MAJOR FINDINGS—Continued
It is important to understand how natural processes affect ground-water quality in order to identify the effects of urban and agricultural development under similar hydrogeologic conditions. In the CAZB, the majority of ground-water basins do not have significant urban or agricultural development. The ground-water quality in these basins is primarily a product of natural processes such as the interaction of ground water with rocks and sediment in the basins (Robertson, 1991).
Natural sources of dissolved-solids and nitrate can control ground-water quality in basins with minimal urban development. Specific-conductance values (an indirect measure of the dissolved-solids concentration) and nitrate concentrations for ground water in basins with minimal urban development increase northwestward from southeastern Arizona toward the central part of the State (figs. 16 and 17). The increasing specific-conductance values can be attributed to a corresponding increase in evaporite deposits in basin sediments from southeast to northwest (Gellenbeck and Coes, 1999). Evaporite deposits in the basins contain minerals such as halite (salt) and gypsum that can be easily dissolved in ground water. (Robertson, 1991). The increasing nitrate concentrations can be largely attributed to natural sources; however, human activities such as agriculture can be a source in some basins. In some locations in the CAZB, high nitrate concentrations in ground water reported prior to any agricultural or urban development indicate that natural sources of nitrate are present in some basins (Hem, 1985; Robertson, 1991; Gellenbeck, 1994; Gellenbeck and Coes, 1999). Dissolution of evaporite deposits, decay of buried organic matter, precipitation, weathering of rocks and soils, and fixation by microorganisms are just a few of the possible sources of naturally occurring nitrate in ground water.
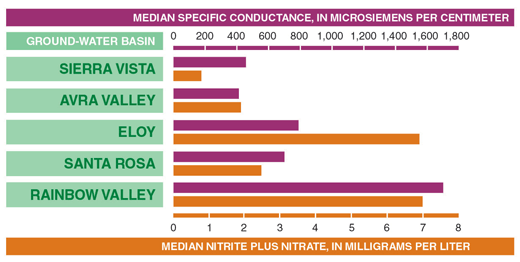 |
| Figure 16. Nitrate concentrations and specific conductance values in ground water from basins with minimal urban development increase from the southeast to the northwest. (Basins shown below in figure 17.) |
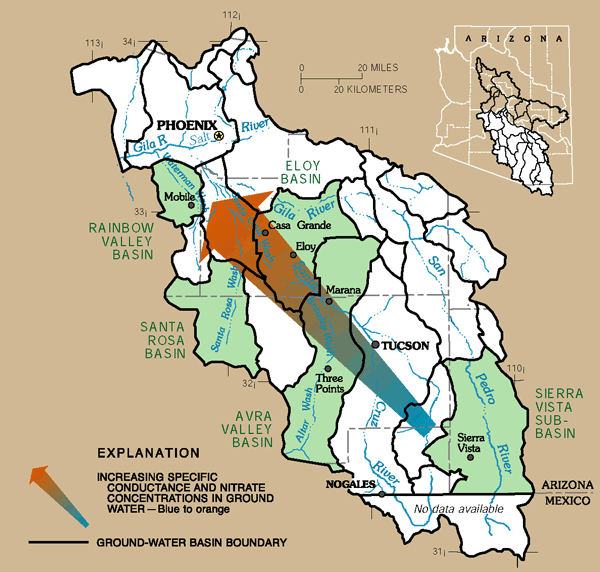 |
| Figure 17. Increasing specific conductance values in ground water from southeast to northwest can be attributed to an increase in soluble evaporite deposits in basin sediments. Increasing nitrate concentrations in the same direction may be the result of naturally occurring nitrate and of human activities that include agriculture. |
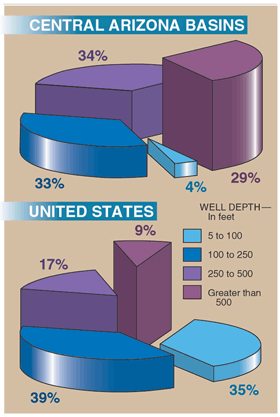
Quality of deep, older ground water unaffected by human activities In general, ground water in Arizona is replenished
(recharged) at very slow rates because of little precipitation,
high evaporation losses, and the long distance water must travel
to recharge deep aquifers. In the CAZB, 63 percent of the wells
