the importance of vegetation to terrestrial studies we have include spectra of trees, shrubs, grasses, flowers, leaves, lichens and other microorganisms. The vegetation types are representative of the types we have encountered and studied in our published works. Terrestrial remote sensing also encounters man-made materials, so we have included spectra of plastics, roofing materials, processed wood, paint and other man-made materials. Spectra of liquids from water to organic liquids mixed with minerals are also included. Together the spectral library encompasses a broad range of materials that may exist in a remotely sensed image.
In some cases, several spectra were measured to span a solid solution series and/or a grain size series. We tried to include spectra of all mineral classes, particularly those important to imaging spectroscopy remote sensing. In other cases, we have studied particular solid solution series because we are mapping them in the field with imaging spectroscopy or studying that mineral in detail. This explains, for example, why there are so many alunite, olivine, and topaz samples in the database. Future releases of the database will likely include additional spectra of other solid solution series.
Spectral Library Chapter Organization
The digital spectral library is organized in chapters:
The organization of some samples into a particular chapter was sometimes in a "gray area." For example, pure minerals are often difficult to find or it may be difficult to process the sample to purify the mineral. Such a sample could be considered a mixture. We placed some samples like this in the Minerals Chapter because we are using the sample and its spectra to match to pure minerals. We tended to place in the mixture chapter (Chapter 2: S) samples which show specific mixture spectral properties that we use for identification and mapping of that mixture. Usually these will include overlapping absorption features.
Spectrometers
The spectra in our library were measured on at least one of 4 different
spectrometers: 1) a Beckman 5270 covering the spectral range 0.2 to 3
microns, an Analytical Spectral Devices (ASD) portable field
spectrometer covering the range from 0.35 to 2.5 microns, 3) Nicolet
Fourier Transform Infra-Red (FTIR) Interferometer Spectrometer covering
the range from about 1.3 to 5.2 microns, and 4) the NASA Airborne
Visible-Infra-Red Imaging Spectrometer (AVIRIS) covering the range 0.4
to 2.5 microns. The spectral resolution of these instruments is shown
in Figure 1.
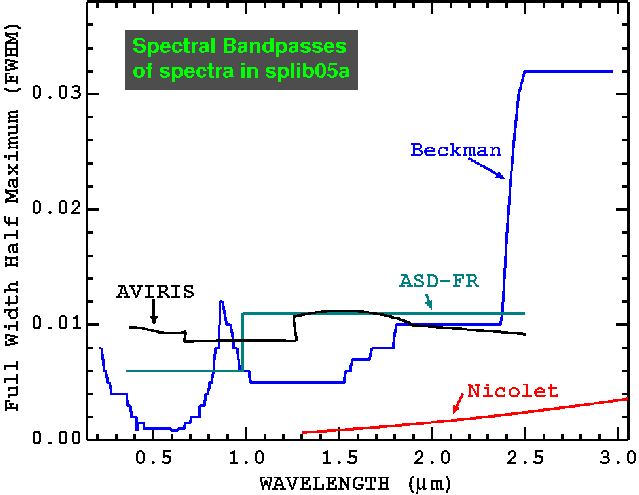
Figure 1. The spectral bandpass for the spectrometers used to obtain spectra for digital spectral library splib05a. The bandpass is expressed as the Full Width at Half Maximum (FWHM).
Spectra run on our modified Beckman 5270 spectrometer (Clark et al., 1990b) from 0.2 to 3.0 microns were corrected to absolute reflectance. This instrument measures directional-conical light as described in Clark et al., 1990b. Samples were run with a signal-to-noise ratio of at least 500 at a reference reflectance level of 1.0. A few minerals were also run over a slightly smaller wavelength range because of sample size limitations. For example, small sample quantities, necessary for purity, were measured using apertures in the beam to restrict the spot size of the spectrometer. This reduced light made integration times longer and the achievable range was sometimes reduced, typically to 0.3 to 2.7 µm. This also, in some cases, limited the signal-to-noise ratio that was achievable. It is not possible with the current instrumentation to substantially improve the spectral data on small volume samples. The ice sample, measured at 77o K, only includes the infrared range, 0.8 to 3.0 µm.
Spectra measured on the ASD spectrometer used a directional light source and fiber-optic probe to collect light. The incidence angle was variable but typically ranges from 20 to 40 degrees, as did the emission angle. Spectra are corrected to absolute reflectance using a Spectralon standard with correction methods similar to that described in Clark et al., (1990b).
