U.S. GEOLOGICAL SURVEY
Scientific Investigations Report 2004-5090
Vertical Distribution of Trace-Element Concentrations and Occurrence of Metallurgical Slag Particles in Accumulated Bed Sediments of Lake Roosevelt, Washington, September 2002
Trace Elements in Pore Water and Sediment
The concentrations of trace elements in sediment and pore-water matrixes were determined for samples collected at several depth intervals (surface to 1 cm, 1 to 2 cm, and 9 to 11 cm) in shallow cores collected at sites CCR-668, CCR-692, and CCR-705 (table 7, at back of report ). Concentrations of mercury were not determined in pore water or sediment samples because the pore-water sample volume was insufficient for mercury analysis. Some uncertainty is associated with the dissolved concentration of trace elements in pore water because the concentrations of iron and zinc were quite different in replicate samples of pore water obtained from the 9 to 11 cm interval from site CCR-668. Concentrations of arsenic, cadmium, copper, lead, and zinc of the sediment matrix (table 7) also was determined for the sediments collected for pore-water analysis. Trace-element concentrations determined for pore-water sediments typically were within about 15 percent of trace-element concentrations reported for the sediments from the same or nearby horizon in the cores that were collected to determine concentration profiles within the accumulated sediments at each site (table 5).
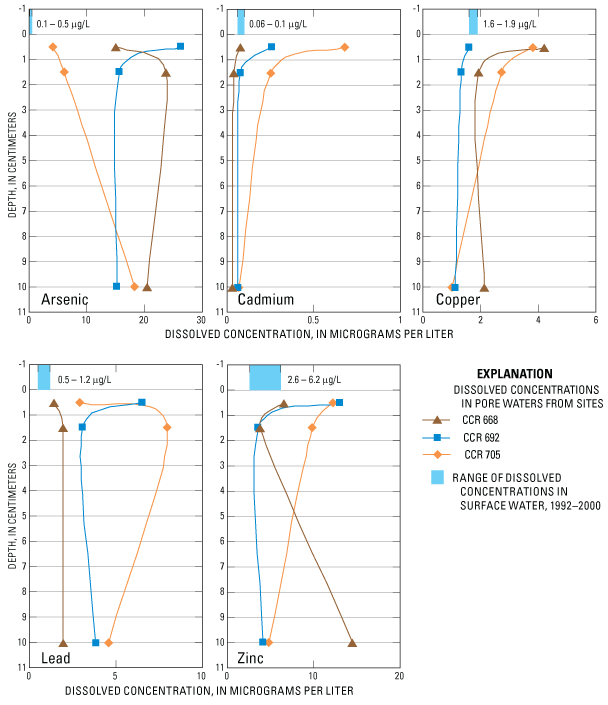
Concentrations of dissolved arsenic, cadmium, copper, lead, and zinc in pore waters collected from these sediments (fig. 8) were often larger than ambient dissolved concentrations measured in Columbia River water at the Northport sampling station between 1992 and 2000 (Bortleson and others, 1994; Kelly and others, 2001). Thus, concentrations of these trace elements dissolved in pore water is most likely due to their close association to the elevated trace-element concentrations in the adjacent sediments through either leaching or remobilization of trace elements from the sediment phase to the pore-water phase. If present, slag may be a continuous source of zinc, and copper, which are present in slag in comparatively large concentrations. Recent studies have clearly shown that copper, lead, zinc, and other trace elements are readily leached from similar metallurgical slags (Parsons and others, 2001; Lottermoser, 2002; Piatak and others, 2004), although earlier studies by the Trail smelter report that slag was relatively inert with respect to the leaching of trace elements based on U.S. Environmental Protection Agency's Toxicity Characteristic Leaching Procedure (Cominco, 1991).
Figure 8. Vertical distribution of concentrations of dissolved arsenic, cadmium, copper, lead, and zinc in pore waters from sediment core sampling sites CCR-668, CCR-692, and CCR-705, Lake Roosevelt, Washington.
Some trace-element concentrations in pore water show a gradient of decreasing concentrations with depth that is opposite the trace-element concentration gradient in the sediment. Cadmium, copper, and to a lesser extent zinc follow this pattern. No clear pattern was noted for the pore-water concentration gradients of arsenic and lead. The precipitation of metal sulfides may explain the smaller pore-water concentrations of some trace elements at depth. This explanation is supported by the lack of sulfate in pore water from cores CCR-668 and CCR-692 and the decreasing sulfate concentrations in the deeper intervals of core CCR-705. Under reducing conditions, which are thought to be present in sediments with pore water iron concentrations greater than 1,000 mg/L, sulfate reducing bacteria can readily convert sulfate to sulfide ions. In the presence of sulfide ions, dissolved metals such as iron, lead, mercury, and cadmium can form highly insoluble precipitates that are removed from solution.