sampled that draw water from aquifers used for drinking water were
at least 250 feet deep compared to 26 percent of NAWQA wells sampled
nationwide (fig. 18). Recharge takes longer to reach deep aquifers
than shallow aquifers; therefore, deep ground water typically was
recharged earlier and is older than shallow ground water. Tritium
age dating of ground water (see below) confirms that 55 percent
of the wells sampled in the CAZB yielded ground water that was recharged
prior to 1953 and possibly thousands of years earlier. For example,
some ground water in the Upper Santa Cruz Basin was determined to
be about 6,500 years old (Kalin, 1994). Across the Nation, only
27 percent of the NAWQA wells sampled for tritium yielded ground
water that was recharged prior to 1953. |
||
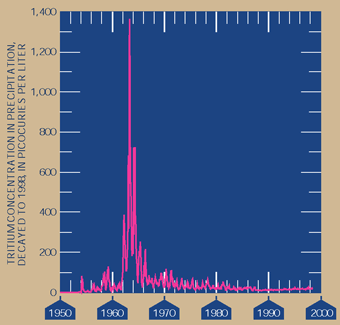
Method for dating ground water Tritium is a radioactive isotope that can be used to estimate whether ground water has been recharged before or after 1953. Large quantities of tritium were released to the atmosphere during testing of thermonuclear weapons from 1952 until the late 1960s. Atmospheric tritium is incorporated into water molecules in the atmosphere prior to precipitation and recharge to ground water at a given time is controlled by both the quantity of tritium in the atmosphere when precipitation and recharge occur and the radioactive decay rate of tritium. Ground water that does not contain detectable tritium (less than 2.5 picocuries of tritium per liter) can be assumed to have been recharged prior to 1953, and ground water that does contain detectable tritium (more than 2.5 picocuries of tritium per liter) is assumed to contain some component of ground water that was recharged after 1953. Trituim concentrations in precipitation are a guide for determining when ground water was recharged. |
Concentrations of arsenic, fluoride, and molybdenum exceeded drinking-water standards in samples from major aquifers. The median arsenic concentration in ground water for the three CAZB basins sampled was 4 µg/L. One sample from the Upper Santa Cruz Basin and one sample from the West Salt River Valley exceeded the current MCL for arsenic of 50 µg/L; however, a new, lower standard of 5 µg/L has been proposed by the USEPA because of the cancer risk posed by arsenic in drinking water (U.S. Environmental Protection Agency, rev. August 25, 2000). When arsenic concentrations in ground water sampled in the CAZB are compared to the proposed standard, more than 50 percent of samples from aquifers in West Salt River Valley that are used for drinking water exceed 5 µg/L. Seventeen percent of sampels in the Upper Santa Cruz Basin and 10.5 percent of samples in the Sierra Vista subbasin exceed 5 µg/L. The USEPA may not settle on 5 µg/L, but the new standard is likely to be significantly lower than the current MCL.