Spectra were also run on a commercial Nicolet Fourier Transform Infra-Red (FTIR) interferometer spectrometer. Most spectra were run on a Nicolet model 740 until 1998 when spectra were run on a Nicolet Magna. Spectra were measured over a range from about 1.3 to 150 microns, but we report here data only to 5.2 microns. Longer wavelength data will be presented in a future library. To cover the factor of 100 in wavelength range from 1.5 to 150 microns, two detectors and two beam splitters had to be used. For all samples, the near IR (1.3 to 5 microns) (referred to here as the NIR) and the mid-IR (~2.5 to 25 microns) (MIR) were measured with the same beam splitter and with no change in the sample. On the Nicolet 740, only a change in light source was required: a simple change of a lever. On the Nicolet Magna, an internal mirror automatically switched the light source and detector. With some setup changes in an instrument or between instruments, a wavelength region was measured at a different time, thus each sample was put back in its bottle between measurements. There was sufficient overlap in the data to confirm consistency across the spectral boundary.
The AVIRIS spectra in this library are of samples where it would be difficult to make measurements, like tall trees, with normal field portable spectrometers (requiring cranes or other means to lift the spectrometer and operator above the sample). Calibration of AVIRIS data is discussed in Clark et al., (2003a).
The name for each sample occurs in 3 places: 1) the specpr (Clark, 1993) title field for the spectrum, 2) the specpr title field for the description entry, and 3) after the keyword in the description. We have tried to use only proper mineral names as given in Fleischer (1980), Fleisher and Mandarino (1995), and Klein and Hurlbut, (1999). Some users of the library may be unfamiliar with all the mineral names. For example, if you want to find all scapolites, you would have to know that Dipyre was a scapolite if you only looked at the specpr title fields. Because of the 40 character limit in a specpr title field, we could not include all common names there. However, we used the keywords (described below) in the description for each sample. Here you could search for scapolite and you would find all entries in the "scapolite group" (Dipyre, Marialite, Meionite, and Mizzonite).
We have used specific mineral names except in a few cases where we still do not have sufficient data. For example, technically, there is no "hornblende," only ferro-hornblende and magnesio-hornblende. We have two samples where we cannot make the distinction, so they are labeled hornblende.
The specpr title field is limited to 40 characters. Some sample names, especially mixtures are too long to fit in this space. Thus names are abbreviated. The abbreviations used in this library are listed here: click here.
Sample Documentation
Each spectrum has a sample description page describing the origin and sample purity from available data. The sample description pages follow those from Clark et al., (1993) with keyword=value or keyword followed by text and end_keyword. However, the description pages are now in html format. The html format still contains the keyword organization. Different sample types have different documentation formats. For example minerals require some different documentation from plants.
Templates for the documentation types are:
It is beyond our budget and manpower assigned to the work to complete every entry in the description pages for every sample. Thus, the description pages show what we have completed on these samples to date.
This spectral library includes an updated set of sample description pages that also apply to the Clark et al. (1993) library. Many new X-Ray diffraction (XRD) analyses have been completed, so most mineral samples now have XRD data.
Spectral Purity and Spectral Range Flags
Each spectrum has spectral purity information, spectral range, which spectrometer was used to make a measurement and these are coded in the Specpr title (Table 1). In the Clark et al., (1993) spectral library, the code was: W1R1Bx. The "W" stands for wavelength region followed by the region measured (Table 2). Region 1 is a nominal range of 0.2 to 3.0 microns of the Clark et al., (1993) spectral library. The "R" stands for resolution, followed by the resolution index. The "x" is a letter code for the spectral purity in indicated spectral range. The wavelength, resolution and spectrometer codes in this library are as follows. The "W1" refers to wavelength region 1, as before. Some Nicolet spectrometer data are included in this library that extends to 5.2 microns
The spectral ranges are (the number after the W):
| Spectral Ranges | ||
|---|---|---|
| Spectral Range Number | Range | |
| 1 | 0.2 - 3 microns | (50000 - 3333 cm-1) |
| 2 | 1.5 - 6 microns | (6666 - 1666 cm-1) |
| 3 | 6 - 25 microns | (1666 - 400 cm-1) |
| 4 | 20 - 150 microns | (500 - 66.7 cm-1) |
For this release, splib05a, only spectral region 1 is evaluated. Nicolet data in this library also cover wavelength region 2. It is our intent to include Nicolet data to 150 microns in a future release of the library.
