 |
|
|
||||
| U.S. Geological Survey Open-File Report 2004-1350 |
Chapter 6. Stable Isotopes of Foraminifera from Pocomoke Sound |
| T. M. Cronin 1, R. Thunell 2, C. Saenger 1, R. R. Seal, II 1, and C. Vann 1 |
1U.S. Geological Survey, Reston, Virginia 20192; 2Department of Geological Sciences, University of South Carolina, Columbia, South Carolina 29208 |
IntroductionAnalysis of the oxygen and carbon isotopic composition of the calcitic (CaCO3) shells of foraminifers is a common tool in paleoenvironmental reconstruction because the ratios of heavier (18O, 13C) to lighter (16O, 12C) isotopes are influenced by factors such as the temperature and salinity of the water in which the foraminifer secretes its shell. The oxygen isotopic composition of foraminifer calcite is often expressed as δ18Oforam in the following relationship: 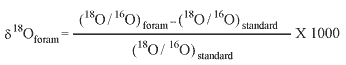
where values are expressed as per mil (‰). A similar relationship is used to express the δ13Cforam of foraminiferal calcite. The standards usually used for oxygen and carbon isotopes are Standard Mean Ocean Water (SMOW) and a belemnite from the Cretaceous Pee Dee Formation (PDB) in North Carolina, respectively (see Berger, 1979; Arthur and others, 1983). Although foraminiferal isotopic ratios are commonly used in deep-sea paleoceanography, this method also holds promise for understanding coastal and estuarine hydrological variability. This chapter describes preliminary results on the oxygen (δ18O) and carbon (δ13C) composition of the foraminifer Elphidium recovered from sediment cores PC-2B and PC-6B from Pocomoke Sound and compares these records to isotopic curves from the mainstem Chesapeake Bay. |
Material and MethodsA total of between 5 and 10 specimens of Elphidium were picked at the USGS laboratories in Reston, Virginia, from washed sediment in the > 150-µm size fraction at approximately 2-cm spacing from each core (PC-2B, PC-6B). An effort was made to select specimens having the best shell preservation with little of no signs of dissolution. In general, foraminifers are extremely well-preserved in sediments from Pocomoke Sound, showing no visible signs of post-mortem physical or chemical alteration. The oxygen and carbon stable isotope analyses on foraminifera were carried out at the University of South Carolina using a VG Optima stable isotope ratio mass spectrometer equipped with an Isocarb single acid bath carbonate preparation system which permits automated runs of 40 samples. This system has the capability of analyzing small carbonate samples for isotopic composition (<20 micrograms). |
Age Model for Pocomoke CoresIn order to evaluate the temporal trends in Pocomoke isotopes and establish correlations to cores in other parts of the bay, we constructed age model for Pocomoke cores PC-2B-3 and PC-6B-2 using a combination of age data from several sources. These include patterns in anthropogenic lead and Cesium-137 (137Cs) from core PC-6B-2 (Holmes and Marot, Chapter 2, this volume), the peak in ragweed (Ambrosia) pollen from several Pocomoke Sound cores (Willard and Bernhardt, Chapter 7, this volume), and radiocarbon dates from Pocomoke cores (Cronin and others, Chapter 4, this volume). Together with the lead, radio-isotopic and pollen data, we also used foraminiferal oxygen isotopic data as a means of correlating cores based on downcore isotopic excursions observed in all the cores (see below). |
Foraminiferal Isotopic ResultsFigure 6.1 shows the δ18Oforam and carbon δ13Cforam for the two cores plotted against the age model discussed below; the isotopic data are summarized in Table 6.1. The mean δ18Oforam values for all samples from PC-2B and PC-6B were -1.61 ‰ and 1.20 ‰ respectively. The δ13Cforam values are likewise lighter at the PC-2B site (-3.29 ‰ versus -3.00 ‰ at PC-6B). These patterns show that mean isotopic values are lighter (more negative) at core sites located closer to the source of fresh water and probably reflect the influence of isotopically light oxygen in water nearer to shore and river mouths and terrestrially derived organic material (see next section). We also computed the mean δ18Oforam and δ13Cforam values for the pre- and post-1900 intervals in order to see if any long-term change had occurred (Table 6.1). Pre- and post-1900 δ18Oforam and δ13Cforam for core PC-2B show no