A second set of samples was collected during the snowmelt runoff period between May and June 1996 to compare the metal loads in water and colloids between low- and high-flow conditions (fig. 9). Results of determinations from these samples are in appendices III and IV. Only three sites were sampled during high flow, so there only is an outline of spatial detail. The samples, however, permit some generalizations about the loading of metals under two different flow regimes.
In the absence of acid-mine drainage, concentrations of dissolved metals generally decrease as flow increases. At high flow from snowmelt runoff, one would expect water quality standards to be achieved. Concentrations of some metals, however, did increase with higher flow, mostly the colloidal concentrations, creating the opportunity to exceed toxicity levels for some organisms. Among the dissolved metals sampled, zinc was the only metal that exceeded the acute toxicity standard. Zinc concentrations far exceeded the acute toxicity standard at site A-72 below Silverton, and at the two downstream sites (Durango and Cedar Hill), it was about twice the acute toxicity standard. Other metal concentrations either exceeded the chronic toxicity standard or were very close to the standard. Concentrations of copper did not exceed the standard, but were only a few micrograms per liter below the standard. During high flow, colloidal iron was greater than 1 mg/L, the standard for "total recoverable" iron concentrations, at both downstream sites. The concentrations of dissolved lead were below detection (<0.01 mg/L).
The samples collected at high flow contained some sand-sized particles that were screened out before measuring the metal concentrations in the colloids. This indicates that there was some unquantified metal transport associated with suspended particulate material (>62 mm) at high flow associated with these particles. Unfortunately, this component was not analyzed and has been discarded.