Following each spectral range number is a lower case letter (the "x" in Table 1) signifying the spectral purity of the spectrum for that wavelength range.
a: The spectrum and sample are pure based on significant supporting data available to the authors. The sample purity from other methods (e.g. XRD, microscopic examination) indicates essentially no other contaminants. Spectrally, no contaminants are seen in the spectrum.
b: The spectrum appears spectrally pure. However, other sample analyses indicate the presence of other minerals that probably affect the absolute reflectance level to a small degree, but do not add any significant spectral features in that region. The spectral features of the primary minerals may be slightly less intense, but the feature positions and shapes should be representative. For example, in the visible-NIR wavelength regions (W1 or 2), quartz would tend to increase the reflectance level and decrease absorption band strength, but would not add any measurable features to the spectrum. Such a sample would rate a "b." In a few cases, where we have little support data, but the spectra for that mineral are well known, we assigned the spectral purity based on the spectral data, possibly along with information derived from a microscopic examination of the sample. There are a few "b" ratings assigned this way.
c: The spectrum is spectrally pure except for some weak features with depths of a few percent or less caused by other contaminants. For example, some minerals may have some slight alteration that is apparent. Spectroscopic detection of alteration minerals is easier for more transparent minerals. For example, some of the albite spectra show weak 2.2-micron features due to alteration. From the knowledge of the mineral formula, one can often tell which features do not belong to the mineral. Albite, for instance, does not have OH in the formula, so water or hydroxyl features (1.4, 1.9, 2.2 microns in the spectrum) are not due to albite. However, you could argue that incipient alteration due to weathering is common in minerals at the Earth's surface. Thus, spectral bands due to weathering are somewhat characteristic of many samples (e.g. feldspars), even if they are not a property of the pure mineral. Spectral features of alteration minerals might be useful in some cases, but such alteration and their spectral effects still reduce spectral purity as far as this library is concerned. Note, also, that almost every sample has some absorption near 3 micron due to water in the sample. Water adsorbs onto mineral grains and is common. Water absorptions at 3 microns are not considered contaminants and do not affect spectral purity.
d. d: Significant spectral contamination. The spectrum is included in the library only because it is the best sample of its type currently available and the primary spectral features can still be recognized (it may also have a better spectral purity in another wavelength range). However, the spectrum should be used with care. The sample description should be consulted as a guide to which features are a part of the actual mineral. This sample may be purged from the database in future releases as better samples become available.
?: There are insufficient analyses and/or knowledge of the spectral properties of this material to evaluate its spectral purity. In general we have included such samples because we believe their spectra to be representative. These are samples for which we are concentrating future analyses in order to resolve the purity issue. Note that there may be sufficient knowledge to assign spectral purity in one spectral region, but not another.
Mixtures, soils, rocks, coatings can be complex. The spectral purity of complex mixtures can still be high (assigned "a" status; see the Spectral Purity discussion below), if the goal of the mixture is to document the spectral features in such a mixture. Mixtures that have additional contaminants not intended for documenting that mixture would have a lower spectral purity than if the contaminant were not present, following the guidelines above.
The spectral purity should be consulted before use of spectra to see if the sample is appropriate for the intended purpose. For example, examination of the spectra for hypersthene PYX02 will show that the spectral purity is a "c," meaning contaminating spectral features overlap diagnostic hypersthene spectral features. However, as documented in the sample description, the pyroxene sample contains a small amount of tremolite alteration. While the tremolite absorptions are narrow absorptions at 1.4 and 2.3 microns, the pyroxene absorptions are very broad. If an analysis can ignore the narrow contaminant absorptions, then the sample can be used as an excellent reference spectrum for hypersthene.
Another example of spectral purity: consider orthoclase, sample NMNH142137. The spectral purity is a "b." There are weak alteration mineral absorptions in the 2 to 2.5 micron spectral region and are not indicative of feldspars. Most feldspars for which we have seen or measured spectra show alteration to muscovite at 1.4 and 2 to 2.5 microns. These absorptions are not due to feldspars and should not be used to identify feldspars.
The spectrum for each mineral should be evaluated for contaminant features and how those features might interfere with a particular analysis. Knowledge of the spectral properties of minerals and other materials is important in using any spectral database as reference spectra for identifying other minerals and materials. See Clark (1999) and references therein for discussions of spectral properties of materials and principles of spectroscopy.