The median concentration of fluoride was 0.5 µg/L; about 2 percent of the smaples exceeded the current MCL for fluoride of 4 µg/L. The median concentration of molybdenum was 3 µg/L; about 1 percent to the samples exceeded the current lifetime health advisory for molybdenum of 40 µg/L established by the USEPA.
 |
| Figure 19. Samples are collected at the well head for randon analysis to prevent possible sample contamination from exposure to the atmosphere. |
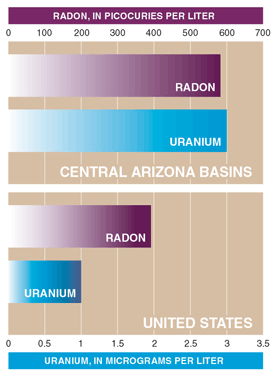
Radon and uranium are detected in most ground-water samples. Radon is a colorless and odorless radioactive gas that is carried in the water pumped from wells (fig.19) and released to indoor air by activities such as cooking and showering. Breathing radon increases the risk of lung cancer (U.S. Environmental Protection Agency, rev. October 18, 1999). Radon is naturally formed in rocks and soils from the radioactive decay of radium, an intermediate product in the uranium decay process. In the CAZB Study Unit, radon was present in 100 percent of the samples, and uranium was detected in 90 percent of the samples. The median concentrations for radon and uranium were 584 picocuries per liter and 3 micrograms per liter, respectively. Currently (2000), there are no USEPA MCLs for radon and uranium; however, proposed MCLs could result in increased costs for water suppliers to treat drinking water for these constituents or find alternate supplies. Additional costs would probably be passed on to the water user (see information on proposed standards for arsenic, radon, and uranium on p. 20).
Effects of Human Activities on Ground-Water Quality
The contamination of major aquifers is largely controlled by hydrology and land use (U.S. Geological Survey, 1999). In the CAZB Study Unit, deep ground water that was recharged prior to 1953 typically has not been affected by human activities (see p. 18). In areas with recent recharge (after 1953), ground water is more likely to be contaminated by nutrients and man-made chemicals associated with urban and agricultural land uses.
Ground-water quality deteriorates in irrigated areas. Irrigation water that seeps downward is a principal source of ground-water recharge in irrigated areas of the CAZB. Dissolved-solids concentrations in seepage can be as much as five times those in the original irrigation water (Bouwer, 1990) because of concentration by evaporation and plant use (see p. 9). The greater the dissolved-solids concentration in the applied irrigation water, the greater the concentration in the seepage moving downward to the ground water.
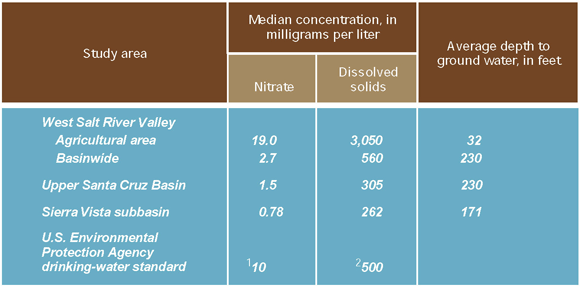
To determine the effects of irrigated agriculture on shallow ground-water quality, nine monitoring wells were drilled and sampled in the southwestern part of the West Salt River Valley (see Study Unit Design, p. 26). Because the average depth to ground water in the nine wells is 32 feet (table 1) compared to 230 feet for wells sampled basinwide, irrigation seepage does not have to travel far to reach the shallow ground water in the agricultural area. Sources of irrigation water in this area include treated sewage effluent, water from the Salt River and CAP canal, irrigation return flows, and ground water. Dissolved-solids concentrations of these sources range from about 900 mg/L for treated sewage effluent (Tadayon and others, 1998) to 650 mg/L for CAP water and 470 mg/L for Salt River water (Salt River Project, 1997).