Measured stream flow discharge was multiplied by concentrations and converted to units of kilograms per day (kg/day) to evaluate the instantaneous synoptic metal load. There was a clear distinction between metals transported in the dissolved load and those transported in the colloidal load. Both dissolved and colloidal metals, however, had the same downstream profile of metal loading. The profile shows a sharp increase contributed by the inflows of Cement and Mineral Creeks near Silverton and then a gradual decrease during flow through the Animas Canyon and on downstream. The decrease, or attenuation, of the dissolved load resulted either from an increase of the colloidal load, or in sorption of metals onto material in the bed sediments. Attenuation of the colloidal load results from deposition of aggregated colloids to the stream bed and incorporation into the bed sediments.
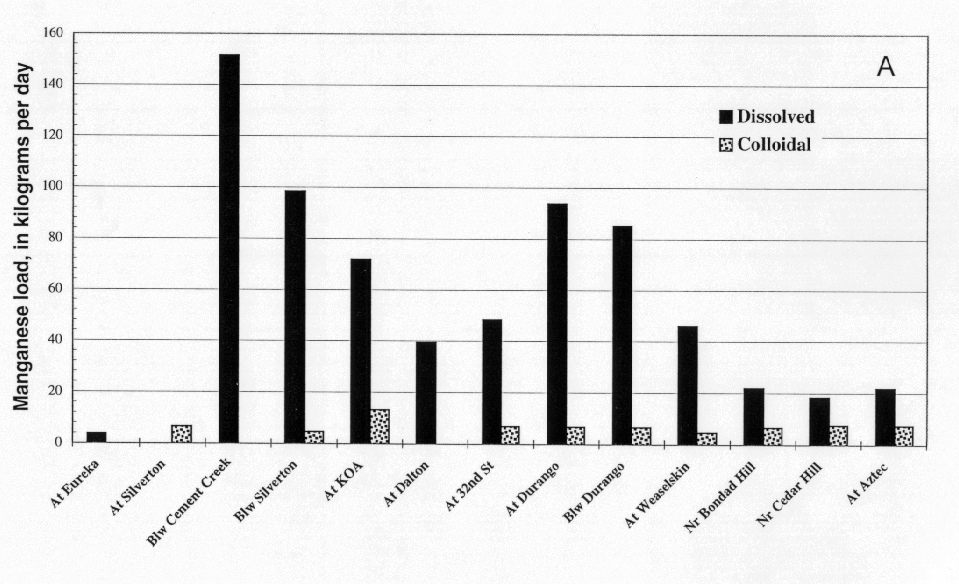

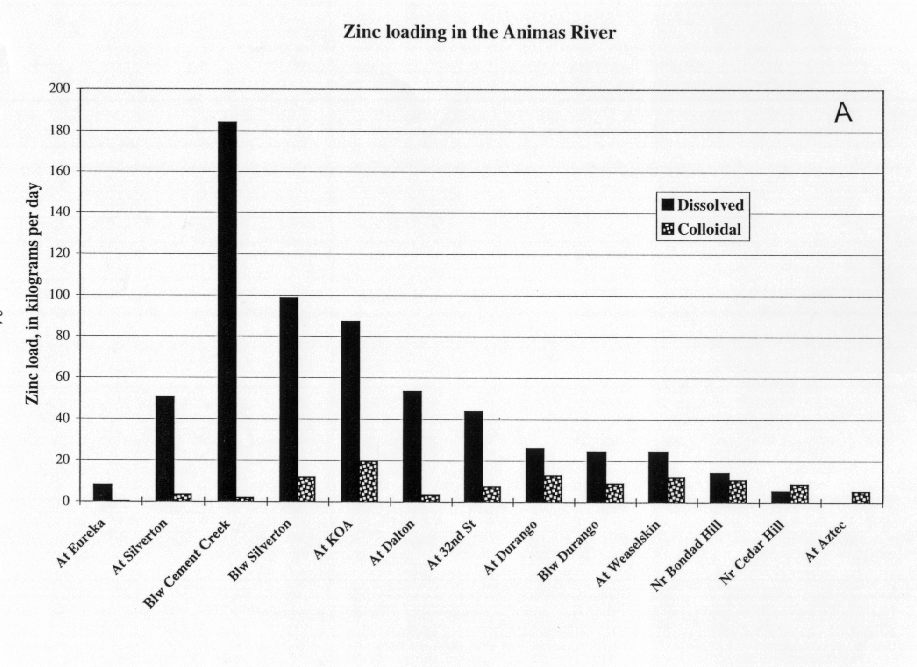

Manganese and zinc were transported mostly in the dissolved load of the Animas River (figs. 12A and 13A). The inflows from Cement and Mineral Creeks near Silverton were mostly as dissolved manganese and zinc (figs. 12B and 13B). In the mixing zone downstream from each creek, there was no partitioning of the dissolved metals to the colloidal phase, most likely because in the pH range between 6.5 and 7.5, and neither manganese nor zinc tended to sorb to or form colloids in this pH range. Although there was no partitioning to colloidal loads, both the dissolved loads of manganese and zinc decreased downstream indicating that these two metals were lost to the bed sediments. In the bed sediments, zinc, in particular, could increase the toxicity of sediments for aquatic organisms. In the Durango area, there was an increase in the dissolved manganese load, but not in the dissolved zinc load. The manganese may have been from urban sources, but more likely it is from sedimentary rocks that crop out in this section of the basins that are supplying detritus to the drainage.
