| Lake Pontchartrain
Basin: Bottom Sediments and Related Environmental Resources |
Continuous Sediment Sampling and Analysis System
Appendix A. Quality Assurance and Control
Several steps were taken to ensure that the systems used to perform the
survey were operating properly at all times. The methods for the quality assurance and
control were documented in the Quality Assurance Project Plan (QAPP) for this
project (CAIS, 1997). The topics addressed
during the quality control measures are addressed in the following text.
- Precision and accuracy
- Total sample concentrations
- Interlab sample comparison
Precision and Accuracy
A replicate sample analysis was performed for the NIST 2704 wafer.
Table A1 shows the results of the replicate analyses processed from the NIST Standard
2704. The precision and accuracy results were generated by repeating XRF analysis on the
same wafer at least five times. All analytes for the filter sample were within the
expected range of precision and accuracy.
Table A1. XRF Data Quality Measurements for Filters
| System |
Analyte |
Precisiona |
Precisionb |
Precisionc |
Accuracyc |
XRF
wt% |
Al |
±2.4% |
±2.3% |
±25% |
±25% |
| Si |
±1.4% |
±5.6% |
±25% |
±25% |
| |
S |
±5.4% |
±8.5% |
±25% |
±25% |
| |
Fe |
±1.2% |
±1.8% |
±25% |
±25% |
| |
Ca |
±1.6% |
±3.6% |
±25% |
±25% |
| |
K |
±0.7% |
±N/A |
±25% |
±N/A |
| |
Ti |
±1.0% |
±3.7% |
±25% |
±25% |
| |
Mg |
±4.9% |
±2.8% |
±25% |
±25% |
| |
|
|
|
|
|
| ppm |
Cr |
±4.7% |
±3.9% |
±25% |
±25% |
| |
Mn |
±2.6% |
±1.7% |
±25% |
±25% |
| |
Ni |
±19.3% |
±10.2% |
±25% |
±25% |
| |
Cu |
±1.8% |
±0.4% |
±25% |
±25% |
| |
Zn |
±1.5% |
±0.2% |
±25% |
±25% |
| |
Zr |
±9.0% |
±3.9% |
±25% |
±25% |
| |
Sr |
±6.8% |
±3.9% |
±25% |
±25% |
| |
Cd |
±28.4% |
±12.2% |
±40% |
±40% |
| |
Sb |
±37.9% |
±9.0% |
±40% |
±40% |
| |
Sn |
±14.6% |
±13.9% |
±40% |
±40% |
| |
Ba |
±8.3% |
±3.7% |
±25% |
±25% |
| |
Pb |
±2.3% |
±1.7% |
±25% |
±25% |
a Relative standard deviation based on replicate analysis of NIST 2704
filter.
b Difference from true value based on replicate analysis of NIST 2704 filter.
c Acceptance/rejection values.
Total Sample Concentration
Analytical totals for the samples analyzed from the 1997 Lake
Pontchartrain survey ranged from approximately 85% to 115%. This range in totals was
reflected in the individual elemental concentrations and caused a scatter effect in the
final results. When the data were plotted in a metal to metal format that generally shows
correspondence, there was only scatter showing little to no correlation. The problem of
variable total concentration has been solved by normalizing the data to a 90% total. By
normalizing the data to 90%, the variation is greatly reduced and the data plots show
considerably less scatter. Figure A1 gives an example of the relationship of
aluminum to silicon for
the 1997 Lake Pontchartrain data. With the exception of a few samples, the correlation
gives a reasonable regression. In addition, mean aluminum values for normalized 1997 data (6.51
wt%) and for normalized 1996 data (6.89 wt%) are very close. The 1997
aluminum range was 3.1 to
9.89 wt% as compared to 2.0 to 9.19 wt% for the 1996 data.
While 90% is somewhat arbitrary, it was chosen because the structural H2O
was determined to be relatively consistent at 8% to 9%, and elemental Na values were
consistently between 0.5% and 2%. The consideration of the structural H2O and
Na, along with the remaining analytes should account for >99% of the elemental makeup
of the sample.