Spectral Measurement Side-Effects
1) Residual Water Absorptions in the Samples. Near 3 microns even anhydrous minerals show absorption due to water adsorbed onto the surfaces of the mineral grains. Our experience has shown that these water absorptions are still present in dry nitrogen purged environments, although slightly weaker, thus the spectra measured on the Nicolet in a dry atmosphere will have different absorption than those same samples measured by other spectrometers like the Beckman (Clark et al., 1993) library in a normal room atmosphere. Spectra of similar samples obtained at other facilities, like those in Hawaii or the east coast of the US have shown that the water absorptions in the spectra from relatively dry Colorado are really quite small in comparison. Placing the sample in a dry nitrogen atmosphere or a vacuum oven has little effect on the water absorption as water from the atmosphere will readsorb onto the sample by the time it reaches the spectrometer. Experiments by the senior author when he was at the University of Hawaii have also shown that most of the adsorbed water remains even under a strong vacuum at room temperatures (Clark, 1981). We decided in general not to heat our samples in order to avoid any temperature induced alteration.
2) Low Signal Artifacts. The Nicolet FTIR near-infrared measurements have an artifact toward shorter wavelengths. From about 2 microns shorter to 1.3-microns, the reflectance curves down giving a convex shape to the spectrum. This roll-over is not correct. Attempts to derive a correction have not been helpful because the amount of roll-over is variable from sample to sample. We include this spectral range because the very high spectral resolution can be quite useful for studying narrow spectral features. The roll-over becomes more intense at wavelengths shorter than about 1.5 microns. We sometimes include data to 1.3 microns if it shows narrow spectral features, but the broader curvature should not be interpreted as spectral structure.
3) Purged Samples and Residual Atmospheric Features.
All Nicolet samples in this library were measured in a dry nitrogen
atmosphere, scrubbed of H2O and CO2 by a Balston
air purification system. The sample and the reflectance standard were each placed in
the sample chamber, and a period of time was allowed to pass before a
measurement was made. The standard and reference were each measured at
approximately the same time after installation into the chamber, so the
purge and any residual atmospheric gases which could cause absorptions
should be roughly comparable and therefore should cancel. This did not
always work perfectly, and residual atmospheric absorptions
(H2O and CO2) are sometimes present at a low level
(e.g. see Salisbury et al., 1991). Such residual absorptions
(Figure 2a, b) are difficult to remove completely because of the intense nature of the
absorptions. Similar features are reported in other databases covering
this spectral region (e.g. Salisbury et al., 1991).
Figure 2a. Residual atmospheric H2O absorptions.
Figure 2b. Residual atmospheric CO2 absorptions.
4) Spectral Measurement Geometry. Measurements with our Nicolet
spectrometer geometry results in an unusual side effect. The basic
geometry of the Nicolet spectrometer bi-conical reflectance attachment
is shown in Figure 3a, 3b. The reflectance attachment focuses the
spectrometer beam onto a spot about 2 mm in diameter (Figure 3b) via an
elliptical mirror. A second mirror collects the reflected light and
sends it to a detector. The small spot size produces side effects at
wavelengths where the sample is weakly absorbing and multiple scattering
of reflected photons is a significant portion of the signal. Some
photons will scatter from grain to grain, leaving the surface outside
the focus of the collection mirror, and therefore are not seen by the
detector (Figure 3b). This results in a relative loss in signal, and a
measured lower reflectance at wavelengths where multiple scattering is
significant. This is illustrated in Figure 4, where a Nicolet
measurement is compared to a spectrum from the Clark et al.,
(1993) library. The spectra are scaled to match at 2.25 microns
in this case. The
Nicolet measurements show weaker 2.16, 1.75, and
2.3-micron absorptions due to this effect. The Nicolet measurement is
lower in reflectance at wavelengths smaller than 2-microns also due to
this effect. The increased spectral structure in the Nicolet spectrum
near 2.5 microns is due to the much better spectral resolution.
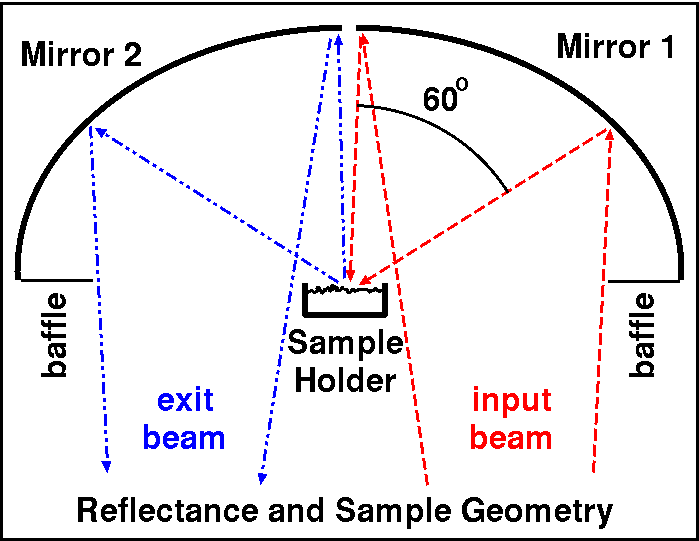
Figure 3a. The sample viewing geometry for the Nicolet spectrometer
was biconical. The measurement had maximum
60-degree entrance and exit beams (30-degree average), with about a 60 phase
angle.