The median dissolved-solids concentration in water from the nine shallow wells exceeded 3,000 mg/L (table 1). In addition, the effects of nitrate from fertilizer applications and reuse of irrigation return flows were evident from the median nitrate concentration that was nearly twice the MCL of 10 mg/L (table 1).
|
Table 1. Median concentrations of nitrate and dissolved solids were highest in shallow ground water from an agricultural area in the West Salt River Valley.
|
|
During 1991-98, arsenic, radon, and uranium were measured in ground-water samples from 36 NAWQA Study Units across the United States. If the ground-water samples from these Study Units are representative of ground water across the Nation, MCLs (see Glossary) proposed by the USEPA for these constituents will affect many water suppliers and municipalities in the United States. Because ground-water supplies in many parts of the Nation will likely exceed the proposed MCLS, public-water systems would be required to either specifically treat their water to decrease concentrations of the constituents or find alternative sources of supply. Costs of these options would probably be passed on to water users. |
||
| Arsenic
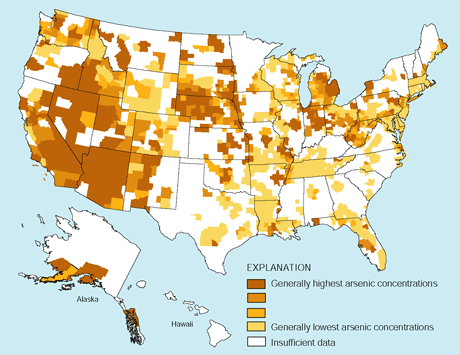
The current USEPA MCL for arsenic in drinking water, 50 µg/L, is under review after recognition of the risks of developing cancers (National Research Council, 1999). In 2000, the USEPA proposed a new, lower arsenic MCL of 5 µg/L (U.S. Environmental Protection Agency, rev. June 2, 2000). In samples collected in three CAZB basins, only 2 percent exceeded the current USEPA MCL; however, 32 percent exceeded the lower, proposed MCL. In samples collected across the Nation, only 0.6 percent exceeded the current USEPA MCL; however, 14 percent exceeded the lower proposed MCL. Arizona, including the CAZB, is among the areas in the Nation where 10 percent or more of ground-water samples are likely to exceed the lower MCL (figure at right; Welch and others, 2000). |
||
Radon and uranium Currently, USEPA MCLs for radon and uranium do not exist. Because of public health concerns, including increased risks for developing lung cancer, the USEPA proposed an MCL of 20 µg/L for uranium in 1991. In 1999, the USEPA proposed an MCL of 300 picocuries per liter (pCi/L) for radon; however, if States or water suppliers implement methods to lower radon levels in indoor air, they would only be required to meet an Alternate MCL (AMCL) of 4,000 pCi/L (U.S. Environmental Protection Agency, rev. April 21, 2000; U.S. Environmental Protection Agency, rev. October 18, 1999). Of the ground-water samples collected in the three CAZB basins, 9 percent exceeded the proposed uranium MCL, 91 percent exceeded the proposed radon MCL, and 1 percent exceeded the proposed radon AMCL. In the NAWQA samples collected nationwide, 4 percent exceeded the proposed uranium MCL, 61 percent exceeded the proposed radon MCL, and 4 percent exceeded the proposed radon AMCL (Dennis Wentz and others, U.S. Geological Survey, written commun., 1999). The Study Units with the highest radon concentrations were in the Colorado Rockies and the Eastern United States. Median concentrations of radon and uranium in the CAZB Study Unit were higher than median concentrations for the United States (figure at right). |
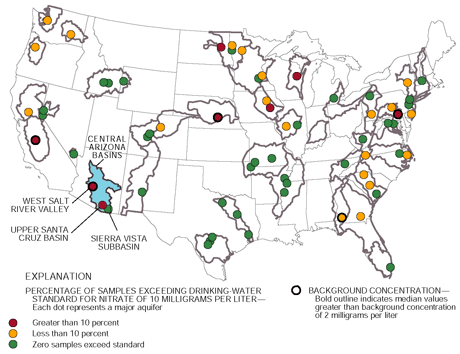
The study of water quality in deep aquifers
that provide drinking water in the West Salt River Valley is one
of eight NAWQA ground-water studies nationwide that had more than
10 percent of samples that exceeded the USEPA MCL for nitrate of
10 mg/L (figure at right). Of 35 samples collected basinwide in
the West Salt River Valley, 34 percent had concentrations greater
than the USEPA MCL. Seventy-eight percent of shallow ground-water
samples from the agricultural land-use study in the West Salt River
Valley had concentrations greater than the USEPA MCL for nitrate.
