
Chapter 9
By
Ellen Thomas1, Taras
Gapotchenko 1, Johan C. Varekamp1,
Ellen L. Mecray2 and
Marilyn R. Buchholtz ten Brink2
The health of the LIS ecosystem is further affected by sediment pollution.
Atmospheric deposition and river-borne fluxes of pollutants, both organic
and metallic, led to the pollution of the top section of sediments (Mecray
and Buchholtz ten Brink, 2000; Varekamp and others, 2000). In addition,
some locations became by outflow from sewage treatment plants (Buchholtz
ten Brink and others, 2000).
Records of abundant biota which easily fossilize may reflect the effects
of anthropogenic and natural environmental changes in LIS. Benthic
foraminifera are such biota and are abundant in LIS. A detailed
study of LIS benthic foraminifera was conducted in the late 1940s (Parker,
1952), followed by an exhaustive study in the 1960s (Buzas, 1965).
LIS faunas are classified as 'marginally marine' and typically have a low
species diversity. The most abundant species have high tolerances
for fluctuations in temperature and salinity, as well as for low oxygen
conditions and environmental pollution (Alve, 1995).
We compare data on the composition of benthic foraminiferal assemblages
in LIS in 1996/1997 with the data collected in 1948 (Parker, 1952) and
the early 1960s (Buzas, 1965).
The grab samples were processed as described in Mecray and Buchholtz
ten brink (2000). Part of the freeze-dried samples was wet-sieved over
a 0.062-mm screen for foraminiferal analysis. The complete fraction larger
than 0.062 mm was used. The grab samples were not stained to detect
living specimens, so we could only study total faunas (living and dead
specimens), and compare these with similar data in Buzas (1965).
At several locations a sub-sample of the grab samples was placed in formaldehyde.
These samples were not buffered for long-term storage, and all carbonate
material was dissolved at the time of analysis. We processed and
studied several of these samples in order to determine whether agglutinant
specimens had been affected by freeze-drying. We found no differences between
agglutinant taxa abundance in the formaldehyde and the freeze-dried samples.
We prepared splits of the samples to contain at least 100 specimens of
foraminifera. Specimens were picked from all samples, identified to species
level, and stored in foraminiferal slides. SEM micrographs of selected
foraminifera are shown in Appendix 1.
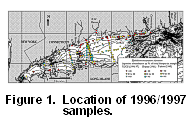 on
benthic foraminifera collected in 1996/1997 with data published on faunas
collected in the early 1960s by Buzas (1965) and in the late 1940s by Parker
(1952). Distributional maps and plots show the relative abundances
of E. excavatum clavatum (Micrograph
1; Figs. 1, 2),
Buccella
frigida (Micrograph 2 -ventral
view; Micrograph 3 - dorsal view;
Figs.
3, 4),
Eggerella advena
(Micrograph 4;
Figs.
5, 6), and Ammonia beccarii
(Micrograph 5- ventral view;
on
benthic foraminifera collected in 1996/1997 with data published on faunas
collected in the early 1960s by Buzas (1965) and in the late 1940s by Parker
(1952). Distributional maps and plots show the relative abundances
of E. excavatum clavatum (Micrograph
1; Figs. 1, 2),
Buccella
frigida (Micrograph 2 -ventral
view; Micrograph 3 - dorsal view;
Figs.
3, 4),
Eggerella advena
(Micrograph 4;
Figs.
5, 6), and Ammonia beccarii
(Micrograph 5- ventral view; 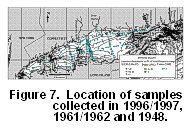 Micrograph
6 - dorsal view; Figs. 7,
8)
for the late 1940s, early 1960s, and late 1990s. The low diversity
benthic foraminiferal faunas of the 1960s were dominated by Elphidium
excavatum clavatum, with common Buccella frigida and Eggerella
advena. Elphidium incertum (Micrograph
7) was present in many samples; Trochammina squamata (Micrograph
8 - ventral view; Micrograph 9
- dorsal view) was rare.
Micrograph
6 - dorsal view; Figs. 7,
8)
for the late 1940s, early 1960s, and late 1990s. The low diversity
benthic foraminiferal faunas of the 1960s were dominated by Elphidium
excavatum clavatum, with common Buccella frigida and Eggerella
advena. Elphidium incertum (Micrograph
7) was present in many samples; Trochammina squamata (Micrograph
8 - ventral view; Micrograph 9
- dorsal view) was rare.
The 1996/1997 faunas (Table 1) were
still dominated by Elphidium excavatum clavatum and Buccella
frigida, and E. incertum was present in many samples, T.
squamata was rare. Eggerella advena, however, was rare
in all samples, and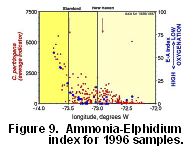 species diversity decreased. Ammonia beccarii was rare (<5%)
in 1961, but formed up to 27% of the assemblage in western LIS in 1996/1997.
As a result the Ammonia-Elphidium index (Fig.
9), high values of which have been linked to low oxygenation in
the Gulf of Mexico (Sen Gupta and others, 1996), increased strongly in
western LIS, where seasonal hypoxia is most pronounced (www.epa.gov/region01/eco/lis/lodo.html).
The A-E index shows a strong, positive correlation with counts of the bacterial
spore Clostridium perfringens, a sewage outfall indicator (Buchholtz
ten Brink and others, 2000).
species diversity decreased. Ammonia beccarii was rare (<5%)
in 1961, but formed up to 27% of the assemblage in western LIS in 1996/1997.
As a result the Ammonia-Elphidium index (Fig.
9), high values of which have been linked to low oxygenation in
the Gulf of Mexico (Sen Gupta and others, 1996), increased strongly in
western LIS, where seasonal hypoxia is most pronounced (www.epa.gov/region01/eco/lis/lodo.html).
