The geochemical and lead-isotopic results from the bed-sediments from Cement Creek are summarized in figures 24-25. Geochemical profiles of Cement Creek differ substantially from those of Mineral Creek or the upper Animas River. Two samples labeled as "Other" are important in discerning the influence of natural weathering and spring seepage processes from mining activities on metal concentrations in the bed sediments. The sample upstream from the first bed-sediment sample of Cement Creek is from a ferricrete deposit on the west-facing slope of Cement Creek. A ferricrete is any rock cemented by iron oxides. Ferricretes may contain rounded or angular rock fragments cemented together by iron oxides that precipitate from natural weathering of mineral deposits. The second "Other" sample is from an iron bog forming below a spring on the west side of Cement Creek in the creek bed. Since this sample was collected from a small terrace just above Cement Creek at the low-flow stage, the chemistry of the sample will be affected by bed sediment material and water from Cement Creek entrapped during high-flow conditions. However, the very different geochemistry of the iron-hydroxide precipitates clearly shows that most of the metals in the iron bog are deposited from a different source.
Arsenic concentrations in bed sediments are elevated ranging from 23 to 43 times CAV (42-78 ppm) and the profile (fig. 24A and B) of both the partial and the total digestion data indicate that the arsenic concentration in the bed sediments increases significantly below the inflow at the Gladstone Mine and the confluence below Tiger Gulch. This increase is due to a higher arsenic in the iron-hydroxide phases and may represent accumulation of colloidal materials in the bed sediments. As we stated above, if the elevated arsenic concentrations reflect the addition of the chimney-type deposits, the increased arsenic levels could represent an unsampled source of metals coming from Prospect Gulch which drains the east side of Red Mountain. Copper, lead, and zinc do not show a significant change over this stream reach. The sample from Topeka Gulch, a small tributary drainage which has not been substantially affected by mining, contained similar concentrations (46 ppm) of arsenic to those found in the bed sediments of Cement Creek, but much less of it is in the labile phase (fig. 24B). In contrast, the arsenic concentration in the ferricrete sample was less than 10 ppm and in the iron bog sample was 6.7 ppm. The arsenic concentration in the bed sediments of Cement Creek at Memorial Park in Silverton is about twice that in either Mineral Creek or the upper Animas River at site A-72 in Silverton.
Cobalt concentrations in the bed sediments of Cement Creek are low and relatively constant at about 13 ppm (fig. 24C). The range is from about 10-30 ppm, near the CAV and showing a similar range to the other two major tributaries. Cobalt in the ferricrete sample is quite low, but in the iron bog sample is very similar to that in the bed sediments. Cobalt in the bed sediment from Topeka Gulch is higher than that in bed sediments in Cement Creek.
Copper concentrations (fig. 24D) in the headwaters of Cement Creek are elevated to about six times CAV (410 ppm). Copper concentrations in the ferricrete sample are also elevated (360 ppm) suggesting that some component of the elevated copper concentrations in the headwaters of Cement Creek may represent premining conditions. However, there are a few small mines in Ross Basin which were not sampled that may contribute copper and other ore-related metals to this upper reach of Cement Creek. Over most reaches of Cement Creek, the copper concentration is between 1-2 times CAV. The overbank sediment from just below the confluence of the South Fork of Cement Creek contains 290 ppm copper indicating that this sediment was transported from the uppermost reach of Cement Creek.
Lead concentrations (fig. 24E) range from 670 ppm (52 times CAV) at the headwaters to 320 ppm (25 times CAV) at Memorial Park in Silverton. Lead concentrations in both the ferricrete and the iron bog samples are much lower, in the 5 ppm range. The concentration of lead in the bed sediments from Topeka Gulch is about six times CAV, but substantially below that found in Cement Creek. Again, the lead concentration in the overbank sediment from below the confluence with the North and South Forks of Cement Creek is much higher than that in the sample from Cement Creek at this location.
Strontium concentrations in all samples except those from the ferricrete and the iron bog are relatively constant, averaging 250 ppm and are lower than CAV (384 ppm). The profile is relatively flat (fig. 24F). Strontium in solution behaves conservatively and is not sorbed or retained by the iron-hydroxide minerals thus, strontium concentrations in the ferricrete and spring seep samples (iron bogs) are very low.