Only 10 percent of samples from the Upper Santa Cruz Basin and none
of the samples from the Sierra Vista subbasin had concentrations
that exceeded the USEPA MCL for nitrate. |
The highest concentrations of nitrate and dissolved solids were in shallow ground water beneath an irrigated agricultural area. Shallow ground water from the agricultural land-use study area in the West Salt River Valley had median concentrations of nitrate (19 mg/L) and dissolved solids (3,050 mg/L) that exceeded the USEPA MCL and SMCL, respectively (table 1). Nitrate and dissolved solids from irrigation and agricultural practices are accumulating in shallow ground water (see p. 9 and 11). The shallow ground water in this area is not used for drinking water, and clay beds reduce the likelihood of contamination of the aquifers below that are used for drinking water (see p. 22).
Deeper ground water from urban, rangeland, and agricultural areas in other parts of the West Salt River Valley had a median nitrate concentration that was less than the MCL of 10 mg/L; however, the median concentration of dissolved solids exceeded the SMCL of 500 mg/L (table 1). Median concentrations of nitrate from the Upper Santa Cruz Basin and the Sierra Vista subbasin also were less than the MCL, and median concentrations of dissolved solids were less than the SMCL (table 1).
 |
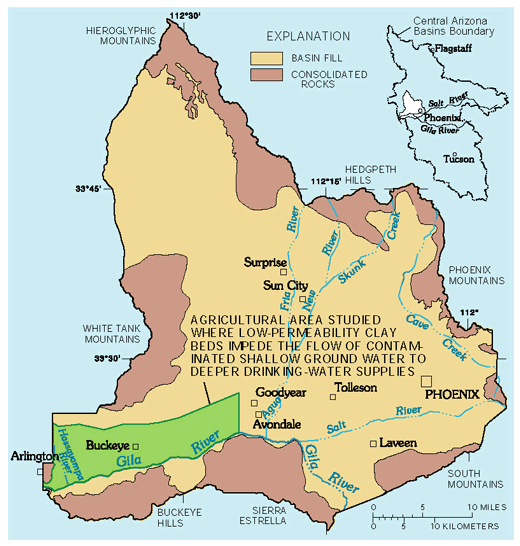
Clay beds that currently protect deep ground water from contamination may not do so in the future. In the agricultural land-use study area of the West Salt River Valley (fig. 20), the tops of low-permeability clay beds are about 150 to 400 ft below the land surface. These clay beds impede the downward movement of irrigation seepage and reduce the likelihood of contaminants reaching deeper drinking-water supplies. Domestic wells in the area yield water from beneath the protective clay beds. Ground water above the clay beds has higher nitrate and dissolved-solids concentrations than ground water from beneath the clay beds (fig. 21). In this area, ground-water samples from above the clay beds had a median dissolved-solids concentration of 3,050 mg/L and a median nitrate concentration of 19.0 mg/L (table 1). Ground-water samples from below the clay beds had a median dissolved-solids concentration of 702 mg/L and a median nitrate concentration of 1.9 mg/L. Care must be taken in drilling and completing drinking-water wells below the clay beds to ensure that shallow ground water above the clay beds does not contaminate the well and aquifer below.