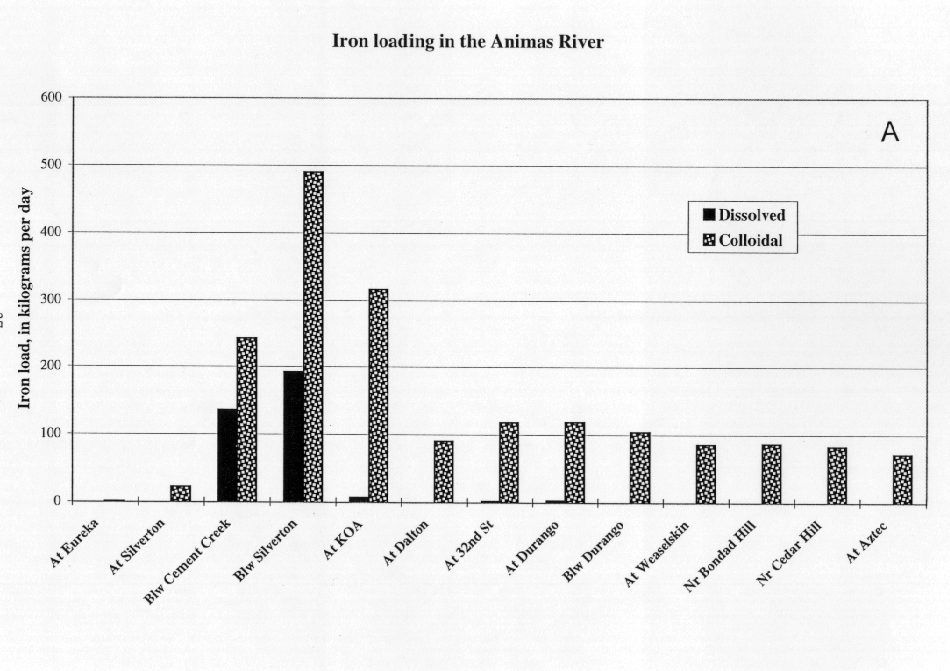
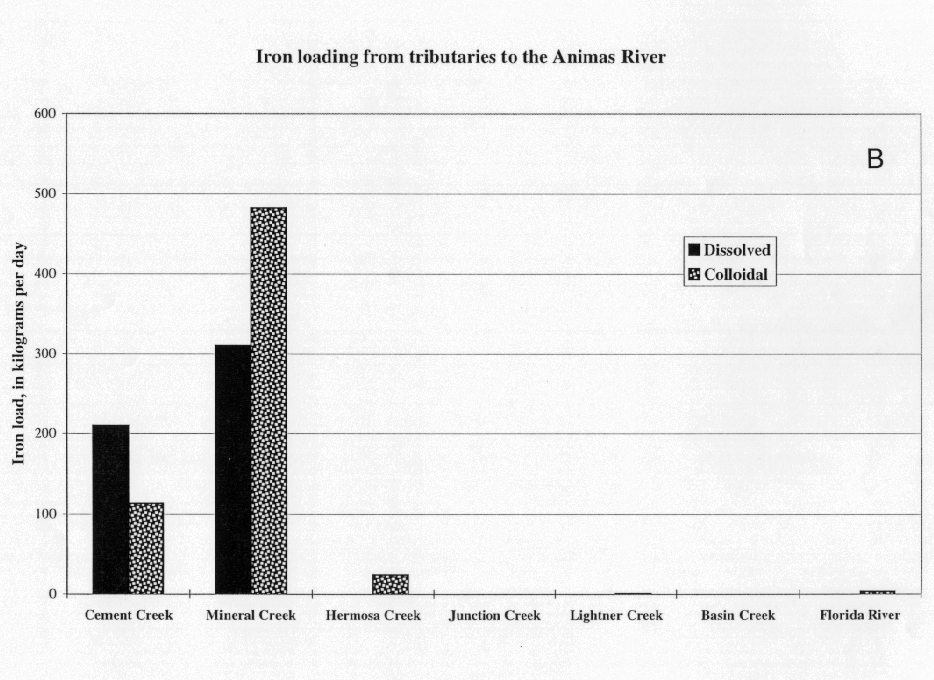
The profiles of colloidal loads of iron and aluminum were very similar (compare the data in figs. 14A and 15A), and the masses of colloidal iron and aluminum were nearly the same. The inflow of both metals from Cement Creek primarily was as a dissolved load, whereas the inflow from Mineral Creek was mostly as a colloidal load (figs. 14B and 15B). This difference in the chemical form of the load was the result of the different pH conditions in the two streams. In the mixing zone, downstream from the confluence with Cement Creek, the dissolved iron and aluminum loads from Cement Creek were partitioned almost completely to colloidal loads. This process occurred within tens of minutes as the water traveled 1 km from Cement Creek to Mineral Creek.