appreciable change during the last 100 years. In core PC-6B, post-1900 δ18Oforam values are about 0.17 ‰ heavier than values prior to 1900, whereas δ13Cforam values are on average about 0.3 ‰ lighter since 1900. Figure 6.1 also shows significant decadal variability in both δ18Oforam and carbon δ13Cforam at both core sites. δ18Oforam values prior to ~1900 AD have about a 2 ‰ range of variability; after 1900, δ18Oforam values exhibit greater negative and positive excursions, which are more obvious in core PC-2B. Similarly, the δ13Cforam values similarly have about a 2 ‰ range at PC-2B prior to 1900 AD and about 1.5 ‰ at PC-6B, and the range of variability increases markedly after 1900 AD, especially in PC-2B. The most obvious isotopic "events" are the positive excursions in both cores and in both oxygen and carbon isotopes just prior to 1800, around 1900, and during the late 1950s and early 1960s. The sharp negative excursion around 1950 in foraminiferal δ18O and δ13C is also one of the more striking aspects of the isotopic record. |
Factors Influencing Foraminiferal Isotopic CompositionOxygen isotopes The first factor to consider when interpreting isotopic composition of foraminiferal shells is whether the particular foraminiferal species or genus secretes its shell in isotopic equilibrium with the surrounding seawater. This complication has been referred to as the "vital effect" and it requires researchers to calibrate isotopic records from two different foraminiferal genera when more than one foraminiferal genus is analyzed in a core. Such a situation exists in studies of deep-sea planktonic and benthic foraminifers. Surprisingly, relatively little exist on oxygen isotopic vital effects in shallow water estuarine foraminiferal species. Nonetheless, Grossman (1984, 1987) conducted a survey of isotopes in many benthic foraminiferal genera and data for Elphidium suggest that the isotopic composition of its shell is out of equilibrium with the seawater in which it grows by about 0.8 per mil for δ18O. Thomas and others (2000) also conducted a preliminary study of isotopes of Elphidium excavatum from Long island Sound at salinities similar to those of Pocomoke Sound (20-30 ppt) and found an offset of about 1.1 ‰. They pointed out the vital effect is not critical in cases where the same species are being analyzed in a study, such as the current one. Assuming there is no temporal variability in the vital effect for the species of Elphidium living in Chesapeake Bay, their oxygen isotopic composition should be influenced primarily by the salinity and the temperature of the water in which they secrete their shell. Epstein and others (1953) determined the relationship between δ18O of carbonate and temperature expressed in the following equation: 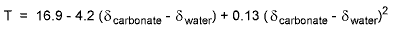
where T is temperature (°C), δcarbonate is the per mil difference between carbonate and standard mean ocean water (SMOW), δwater is the difference between δ18Owater and that of SMOW. In general, other factors being equal, a one per mil change in δ18Oforam is equivalent to a temperature change of about 4°C. In addition to temperature, the isotopic composition of estuarine waters is also influenced by the mixing of isotopically heavy marine water with isotopically light freshwater from regional precipiation and freshwater discharge. Thus, there should be a strong relationship between estuarine salinity and δ18Owater. We obtained δ18Owater values from a transect of water samples taken along the salinity gradient in the Potomac River and the polyhaline region of Chespapeake Bay in order to examine the modern δ18Owater gradient (Figure 6.2; http://geology.er.usgs.gov/eespteam/Atlantic/index.htm.). We included in Figure 6.2 δ18Owater values from oceanic water from the adjacent Atlantic continental shelf obtained from Chapman and others (1986, also Chapman and Beardsley, 1989) and Khim and Krantz (1996). This plot demonstrates the following salinity - δ18Owater relationship: 