Labile zinc concentrations (fig. 24G) are relatively constant over the course of Cement Creek near 200 ppm (2.6 times CAV) except for the sample from the headwaters where the zinc concentrations are slightly elevated (280 ppm or 3.7 times CAV). Contrasting the leach results from the bed sediments with the total zinc concentration data (fig. 24H) shows that about 33 percent of the zinc in the bed-sediment sample from the headwaters is labile whereas 67 percent is bound in more resistant sulfide mineral phase sphalerite. Microscopic identification of mineral phases and emission-spectrographic analysis of the residue from the leachate confirms that sphalerite, rather than tetrahedrite is present in the residual phase. The concentration of sphalerite-bound zinc in bed sediments remains relatively constant downstream from the Gladstone Mine, where it is 1,200 ppm, to Memorial Park in Silverton, where it is 1,000 ppm. The total zinc concentration in bed sediments at the confluence of Cement Creek with the upper Animas River is about 1,200 ppm or about 16 times the CAV.
Vanadium concentrations remain constant (averaging 143 ppm) throughout the course of Cement Creek at concentrations near CAV (fig. 24H). The vanadium profile indicates that there are no samples from Cement Creek that are biased by hydrologic concentration of either iron-hydroxide minerals or placering of heavy minerals.
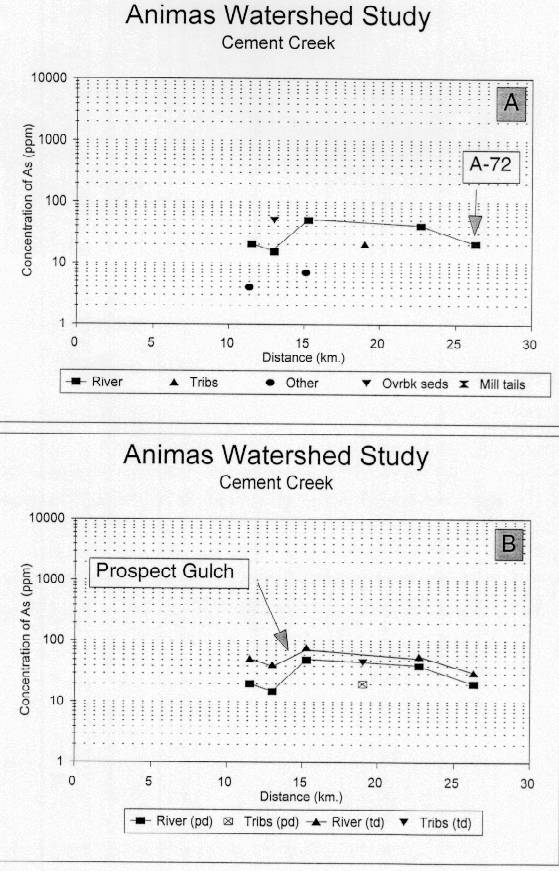