The A-E index shows a strong, positive correlation with counts of the bacterial
spore Clostridium perfringens, a sewage outfall indicator (Buchholtz
ten Brink and others, 2000).
What caused these dramatic faunal shifts? A comparison of oxygen isotope data for E. excavatum clavatum collected in the 1960s and 1990s (Thomas and others, 2000) suggests no significant salinity changes over this time period. Carbon isotope data suggest that organic matter oxidation in LIS increased dramatically over the last 35 years, especially in western LIS. Core studies (Varekamp and others, 2000) indicate that the abundance of the sewage indicator spore increased over the last few decades. Therefore we suggest that both the bloom in A. beccarii (Fig. 9) in western LIS and the lighter carbon isotope values may be related to increased sewage inputs over the last 35 years.
To open georeferenced displays of the distributional themes in ESRI's
ArcView program make sure the application is loaded on your computer.
Users should go to the lisound directory located on the top
level of this CD-ROM and double click on the lisound.apr
project file. The individual ArcView shapefiles may also be opened
directly with any Arc application (e.g. ArcInfo, ArcExplorer) and can also
be found on the data page. Further
detailed information can be found on the ArcView
Project File page.
Buchholtz ten Brink, M. R., Mecray, E. L. and Galvin, E. L., 2000, Clostridium perfringens in Long Island Sound Sediments: An urban sedimentary record: Thematic Section, Journal of Coastal Research, in press.
Buzas, M. A., 1965, The distribution and abundance of Foraminifera in Long Island Sound: Smithsonian Institution Miscellaneous Collection, v. 149, no.1, p. 1-88.
Mecray, E.L. and Buchholtz ten Brink, M.R., 2000, Contaminant distribution and accumulation in the surface sediments of Long Island Sound: Journal of Coastal Research, Thematic Section, in press.
New York State Department of Environmental Conservation And Connecticut Department Of Environmental Protection, 1999, http://dep.state.ct.us/wtr/lis/tmdl.htm
Parker, F. L., 1952, Foraminiferal distribution in the Long Island Sound - Buzzards Bay area: Bulletin Museum Comparative Zoology, Harvard College, v. 106, no.10, p. 428-473.
Parker, C. A. and O'Reilly, J. E., 1991, Oxygen depletion in Long Island Sound: a historical perspective: Estuaries, v. 14, p. 248-264
Sen Gupta, B. K., Turner, R. E., and Rabalais, N. N., 1996, Seasonal oxygen depletion in continental shelf waters of Louisiana: historical record of benthic foraminifers: Geology, 24, p. 227-230.
Sun, M.-Y.; Aller, R. C. and Lee, C., 1994, Spatial and temporary distributions of sedimentary chloropigments as indicators of benthic processes in Long Island Sound: Journal of Marine Research, v. 52, p. 149-176.
Thomas, E., Gapotchenko, T., Varekamp, J. C., Mecray, E. L., and Buchholtz ten Brink, M. R., 2000, Benthic foraminifera and environmental changes in Long Island Sound: Journal of Coastal Research, Thematic Section, in press.
Varekamp, J. C., Buchholtz ten Brink, M.R., Mecray, E. L. and Kreulen,
B.,2000, Mercury in Long Island Sound sediments: Thematic Section, Journal
of Coastal Research, in press.
Figure 2. Relative abundance of Elphidium excavatum clavatum in samples collected in 1948 (Parker, 1952), in 1961/1962 (Buzas, 1965) and in 1996/1996 (Thomas and others, 2000). The color of the background at various levels corresponds with the color of the symbols in Figure 1.
Figure 3. Map of Long Island Sound showing location of the samples collected in 1996/1997 (Thomas and others, 2000), in 1961/1962 (Buzas, 1965) and in 1948 (Parker, 1952). Symbols indicate the year that a sample was collected, colors indicate relative abundance of Buccella frigida. (Fig. 4).
Figure 4. Relative abundance of Buccella frigida in samples collected in 1948 (Parker, 1952), in 1961/1962 (Buzas, 1965) and in 1996/1996 (Thomas and others, 2000). The color of the background at various levels corresponds with the color of the symbols in Figure 3.
Figure 5. Map of Long Island Sound showing location of the samples collected in 1996/1997 (Thomas and others, 2000), in 1961/1962 (Buzas, 1965) and in 1948 (Parker, 1952). Symbols indicate the year that a sample was collected, colors indicate relative abundance of Eggerella advena. (Fig. 6).
Figure 6. Relative abundance of Eggerella advena in samples collected in 1948 (Parker, 1952), in 1961/1962 (Buzas, 1965) and in 1996/1996 (Thomas and others, 2000). The color of the background at various levels corresponds with the color of the symbols in Figure 5.
Figure 7. Map of Long Island Sound showing location of the samples collected in 1996/1997 (Thomas and others, 2000), in 1961/1962 (Buzas, 1965) and in 1948 (Parker, 1952). Symbols indicate the year that a sample was collected, colors indicate relative abundance of Ammonia beccarii (Fig. 8).
Figure 8. Relative abundance of Ammonia beccarii in samples collected in 1948 (Parker, 1952), in 1961/1962 (Buzas, 1965) and in 1996/1996 (Thomas and others, 2000). The color of the background at various levels corresponds with the color of the symbols in Figure 7.
Figure 9. The Ammonia-Elphidium
index (Sen Gupta and others, 1996) for the 1996/1996 samples plotted versus
longitude, and compared with the number of spores of Clostridium perfringens,
a sewage indicator (Buchholtz ten Brink and others, 2000).
Table 2. Numerical abundances
of the most common species of foraminifera. A copy of this table (table2.xls)
may also be downloaded in Microsoft Excel format format.