Lower mesohaline waters have δ18Owater values around -6 to -5 ‰, polyhaline waters near -3 ‰ and marine waters -1 ‰. Ingram and others (1996) found a generally similar δ18Owater gradient in San Francisco Bay. The few fresh water samples shown in Figure 6.2 exhibit a wide range of δ18O values as would be expected from the influence of short-term temporal and spatial variability in precipitation sources throughout the watershed region. An additional factor to consider when interpreting foraminiferal oxygen isotopes is the fact that the growth of continental ice sheets during glacial periods causes large quantities of isotopically light fresh water to be stored on land as ice (as much as 120 m of sea-level equivalent). Glacial-interglacial ice-volume cycles occur over tens to hundreds of thousands of years and thus the ice volume effect is not a factor over the decadal to centennial timescales of the current study. Carbon isotopes Carbon isotopic ratios in estuarine water and sediments are influenced by the source of the organic carbon, which is influenced largely from the mixing of lighter (~ -26 ‰) terrestrially-derived organic matter and heavier (~20 ‰) marine organic matter. These carbon sources influence the composition of the dissolved inorganic carbon (DIC) taken up by foraminifera during shell growth such that there is an idealized salinity-δ13C gradient across the 0 to 35 ‰ salinity range. However, as shown by Thomas and others (2000) for Long Island Sound, other geochemical processes operating within the estuary can also influence the carbon isotopic composition of the DIC available to foraminifera for shell secretion. The two most important processes are primary productivity, which leads to heavier isotopic values when productivity is high, and oxidation of organic matter in the water column or near the bay bottom, which leads to lighter values (Anderson et al., 1983; Thomas and others, 2000). These processes, especially oxidation in the deep channel of Chesapeake Bay where seasonal hypoxia has occurred for at least a century (Zimmerman and Canuel, 2000; Cronin and Vann, 2003), might account for temporal patterns in δ13Cforam that do not seem to be controlled by salinity (see below). Carbon derived from different types of terrestrial plant can also be a source of variability; C3 plants (trees shrubs) usually have lighter and C4 plants (agricultural plants and grasses) have heavier organic carbon isotopic values. Similarly, marsh plants and submerged aquatic grasses living along the margins of Chesapeake Bay have relatively heavy carbon isotopic values. However, Bratton and others (2003) concluded from a study of carbon isotopes in Chesapeake Bay sediments that neither terrestrial plant type nor coastal marsh and seagrass sources had an appreciable influence on δ13C trends obtained from fine-grained sediments deposited in deeper regions of Chesapeake Bay where our cores were taken and Elphidium lives. Elphidium is also known to have photosynthetic endosymbionts living in its shell, but it is not clear if these influence the Chesapeake foraminiferal carbon isotope composition. Two other factors can influence the δ13C values on benthic foraminifers - vital effects and microhabitat effects. Vital effects in δ13Cforam are caused by either metabolic processes (i.e., algal or bacterial symbionts living in the foraminifera, Romanek and others, 1992) or kinetic processes (related to how fast the organism secretes its shell). Microhabitat effects involve differences in the isotopic composition of DIC in bottom water versus sediment pore water. Chandler and others (1996) found a strong microhabitat effect in experimentally cultured Ammonia from the Gulf of Mexico such that the δ13Cforam for shells secreted their shell in these living epifaunally (in bottom water) versus infaunally (in pore water) the influence of variability in δ13C. |
Paleosalinity During the Last Few Centuries in Chesapeake BayComparison of the δ18Oforam and δ13Cforam records from the two PC-cores and the RD/2209 core from the north-central bay provide evidence that, over the past few centuries, variability in both isotopes is primarily controlled by salinity and the mixing of freshwater and marine endmembers. Figures 6.3 and 6.4 show the Pocomoke oxygen and carbon isotopic records compared with those from the mainstem bay at sites RD/2209 and PTXT-2. Both oxygen and carbon isotope records exhibit correlative decadal-scale trends at the three sites suggesting that salinity was a primary, though not necessarily the only factor, influencing the foraminiferal isotopic composition at these timescales. We also used the oxygen isotopic