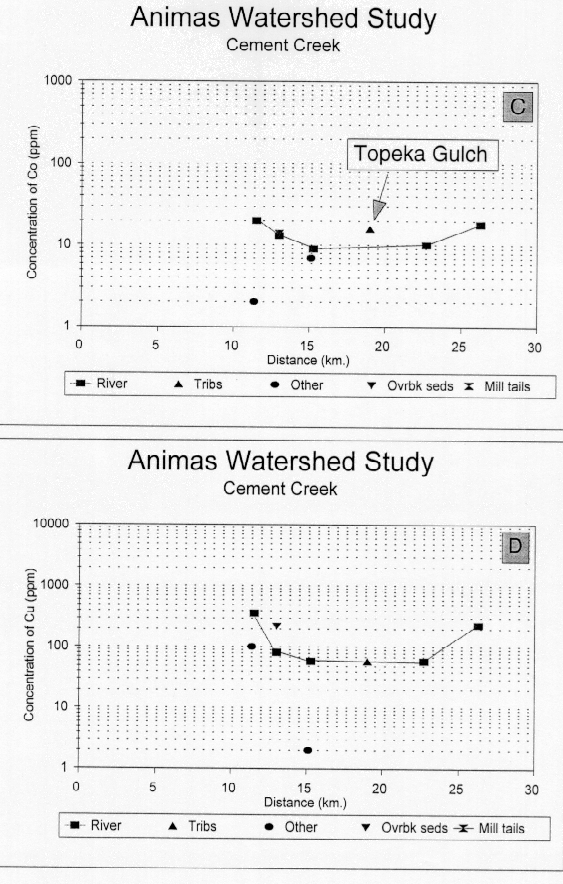
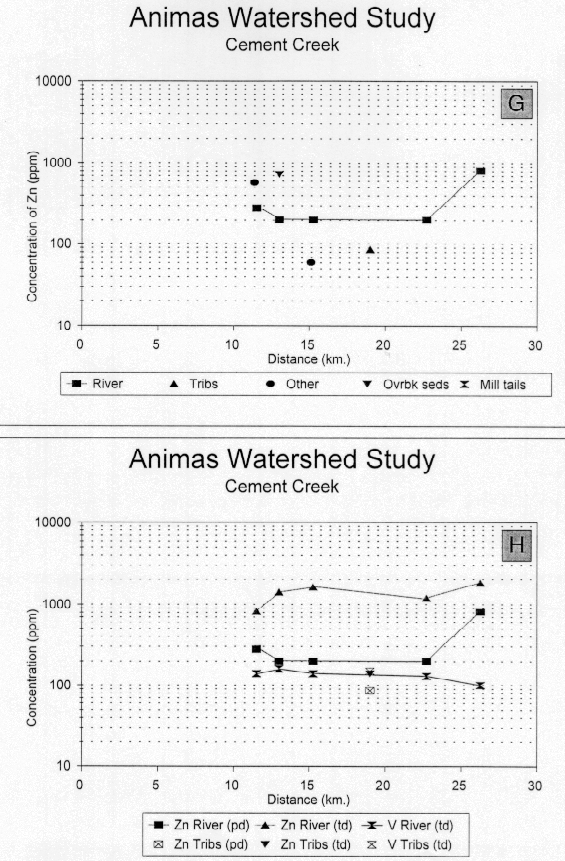
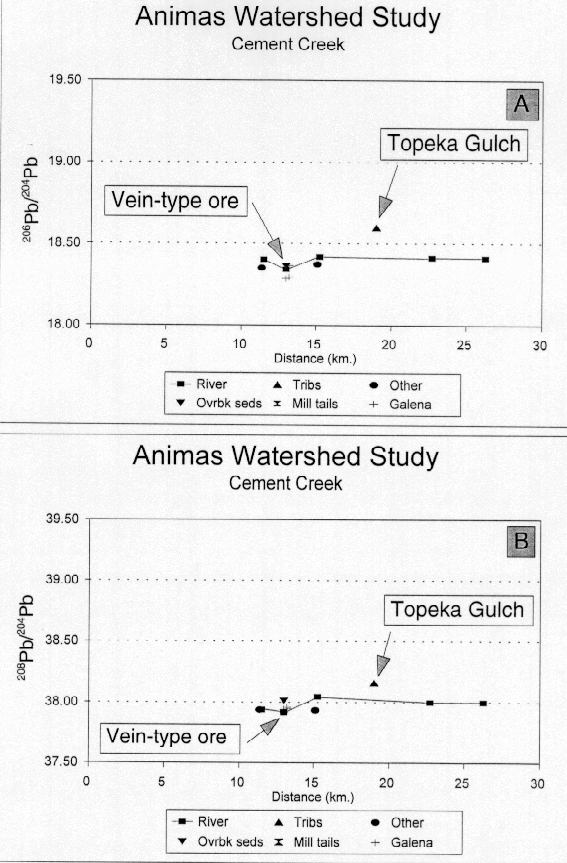 Figure 25. Lead-isotope distribution profiles for Cement Creek plotted against river distance measured
Figure 25. Lead-isotope distribution profiles for Cement Creek plotted against river distance measuredLead-isotopic data from the bed sediments, the overbank sediment, and the ferricrete and iron bog samples closely match the lead-isotopic signature of the Eureka graben vein-type mineralization (figs. 25A and B). The bed-sediment sample from Topeka Gulch, which is largely unimpacted by mining activity, has a composition that differs substantially from the rest of the samples from the Cement Creek drainage and may reflect the contribution of lead from altered, unmined country rock. However, mass balance calculations show that the total contribution of lead of this type to the bed sediment of Cement Creek is small and insignificant. The lead-isotopic data indicated that the majority of the lead in the bed-sediments from Cement Creek below the Gladstone mine matches that of the Eureka graben vein-type ore. Even though the lead-isotopic compositions of the ferricrete and iron-bog samples match the lead-isotopic composition of the vein-type ore, the lead concentrations in these samples are very low and thus, these sources add very little lead to the bed sediments in Cement Creek.