Variability in pore-water concentrations also may occur seasonally due to the changing water levels in the reservoir. Water levels in the reservoir are lowered about 20 to 80 ft during April to June to provide storage volume for snowmelt in the upstream drainage basin. The changing lake level will affect the hydraulic head in the ground-water system discharging to the reservoir, likely increasing ground-water discharge during the drawdown period and possibly inducing the movement of surface water into the reservoir bed when the reservoir is returned to its normal operating water level. The reduction in static pressure during the drawdown period possibly increased the hydraulic gradient across the ground-water/surface-water interface increasing the potential for movement of pore water out of the bed sediments and into the overlying water. Following refilling of the reservoir to full pool elevation in June, the movement of water across the interface may have been reversed for a time when Columbia River water with relatively dilute concentration of trace elements may have seeped in the near-surface bed sediments. Pore-water samples were collected in mid-July, following return of the reservoir to full operating level after the seasonal drawdown of pool elevation to 1,265 ft the previous May. Information is not available to assess these potential effects on the pore-water concentration data.
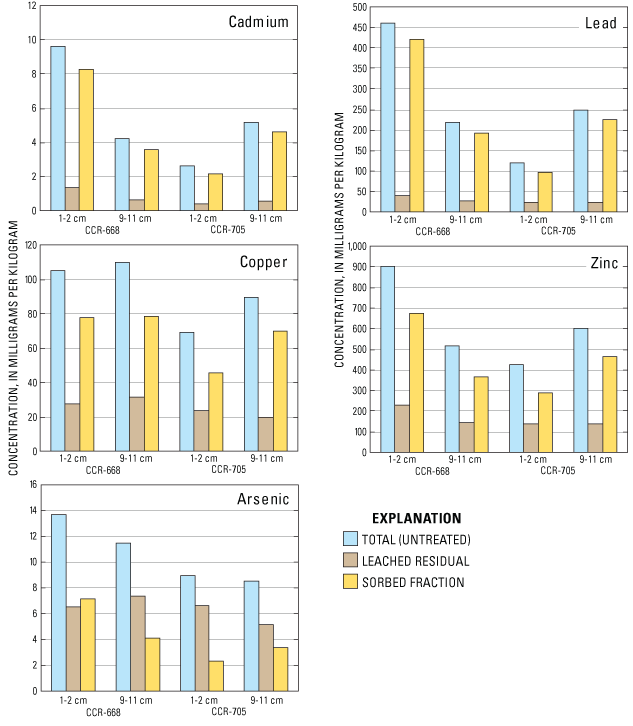
A larger fraction of the trace-element concentrations in sediments of the mid-reach of Lake Roosevelt occur associated with the surface of sediment grains rather than associated with the interior matrix of the sediment grains. Prior to analysis of trace-element concentrations, four sediment samples were treated with hydroxylamine hydrochloride reagent to remove trace elements that are weakly sorbed to the grain surface or incorporated within iron and manganese oxide coatings. Concentrations of trace elements in the residual sediment fraction of the samples treated with hydroxylamine hydrochloride reagent were substantially lower than concentrations in untreated sediment samples (table 7). Concentration data for the total, residual, and leached fractions of arsenic, cadmium, copper, lead, and zinc (table 7) were plotted in figure 9, which shows that most of cadmium, copper, lead, and zinc concentrations are associated with the leached fraction from the surface of the sediment particle or reducible oxide coatings that have precipitated onto sediment surfaces. Thus, most of these trace elements are associated with the surface of the sediment particle where they are relatively accessible and are potentially bioavailable or susceptible to remobilization. Cadmium and lead were most influenced by sediment leaching processes as 85 to 90 percent of these trace elements generally are removed as indicated by residual sediment concentrations that were 10 to 15 percent (fig. 9) of the concentration in the untreated sediment samples. This effect is less pronounced for copper and zinc where 66 to 78 percent of copper and zinc concentrations are removed by exposure to the hydroxylamine hydrochloride reagent yielding residual sediment fractions of copper and zinc (22 to 33 percent), which are over twice those for cadmium and lead. Copper and zinc are the trace elements of concern that have the greatest concentration in the interior matrix of the slag particles, which may partially explain their higher concentration in the residual fraction. Arsenic was present mostly in the residual fraction and not as greatly associated with the surface of the sediment particles as were cadmium, copper, lead, and zinc.
Figure 9. Concentrations of arsenic, cadmium, copper, lead, and zinc in whole sediment and leached residual (following extraction with hydroxylamine hydrochloride) from sediment core sampling sites CCR-668 and CCR-705, Lake Roosevelt, Washington, July 2003.
For more information about USGS activities in Washington, visit the USGS Washington District home page .