records to "retrodictively" estimate paleosalinity following the procedures of Saenger and others (in press). In brief, this approach computes δ18Owater from the δ18Oforam based on the equation in Figure 6.2, and uses statistical models relating bay salinity to Susquehanna River discharge, and discharge to regional precipitation, to calculate paleosalinity trends. Water temperature effect on δ18Oforam is factored out by independently measuring paleotemperature of the water in paired samples using magnesium/calcium ratios in fossil ostracodes (Cronin and others, 2003). Figure 6.4 shows the resulting salinity patterns for the past 500 years and the anchor points used to develop the age model for cores RD/2209, PC-2B and PC-6B. It should be emphasized that because 137Cs and lead data were not available for all cores, and in some cases geochronological dating was carried out on a dedicated core, we used the δ18Oforam curve to "tune" the correlations among the three cores. For example, the stratigraphic position of the lead peak coincides with an isotopically heavy (high salinity) period near 1975 in both RD/2209 and PC-6B. Similarly, the 137Cs peak lies stratigraphically below the lead peak in both cores in an interval yielding slightly lower oxygen isotopic values. The third anchor point is the peak in ragweed pollen around 1880-1890 during a negative δ18O excursion. Despite age uncertainty, the records from the three cores are generally similar in the decadal patterns of salinity. Mean long-term salinity at PC-6B, the most saline of the three sites today, is higher than that at site PC-2B, which in turn is higher than that at site RD/2209. Periods of relatively high salinity corresponding to low freshwater discharge are evident near the turn of the 20th century, between 1910 and 1930, during the 1940s, and during the 1960s. Following relatively low salinity during the 1970s and 80's, there is evidence for increased salinity during the late 1980s and early 1990s at sites PC-2B and RD/2209 (the uppermost sediments at site PC-6B were disturbed). |
ConclusionsThese data allow us to postulate that stable isotope stratigraphy from Chesapeake Bay foraminifera, particularly oxygen isotope records, can be used as a means of correlation of sediment cores within the bay. Moreover, if regional decadal and centennial climate processes influence salinity as we suspect (Cronin and others, 2000, 2003), then it might be possible to extend this concept of isotope stratigraphy to other bays and estuaries because historical large-scale droughts and wet periods are known to have influenced large regions of the eastern United States. Large-scale regional climate variability should in theory have led to similar salinity variability in coastal environments influenced by freshwater river runoff and be manifested in the isotopic record of foraminifers. Future work is planned to further refine our understanding of the salinity signal from foraminferal isotopes with particular emphasis on identifying the long term climatic controls on oxygen isotopes and salinity and decoupling the effects of other processes (productivity and oxidation) on dissolved inorganic carbon and foraminferal carbon isotopes. |
ReferencesAnderson, T. F., and Arthur, M. A., 1983, Stable isotopes of oxygen and carbon and their application to sedimentologic and paleoenvironmental problems, In:, Arthur, M. A., Anderson, T. F., Kaplan, I. R., Veizer, J., and Land, L. S., (eds.), Stable Isotopes in Sedimentary Geology, Society of Economic Paleontologists and Mineralogists Short Course 10, p. 1-151.
Berger, W., 1979, Cushman Foundation Short Course on Foraminifera, pp. 156-198.
Bratton, J. F., Colman, S. M., and Seal, R. R. II, 2003, Eutrophication and carbon sources in Chesapeake Bay over the last 2700 years: Human impacts in context: Geochimica et Cosmochimica Acta, v. 67, No. 18, p. 3385-3402.
Chandler, G. T., Williams, D. F., Spero, H. J., and Xiaodong, G., 1996, Sediment microhabitat effects on carbon stable isotopic signatures of microcosm-cultured benthic foraminifera: Limnology and Oceanography, v. 41, p. 680-688.
Chapman, D. C., Barth, R. C., Beardsley, R. C., and Fairbanks, R. G., 1986, On the continuity of mean flow between the Scotian Shelf and the Middle Atlantic Bight: Journal of Physical oceanography, v. 16, p. 758-772.