The contrast in the behavior of aluminum in Cement and Mineral Creeks illustrates the stronger influence of pH on the mode of aluminum transport. In Cement Creek, at a pH of 3.89, dissolved aluminum was 6.0 mg/L and colloidal aluminum was 0.1 mg/L. In Mineral Creek, at a pH of 6.35, dissolved aluminum was 0.09 mg/L, and colloidal aluminum was 3.6 mg/L. At sites along the Animas River between Cement Creek (24 km) and Dalton Ranch (81 km), most of the aluminum was colloidal, ranging from 1.72 mg/L (26 km) and 1.18 mg/L (24 km), to an average of 0.09 mg/L at sites beyond 81 km downstream. Downstream from 81 km, both dissolved and colloidal loads of aluminum were low.
The mass transfer or partitioning of aluminum from dissolved to colloidal loads quantifies the process influenced by differences in pH (fig. 16). Three sources of dissolved aluminum were represented: 10.4 kg/day from the upper Animas River above the confluence with Cement Creek, 283 kg/day from Cement Creek, and 12 kg/day from Mineral Creek. Adding the inputs from the upper Animas River and from Cement Creek should result in a dissolved aluminum load of 293 kg/day downstream from Cement Creek, but only 88 kg/day were measured. Thus, 205 kg/day of dissolved aluminum were lost, while 240 kg/day of colloidal aluminum was formed. This is a reasonable mass balance for the process of aluminum partitioning from the dissolved to the colloidal phase. In the mixing zone downstream from the confluence with Mineral Creek, there was a net loss of dissolved and colloidal aluminum from the stream. The loss of dissolved aluminum in that mixing zone was 89 kg/day, but there also was a loss of 256 kg/day of colloidal aluminum. This indicates that there was not only a loss of dissolved aluminum to form colloidal aluminum, but also a net loss of colloidal aluminum to the stream bed. These data suggest that aluminum precipitation to form colloidal particles and their aggregation in the water column is rapid it occurred just downstream from Cement Creek. The time required for settling or deposition of the colloidal aggregates to the stream bed sediments is longer it did not occur until the water traveled downstream from Mineral Creek. This sequence is schematically illustrated in figure 16.
The same process occurs for iron and the mass of iron that is lost to the stream bed was very similar to aluminum (fig. 17). The gain in colloidal iron downstream from Cement Creek was comparable to the loss of dissolved load, and there was a net loss of colloidal iron downstream from Mineral Creek. This amount of colloidal iron and aluminum would substantially affect the habitat of the stream bed by filling up pore space normally occupied by benthic invertebrates.


The dissolved copper concentration was below the limit of detection downstream from Cement Creek, even though the concentration entering from Cement Creek was near 6 mg/L. Copper was completely partitioned to the colloidal phase, probably as a result of sorption to colloids of iron and aluminum hydroxide, causing the colloidal concentration to increase to 13 mg/L downstream from Cement Creek. Sorption of copper continued downstream from Mineral Creek, and the colloidal concentration reached a maximum of 23 m g/L. The loss of colloidal copper load was about 5 kg/day downstream from Mineral Creek.
Unlike aluminum and iron, only a small amount of zinc was partitioned from the dissolved to the colloidal phase. In the Animas River downstream from Cement Creek, the dissolved zinc load was 184 kg/day, which was greater than the sum of the individual sources of the upper Animas River and Cement Creek. This increased load may indicate a contribution of zinc from some nonpoint sources, perhaps from dispersed tailings in the alluvium. In the Animas River downstream from Mineral Creek, the load of dissolved zinc was 99 kg/day, which represented a net loss of 131 kg/day of dissolved zinc. This net loss coincided with a gain of colloidal zinc of only 6 kg/day, so most of the dissolved zinc was not partitioned to the colloidal phase. Instead, a substantial loss of dissolved zinc to the stream bed occurred through sorption to the abundant aluminum and iron colloids that had previously settled to the stream bed.
These calculations not only demonstrate the substantial masses involved in the geochemical processes active in the streams, but also indicate the large quantity of colloidal material that becomes stored in the bed sediments. Once the colloids are part of the bed sediments, they influence the bed-sediment chemistry and affect the aquatic habitat. As described in the next section, much of the colloidal material is remobilized during high-flow periods.