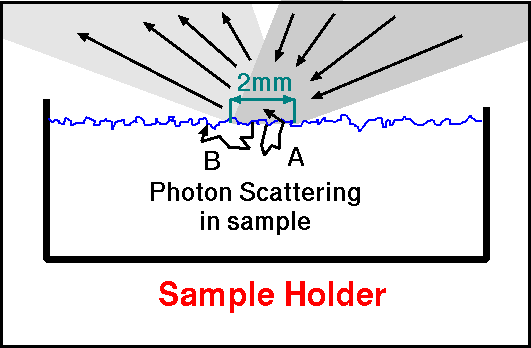
Figure 3b. The small spot measured by the Nicolet spectrometer
results in some loss of photons (labeled B) when the photons are
multiply scattered. Only photons scattered with the exit field of view
(labeled A) are measured. This edge effect is insignificant in the
mid-infrared where absorption coefficients are higher than in
transparent materials at wavelengths less than about 6 microns.
Figure 4. The sample viewing geometry and small spot size of the
Nicolet spectrometer reflectance attachment (Figure 3) results in some
loss of photons. Here, a Nicolet measurement is compared to a spectrum
from the Clark et al., (1993) library. The spectra are scaled to
match at 2.25 microns. The Nicolet measurements show weaker 2.16, 1.75,
and 2.3-micron absorptions due to the loss effect. The Nicolet
measurement is also lower in reflectance at wavelengths smaller than 2
microns also due to this effect. The increased spectral structure in
the Nicolet spectrum near 2.5 microns is due to the much better spectral
resolution compared to that from the Beckman spectrometer.
The narrow focus range of the measurement geometry results in large changes in reflectance level with small changes in the position of the surface of the sample. Because of this uncertainty, data from the Clark et al., (1993) visible-near-infrared library and/or newer spectra were used as a guide to scale the Nicolet to the correct reflectance levels. Because of the light loss effect described above, the reflectance level of samples high in reflectance (greater than about 0.4) in the 1.5-2.5 microns spectral range is accurate only to about 10%. Darker samples should have more accurate levels.
Organic materials at low concentrations can leave residual spectral features (Figure 5).
This usually only affects the region near 3.4 microns.
Figure 5. Illustration of an organic contaminant in Nicolet spectrometer
data.
Wavelength Precision
The wavelength precision of our custom-modified, computer-controlled Beckman spectrometer was checked using Holmium Oxide filters in the visible and the positions of known mineral bands in the near infrared. In particular, we developed pyrophyllite as a wavelength standard because of its many narrow absorption bands (Clark et al., 1990b). The positions of the absorption bands have been checked, using the same pyrophyllite standard, on three FTIR spectrometers. In general, the wavelength accuracy is on the order of 0.0005 microns (0.5 nm) in the near-IR and 0.0002 microns (0.2 nm) in the visible, always a small fraction of the spectral resolution. Wavelength precision for this instrument is further discussed in Clark et al. 1990a.
The wavelength precision of the ASD portable field spectrometer was monitored using a mylar plastic sheet with strong absorption features. The instrument was also calibrated annually by the manufacturer. The position of the mylar spectral features was precisely measured with our Nicolet FTIR instrument, described next.
The wavelength precision of a Fourier Transform Spectrometer is accurate to better than the bandwidth of the system. The Nicolet FTIR systems use a red laser wavelength as a fringe counter in the interferometer, a fraction of the shortest wavelength measured in this library. We also confirmed wavelength positions with our pyrophyllite standard (Clark et al., 1990b, 1993) and its many narrow absorption bands for which we measured their positions on 3 different FTIR spectrometers and our Beckman spectrometer and found excellent consistency.
The wavelength calibration of the NASA AVIRIS system was performed by NASA/JPL (Green et al., 1990, 1996).
Spectral Plots and Data Precision
Plots of the spectra presented here are limited to one of several vertical scales (e.g. 0.0 to 1.1, 1.0, 0.8, 0.7, 0.6, 0.5, 0.4, 0.25, 0.15, and 0.1) and the same horizontal range for easy comparison. The error bars are plotted only when they are above a threshold that allows them to be distinguished on the plot. Most error bars are too small to be distinguished. Each plot shows the specpr title, date and time of acquisition, file name, record number and wavelength set. Not all spectra include error estimates.