Analyses of the tritium from ground water in this area indicated that water above the clay beds generally had been recharged after 1953, and water below the clay beds generally had been recharged before 1953 (see information about age dating ground water on p. 18). Although the clay beds currently reduce the likelihood that irrigation seepage will contaminate the ground water below, future large-scale withdrawals of ground water from below the clay beds could possibly result in the movement of shallow, poor quality water through the clay beds and into the domestic ground-water supply.
|
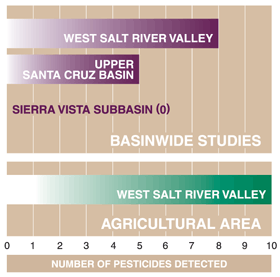 |
| Figure 22. The largest number of pesticides was detected in an agricultural area in the West Salt River Valley. |
Occurrence and distribution of pesticides in ground water in the CAZB reflect both agricultural and urban land uses. Ten pesticides were detected in shallow ground water from the agricultural land-use study area in the West Salt River Valley, west of Phoenix (fig. 22). In other parts of the West Salt River Valley, consisting of agricultural, urban, and rangeland areas, eight pesticides were detected in ground water. Five pesticides were detected in ground water from the Upper Santa Cruz Basin, where there is a mixture of land-use types, but 60 percent of the basin is undeveloped rangeland (Coes and others, 2000). In the Sierra Vista subbasin, where urban and agricultural land uses are minimal (3.3 percent of basin; Coes and others, 1999) and have been minimal in the past, no pesticides were detected in ground-water samples. During 199698, the largest quantities of pesticides used among the three basins were for agriculture in the West Salt River Valley (Ken Agnew, University of Arizona, Pesticide Information and Training Office, written commun., 1999).
Most of the pesticides detected in ground water in the CAZB were herbicides used to control unwanted plants in urban and agricultural areas (fig. 23). Herbicide use in urban areas is indicated by detections of simazine and prometon in the West Salt River Valley and prometon and 2,4-D in the Upper Santa Cruz Basin. These herbicides are used primarily in nonagricultural areas (U.S. Geological Survey, 1999). Detections of atrazine and deethylatrazine (a breakdown product of atrazine) in the West Salt River Valley and the Upper Santa Cruz Basin are an indication that herbicides used in areas of present and historical agriculture are affecting ground-water quality. Atrazine is one of the most heavily used herbicides in agricultural areas in the United States (U.S. Geological Survey, 1999).
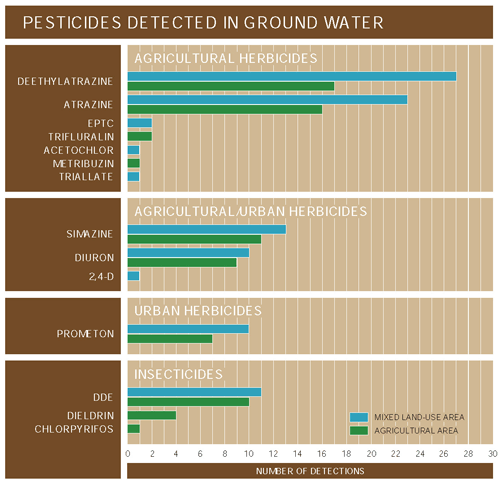 |
| Figure 23. Most of the pesticides detected in ground water in the CAZB were herbicides used in agricultural and urban environments. |
Concentrations of pesticides in ground water did not exceed drinking-water standards or guidelines. Although deethylatrazine, simazine, prometon, DDE, atrazine, and diuron were detected in more than 30 percent of the ground-water samples from the agricultural land-use study area of the West Salt River Valley, none of the concentrations exceeded drinking-water standards or guidelines. Similarly, pesticides detected in ground water from the basinwide sampling in the West Salt River Valley during 199698 did not exceed drinking-water standards or guidelines.