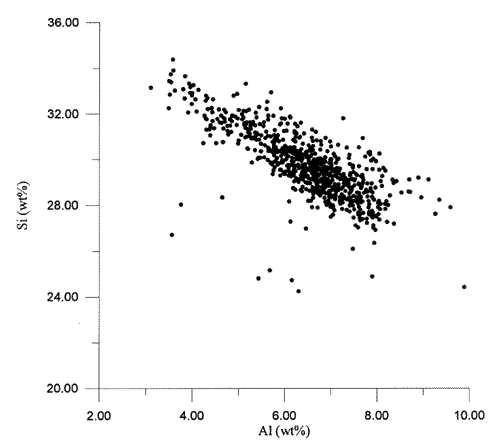
Figure A1. Normalized aluminum to silicon plot for the 1997 Lake
Pontchartrain data.
Interlab Sample Comparison
In order to test the quality of the data, six samples from the 1997
Lake Pontchartrain survey were sent to two labs, the XRF lab at Georgia State University
(GSU) and the analytical lab at Skidaway Institute of Oceanography (Skidaway). The samples
were chosen not because of quality or location but because of extra sediment collected at
these particular stations. The samples were collected during the 1997 survey as bulk
sediment slurry samples from the CS³ processor at approximately the same time that the
corresponding CS³ filter sample was collected. These samples were dried and archived for
future use. Figure A2 shows the results of the interlab comparison of the six Lake
Pontchartrain samples. Five of the samples show a good correlation. One sample, CAIS
Sample 244, appeared to be out of line with its counterparts analyzed by GSU and
Skidaway. Upon reanalyses, the aluminum concentration remained the same as previously reported.
Since this sample was originally analyzed in 1997, and now in 1998 with the same results,
it is mostly likely not a random error on the part of the XRF. Analytical methods, such
as XRF and atomic absorption, may not always agree usually reflecting the differences in
sample preparation. However, it was shown by figure A2 that the data reflect the same
relative difference from one method to another.
The GSU samples were processed into fused glass disks and analyzed by a
Rigaku 3070 wavelength-dispersive spectrometer utilizing a side-window Rh target X-ray
tube (XRF). The sediment was fused using lithium borate flux by heating in a furnace at
1100°C. The ratio of flux to sediment was 9:1, producing a
glass disk that was essentially a borate glass with sediment dissolved in it.
The Skidaway samples were processed using a lithium metaborate fusion
and analyzed by an atomic absorption spectrophotometer (AA) following EPA Method 7020.
Approximately 250 mg of sample was placed in a clean alumina crucible and heated at 900°C for 30
min. After cooling, the sample was reweighed and
percent loss on ignition was calculated. The ratio of lithium metaborate flux to sample
powder is 4:1. A total of 100 mg of ashed sample with 400 mg flux was thoroughly mixed and
then placed in a graphite crucible. The crucible was then placed into a muffle furnace at
1050°C for 8 min., removed, swirled, and returned to the
oven for another 7 min. The glass beads from the crucibles were poured directly into a
wide-mouth bottle containing 50 mL of 1.5N HNO3 spiked with 10 ppm Ge. The
sample was vigorously shaken until all the glass was dissolved. The sample was then
analyzed by the AA for aluminum content.
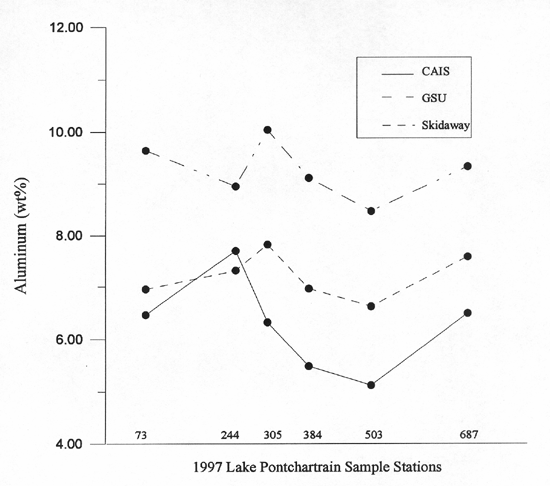
Figure A2. Interlab sample comparison.
 Back to Continuous
Sediment Sampling and Analysis System Back to Continuous
Sediment Sampling and Analysis System
|