Each spectrum was run with a desired signal-to-noise ratio of at least 500 relative to unity reflectance. In practice, it would take too long a time to obtain such a signal-to-noise ratio in regions where the signal is low, so an upper limit to the integration time per channel was also specified with our Beckman spectrometer. Thus, typically at the ends of the spectra (both short and long wavelength ends), the precision drops slightly. Refer to the error bars for each spectrum or examine the channel to channel noise to determine the precision at a given wavelength for any individual spectral channel. The ASD and Nicolet spectrometers typically have higher signal to noise ratios but do not report that data. Thus, signal to noise must be estimated by the spectral channel to spectral channel variability.
Mineral Mixtures and Optical Constants
This spectral library contains pure as well as mixture materials. For computing intimate mineral mixtures (e.g. rocks or soils), radiative transfer algorithms using the Hapke reflectance model (Hapke, 1981) are part of the specpr package. To compute mixture or pure end-member spectra, a set of optical constants are required as a function of wavelength. The algorithms use optical constants so spectra can be calculated as a function of grain size, abundance in a mixture, and viewing geometry. Reflectance spectra of grain-size distributions can also be simulated by computing a mixture of the same mineral (or even several minerals) at several grain sizes.
Pure samples are extremely rare. Spectroscopy, especially over the 0.2 to 150 microns spectral range--a factor of 750 in wavelength range) is very sensitive to low level contaminants at some wavelengths. Spectroscopy can detect a trace contaminant even in samples ground from single crystals that appeared pure. Thus, most samples in this library could be considered a mixture at some level. In other cases, deliberate mixtures are included because of the common associations in which they occur. If you intend to use a spectrum for a particular purpose, confirm the spectral purity to be sure it is appropriate for your needs.
The digital spectral library data are all included in one file in "specpr" format (see Clark, 1993). This file, splib05a, is 19 megabytes in size and has been assembled and managed using the spectral processing software package, specpr. The data are in IEEE binary floating point format. The entire library is assembled, plotted and printed by command files consisting of Unix shell commands, which in turn generate specpr commands to build the library.
Specpr runs on Unix workstations. If the binary file is read on an IBM-PC compatible machine, the floating point numbers need their bytes swapped (this is done in the Livo et al., 1993 program). Ascii versions of all spectra along with plots are presented in the Date Table at the end of this web document.
The organization of the binary data file, in the form of a specpr listing, is shown in Table 3. The listing shows the record number, title, length of the data set (number of channels for spectra; number of bytes for text), and the time and date of data acquisition. Records containing the wavelength sets for the spectra, and records containing the the spectral resolution data sets occur one time near the beginning of the file. The resolution for each spectrometer is shown in Figure 1. Entries with the keyword "DESCRIPT" are sample description records, and contain all the sample documentation. After the DESCRIPT are (usually) two empty records (title ..) for future expansion of the description. Next comes the reflectance data, with the keyword AREF, RREF, or RTGC. The identifier such as "W1R1Bx," which signifies the wavelength range, resolution, spectrometer, and spectral purity which is described below (the "x" is a lower case spectral purity letter code). After the reflectance record is the "errors to previous data" record. These are the standard deviation of the mean for each reflectance value if those values exist, others a placeholder of zeros. The next record in the listing contains the feature analysis for the spectrum. This feature analysis was done using the specpr f45 special function and is described in Clark et al. (1987), Clark (1993).
The Reflectance type, AREF, RREF, or RTGC is defined as follows. AREF is absolute reflectance using standards as described in Clark et al. (1990b). RREF is relative reflectance. All Nicolet spectra used a stainless steel mirror as the standard and are therefore relative to the reflectance of stainless steel. RTGC stands for "Radiative Transfer Ground Calibrated," the method used for all AVIRIS spectra and defined in Clark et al. (2003a).