DDE was detected in 10 (56 percent) of the shallow ground-water samples from the agricultural land-use study area in the West Salt River Valley. Detections of DDE in this area are the result of the persistence of this insecticide breakdown product in the environment and the physical characteristics of the ground-water system in the area. In particular, the shallow depth to ground water in the agricultural land-use study area means that irrigation seepage and recharge, containing pesticides and their breakdown products, do not have to travel far to contaminate the ground water. Clay layers impede the movement of pesticides into the deeper aquifers in the area. The soils in the agricultural area have been identified as a source of DDE for the ground water (Brown, 1993). The only detection of DDE in the West Salt River Valley outside of the agricultural area was in a sample from the northern part of the Phoenix metropolitan area. DDE was not detected in samples from the Upper Santa Cruz Basin or the Sierra Vista subbasin.The large depths to ground water and small amounts of DDT used in most of the West Salt River Valley, the Upper Santa Cruz Basin, and the Sierra Vista subbasin limit the potential for introduction of DDE to the ground water.
|
Pesticides and Human Health Laboratory studies, mostly on animals, have shown that pesticides can cause health problems such as birth defects, nerve damage, cancer, and disruption of the endocrine system (USEPA, rev. June 12, 2000). The health effects on humans are not adequately understood, particularly when estimating the risks of exposure to mixtures of pesticides in water (U.S. Geological Survey, 1999). The USEPA determines risk on the basis of toxicity and exposure to a single pesticide (U.S. Environmental Protection Agency, rev. November 17, 1999), whereas NAWQA studies in the CAZB (see below) and nationwide have shown that most contamination in water occurs as pesticide mixtures (U.S. Geological Survey, 1999). The effects of exposure to low concentrations of pesticide mixtures in drinking water is not known and will require further study to determine if standards or guidelines can be developed. |
Detections of multiple pesticides indicate the complexity of contamination from land-surface activities. No standards or guidelines currently exist for mixtures of pesticides in drinking water because their effect on human health is not known (U.S. Geological Survey, 1999). All 9 wells in the agricultural land-use study area had 3 or more pesticides detected, whereas only 3 of the 35 wells sampled basinwide in the West Salt River Valley had 3 or more pesticides detected, and none of the wells in the Upper Santa Cruz Basin had 3 or more pesticides detected (fig. 24). No pesticides were detected in the Sierra Vista subbasin.
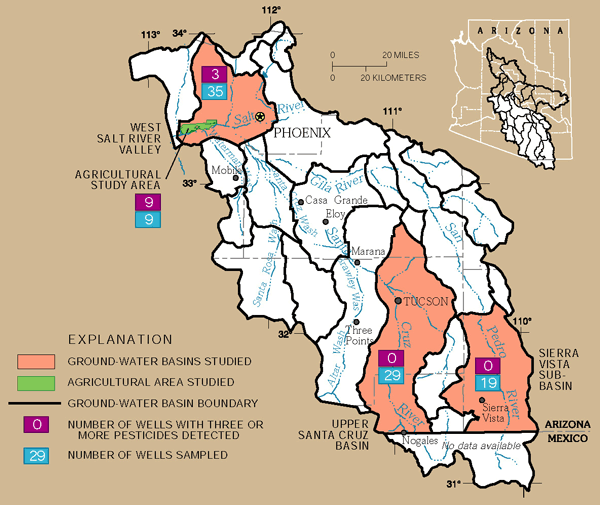 |
| Figure 24. Multiple pesticides were detected in all nine monitoring wells in the agricultural land-use study area of the West Salt River Valley. |
Volatile organic compounds (VOCs), including gasoline compounds, solvents, and refrigerants, have been identified as a major concern for ground-water contamination in Arizona (Marsh, 1994). Leaking underground storage tanks and disposal of solvents have been linked to most of the documented cases of ground-water contamination by VOCs. Electronic- and aerospace-manufacturing facilities use solvents for degreasing and are known to be sources of some of the largest VOC contamination problems in Arizona. Disposal of solvents from these types of facilities has occurred since the 1950s (Marsh, 1994). Dry-cleaning facilities also have been identified as sources of recent ground-water contamination by VOCs. Some municipal supply wells in the urban areas of Phoenix and Tucson are no longer used because of contamination by VOCs (Marsh, 1994).