Chapman, D. C. and Beardsley, R. C., 1989, on the origin of shelf water in the Middle Atlantic Bight: Bulletin American Meteorological Society, v. 19, p. 384-391.
Cronin, T., Willard, D., Karlsen, A., Ishman, S., Verardo, S., McGeehin, J., Kerhin, R., Holmes, C., Colman, S., Zimmerman, A., 2000. Climatic variability in the eastern United States over the past millennium from Chesapeake Bay sediments. Geology. vol. 28, p. 3-6.
Cronin, T. M., Dwyer, G. S., Kamiya, T., Schwede, S., Willard, D. A., 2003. Medieval Warm Period, Little Ice Age and 20th century temperature variability from Chesapeake Bay. Global and Planetary Change. vol 36, p. 17-29.
Cronin, T. M., Robertson, M., Willard, D. A., and Halka, J. Stratigraphy and Age of Pocomoke Sound Sediments: U.S. Geological Survey Open-file Report. This volume.
Cronin, T. M. and Vann, C., 2003, The sedimentary record of anthropogenic and climatic influence on the Patuxent Estuary and Chesapeake Bay ecosystems: Estuaries, v. 26, no. 2a, p. 196-209.
Epstein, S., Buchsbaum, R., Lowenstam, H., and Urey, H. 1953. Revised carbonate-water isotopic temperature scale: Geological Society of America, Bulletin, v. 64, p. 1315-1326.
Grossman, E. L., 1984, Stable isotope fractionation in live benthic foraminifera from the southern California borderland: palaeogeography, palaeoclimatology, Palaeoecology, v. 47, p. 301-327.
Grossman, E. L. 1987. Stable isotopes in modern benthic foraminifera: a study of vital effect. Journal of Foraminiferal Research, v. 17 (1), p. 48-61.
Holmes, C. and Marot, M., Sediment and chemical flux history in the Pocomoke Sound as defined by short lived isotopic analyses: U.S. Geological Survey Open-file Report. this volume.
Ingram, B. L., Conrad, M. E., and Ingle, J. C., 1996, Stable isotope and salinity systematics in estuarine waters and carbonates: San Francisco Bay: Geochimica et Cosmochimica Acta, v. 60, p. 455-467.
Khim, B.-K., and Krantz, D. E., 1996, Oxygen isotopic identity of the Delaware Coastal Current: Journal of Geophysical Research, v. 101, No. C7, p. 16,509-16,514.
Romanek, C. S., Grossman, E. L., and Morse, J. W., 1992, Carbon isotopic fractionation in synthetic aragonite and calcite: Effects of temperature and precipitation rate: Geochimical et Cosmochimica Acta, v. 56, p. 419-430.
Saenger, C., Cronin, T. M., Thunell, R., and Vann, C. in press, Modeling River Discharge, Precipitation, and Estuary Salinity: Application to Holocene Paleoclimate History: The Holocene.
Shackleton, N. J. and Opdyke, N. D. 1973. Oxygen isotope and paleomagnetic stratigraphy of equatorial Pacific core V28-238: Oxygen isotope temperature and ice volumes on a 105 and 106 year scale: Quaternary Research, v. 3, p. 39-55.
Thomas, E., Gapotchenko, T., Varekamp, J. C., Mecray, E. L., and Buchholtz ten Brink, M. R., 2000, Benthic foraminifera and environmental changes in Long Island Sound: Journal of Coastal Research v. 16, p. 641-655.
Willard, D. A., and Bernhardt, C., Pollen Stratigraphy and Land-Use Change, Pocomoke Sound, Maryland: U.S. Geological Survey Open-file Report. This volume.
Zimmerman, A. R., and Canuel, E. A., 2000, A geochemical record of eutrophication and anoxia in Chesapeake Bay sediments: anthropogenic influence on organic matter composition: Marine Chemistry, v. 69, p. 117-137. |
| AccessibilityFOIAPrivacyPolicies and Notices | |
 |
|