Table 3
1 USGS Digital Spectral Library: splib05a 379 Characters of TEXT
2 **************************************** 41 Characters of TEXT
3 **************************************** 41 Characters of TEXT
4 **************************************** 41 Characters of TEXT
5 .. 41 Characters of TEXT
6 Wavelengths USGS Denver Beckman STD 1x 480 02:57:26.00 10/15/1985
8 Bandpass (FWHM) USGS Denver Beckman STD 480 02:57:26.00 10/15/1985
10 Wavelengths Standard ASD FR 0.35-2.5um 2151 21:54:44.00 02/05/2002
16 Bandpass (FWHM) ASD FR 0.35-2.5um 2151 00:00:00.00 01/28/2003
22 Wavelengths to Nicolet 1.3 - 5.2 microns 3325 00:00:00.00 02/07/1994
32 Bandpass (FWHM) Nicolet FTIR 1.3-5.3 um 3325 12:30:00.00 03/24/1988
42 Wavelengths AVIRIS 1996 0.4-2.5 microns 224 00:00:00.00 01/01/1996
43 Bandpass (FWHM) AVIRIS 1996 0.4-2.5 um 224 00:00:00.00 01/01/1996
44 **************************************** 41 Characters of TEXT
45 **************************************** 41 Characters of TEXT
46 ---------------------------------------- 41 Characters of TEXT
47 Acmite NMNH133746 Pyroxene DESCRIPT 5519 Characters of TEXT
51 .. 41 Characters of TEXT
52 .. 41 Characters of TEXT
53 Acmite NMNH133746 Pyroxene W1R1Ba AREF 480 15:18:47.00 03/23/1988
55 errors to previous data 480 15:18:47.00 03/23/1988
57 Acmite NMNH133746 Pyroxene FEATANL 315 15:18:47.00 03/23/1988
59 ---------------------------------------- 41 Characters of TEXT
60 Actinolite HS116.3B DESCRIPT 5923 Characters of TEXT
64 .. 41 Characters of TEXT
65 .. 41 Characters of TEXT
66 Actinolite HS116.3B W1R1Bb AREF 480 08:41:01.00 07/11/1991
68 errors to previous data 480 08:41:01.00 07/11/1991
70 Actinolite HS116.3B FEATANL 396 08:41:01.00 07/11/1991
In the spectral library, any value of -1.23x1034 is considered a deleted point. Because of the inherent floating point inaccuracies of single precision numbers on various computers, values in the range -1.23001x1034 to -1.22999x1034 should be considered deleted points.
The spectral library which containing the spectra, standard deviation error values, sample description pages, and a spectral feature analysis of each spectrum is compiled in a specpr-format binary file. Binary files of the original and instrument convolved spectral libraries will be maintained for ftp file transfer from http://speclab.cr.usgs.gov/spectral-lib.html.
Instrument Spectral Libraries
The intent of the spectral library is to serve as a knowledge base for spectral analysis. Comparison of spectral data is best done when the spectral resolutions of the knowledge base and the spectra undergoing analysis are identical. The specpr software (Clark, 1993) has tools for convolving the spectral library to the resolution and sampling interval of any instrument. The native (laboratory) spectral library will also be convolved to AVIRIS spectral resolution and sampling for the terrestrial instruments, and to Galileo NIMS, MGS-TES, and Cassini VIMS for the planetary imaging spectrometer instruments. Other instrument specific spectral libraries will be convolved as the instruments become operational and when wavelength and bandpass information becomes available. These convolved data files, as well as specpr command files for convolving the database to your own instruments will be made available on the spectroscopy lab web site (http://speclab.cr.usgs.gov/spectral-lib.html) and in the anonymous ftp directory. Each convolved instrument library will be provided in both binary specpr and ascii formats.
ASCII Spectral Data
ASCII data for each spectrum are accessible via the web based Data Table (below) The ascii data consist of 3 columns: wavelength, reflectance, and standard deviation of the mean.
Other resources on spectroscopy can be found at http://speclab.cr.usgs.gov.
Acknowledgements
A successful spectral library has extensive sample documentation. We are indebted to J. S. Huebner, and Judy Konnert of the USGS for their support in analyzing the X-ray diffraction data on minerals for the last couple of years in the splib04 (Clark et al., 1993) library, from which part of this library was derived. We thank the late Norma Vergo for many of the earlier X-ray analyses. Norma's attention to detail has certainly made this spectral library a quality product, and we miss her. Without these dedicated people providing superb analysis and feedback, this library would not have been possible.
Of course, a spectral library needs quality samples. We are indebted to Charlie Alpers, Jim Crowley, Skip Cunningham, Jim Post, Fred Kruse, Jack Salisbury, and Roger Strofrogen for donating excellent samples. Thanks to the British Museum and the National Museum of Natural History for mineral samples. We thank Trude King, Brad Dalton, and Barnaby Rockwell for contributing some samples and spectra. We thank Robert McDougal for help in reducing spectral data on the artificial samples and early examinations of lists of spectra to be included.
This project has been funded by the USGS Minerals Program, the NASA Mars Global Surveyor Thermal Emission Spectrometer Team (R. Clark, Co-I), and the NASA Cassini VIMS Team (R. Clark, Team Member).