VOCs were detected in ground water from all three basins sampled during 199698 (fig. 25). Of the 96 samples collected, 33 (34 percent) contained trichloromethane, 24 (24 percent) contained 1,2,4-trimethylbenzene, and 20 (21 percent) contained tetrachloroethene (otherwise known as perchloroethylene, PCE, a solvent commonly used in dry cleaning). Only two VOC detections exceeded drinking-water regulationsPCE (5.48 µg/L) in the Upper Santa Cruz Basin and 1,2-dibromoethane (0.080 µg/L) in shallow ground water in the agricultural area of the West Salt River Valley.
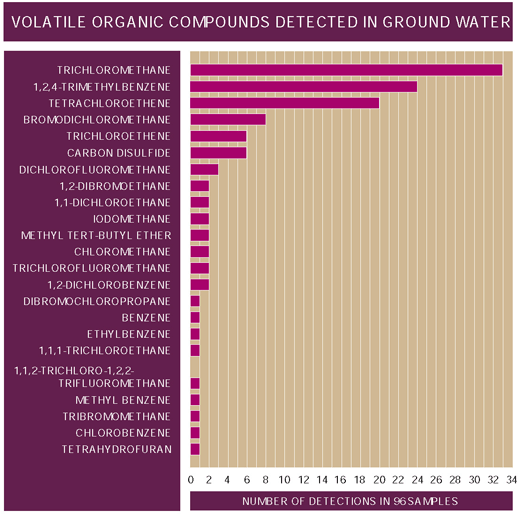 |
| Figure 25. The VOCs trichloromethane, 1,2,4-trimethybenzene, and tetrachloroethene were detected most frequently in ground water. |
Shallow ground water from the nine wells in the agricultural land-use study area had the largest number of VOC detections (35). Ground water from the other 35 wells in the West Salt River Valley had 32 detections. The Upper Santa Cruz Basin (18) and the Sierra Vista subbasin (13) had fewer detections. The larger area of urban land use in the West Salt River Valley appears to be the reason for the greater number of detections there than in the other basins sampled.
Three wells that had five or more VOCs detected in ground water were located in the metropolitan area of Phoenix in the West Salt River Valley. The VOCs detected in these wells were either refrigerants, solvents and chemicals used to make solvents, or gasoline additives. These detections are typical of detections found in small-capacity wells in the metropolitan Phoenix area (Marsh, 1994). Combinations of solvents and gasoline additives are often detected in ground water because their use is widespread, not necessarily because they are from the same source (Squillace and others, 1999).
Detections of VOCs in ground water in the relatively undeveloped Sierra Vista subbasin indicate that ground water in localized areas of the subbasin may be affected by human activities. These detections are not widespread; therefore, the effects of human activity on present-day ground-water quality are not considered significant for the entire subbasin. These detections are an early warning of what could occur in the future in a basin that is presently considered minimally affected by urban activities.
|
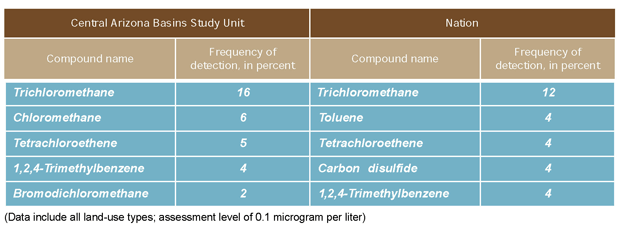
Trichloromethane (chloroform), tetrachloroethene
(PCE), and 1,2,4-trimethylbenzene were three of the five most commonly
detected VOCs in the Nation and in the CAZB when concentrations
above an assessment level of 0.1 µg/L were considered. The
national data collected by the NAWQA Program during 1996-99 represent
ambient ground water for all land-use types.Trichloromethane and
PCE have been shown to cause cancer in laboratory animals from long-term
exposure at concentrations greater than USEPA MCLS.
|
| Table of Contents || Previous Section || Next Section || Glossary U.S. Geological Survey Circular 1213 Suggested citation:
|