References
Clark, R.N., 1980. A Large Scale Interactive One Dimensional Array Processing System, Pub. Astron. Soc. Pac., 92, 221-224. (Software online at http://speclab.cr.usgs.gov).
Clark, R.N., 1981. The Spectral Reflectance of Water-Mineral Mixtures at Low Temperatures, J. Geophys. Res., 86, 3074-3086.
Clark, R.N., T.V.V. King, and N.S. Gorelick, 1987. Automatic Continuum Analysis of Reflectance Spectra: Proceedings of the Third Airborne Imaging Spectrometer Data Analysis Workshop, JPL Publication 87-30, 138-14.
Clark, R.N., A.J. Gallagher, and G.A. Swayze, 1990a. Material Absorption Band Depth Mapping of Imaging Spectrometer Data Using a Complete Band Shape Least-Squares Fit with Library Reference Spectra, Proceedings of the Second Airborne Visible/Infrared Imaging Spectrometer (AVIRIS) Workshop. JPL Publication 90-54, 176-186.
Clark, R.N., T.V.V. King, M. Klejwa, G. Swayze, and N. Vergo, 1990b. High Spectral Resolution Reflectance Spectroscopy of Minerals: J. Geophys Res. 95, 12653-12680.
Clark, R.N., G.A. Swayze, A. Gallagher, T.V.V. King, and W.M. Calvin, 1993. The U. S. Geological Survey, Digital Spectral Library: Version 1: 0.2 to 3.0 microns, U.S. Geological Survey, Open File Report 93-592, http://speclab.cr.usgs.gov, 1340 pages, 1993.
Clark, R.N., 1993. SPECtrum Processing Routines User's Manual Version 3 (program SPECPR), U.S. Geological Survey, Open File Report 93-595, 210 pages. (Software online at http://speclab.cr.usgs.gov).
Clark, R.N., G. A. Swayze, K. E.. Livo, R. F. Kokaly, T. V.V. King, J. B. Dalton, J. S. Vance, B. W. Rockwell, T. Hoefen, and R. R. McDougal, Surface Reflectance Calibration of Terrestrial Imaging Spectroscopy Data: a Tutorial Using AVIRIS, AVIRIS Workshop Proceedings, 2003a. Online at: http://speclab.cr.usgs.gov/PAPERS.calibration.tutorial
Clark, R.N., G. A. Swayze, K. E. Livo, R. F. Kokaly, S. J. Sutley, J. B. Dalton, R. R.McDougal, and C. A. Gent., 2003b. Imaging Spectroscopy: Earth and Planetary Remote Sensing with the USGS Tetracorder and Expert Systems, Journal of Geophysical Research, In Press. http://speclab.cr.usgs.gov/PAPERS/tetracorder
Fleischer, M., 1980. Glossary of Mineral Species, Mineralogical Record, Tucson, 192pp.
Fleisher, M., and Mandarino, J.A., 1995, Glossary of Mineral Species 1995: The Mineralogical Record Inc., Tucson, 280 p.
Green, Robert O., James E. Conel, Veronique Carrere, Carol J. Bruegge, Jack S. Margolis, Michael Rast, and Gordon Hoover, 1990, Determination of the In-Flight Spectral and Radiometric Characteristics of the Airborne Visible/Infrared Imaging Spectrometer (AVIRIS), in Proceedings of the Second Airborne Visible/Infrared Imaging Spectrometer (AVIRIS) Workshop, JPL Publication 90-54, pp. 15-22.
Green, R. O., D. A. Roberts, J. A. Conel, 1996, Summaries of the Sixth Annual JPL Airborne Earth Science Workshop, JPL Publication 96-4, 135.
Hapke, B., 1981. Bidirectional reflectance spectroscopy 1. Theory, J. Geophys. Res. 86, 3039-3054.
Klein, C. and Hurlbut, Jr., C.S., 1999, Manual of Mineralogy, 21st edition, John Wiley and Sons, 681pp.
Livo, E., R.N. Clark, and D. Knepper, 1993, SPVIEW Spectral Plot Program for accessing the U.S. Geological Survey Digital Spectral Library Database with MS-DOS Personal Computers: U.S. Geological Survey, Open File Report 93-593, 26 pages. (Software online at http://speclab.cr.usgs.gov).
Salisbury, J.W., L.S. Walter, N. Vergo, and D.M. D'Aria, 1991, Infrared (2.1-25 microns) spectra of minerals, The Johns Hopkins University Press, Baltimore, 267pp.
| AccessibilityFOIAPrivacyPolicies and Notices | |
| |
|