Data Series 862
1U.S. Geological Survey, St. Petersburg, Fla.
2University of South Florida, Department of Geology, Tampa, Fla.
3U.S. Geological Survey, Woods Hole, Mass.
4University of South Florida, College of Marine Science, St. Petersburg, Fla.
U.S. Department of the
Interior
U.S.
Geological Survey
St.
Petersburg Coastal and Marine Science Center
Publications are available from USGS Information Services, Box 25286, Federal Center, Denver, CO 80225-0046 (telephone 1-888-ASK-USGS; e-mail: infoservices@usgs.gov).
Page Contents: |
|
This database, identified as U.S. Geological Survey Data Series 862, has been approved for release and publication by the U.S. Geological Survey (USGS). Although this database has been subjected to rigorous review and is substantially complete, the USGS reserves the right to revise the data pursuant to further analysis and review. Furthermore, it is released on condition that neither the USGS nor the U.S. Government may be held liable for any damages resulting from its authorized or unauthorized use.
This disc is readable on any computing platform that has standard CD driver software installed. The minimum software requirements are a Web browser, a portable document format (PDF) reader, and a text editor. If you cannot fully access the information on this page, please contact USGS Information Services at infoservices@usgs.gov or 1-888-ASK-USGS.
Carbon dioxide (CO2) in the atmosphere is absorbed at the surface of the ocean by reacting with seawater to form a weak, naturally occurring acid called carbonic acid. As atmospheric carbon dioxide increases, the concentration of carbonic acid in seawater also increases, causing a decrease in ocean pH and carbonate mineral saturation states, a process known as ocean acidification. The oceans have absorbed approximately 525 billion tons of carbon dioxide from the atmosphere, or about one-quarter to one-third of the anthropogenic carbon emissions released since the beginning of the Industrial Revolution (Sabine and others, 2004). Global surveys of ocean chemistry have revealed that seawater pH has decreased by about 0.1 units (from a pH of 8.2 to 8.1) since the 1700s due to the absorption of carbon dioxide (Caldeira and Wickett, 2003; Orr and others, 2005; Raven and others, 2005). Modeling studies, based on Intergovernmental Panel on Climate Change (IPCC) CO2 emission scenarios, predict that atmospheric carbon dioxide levels could reach more than 500 parts per million (ppm) by the middle of this century and 800 ppm by the year 2100, causing an additional decrease in surface water pH of 0.3 pH units. Ocean acidification is a global threat and is already having profound and deleterious effects on the geology, biology, chemistry, and socioeconomic resources of coastal and marine habitats (Raven and others, 2005; Ruttiman, 2006). The polar and sub-polar seas have been identified as the bellwethers for global ocean acidification.
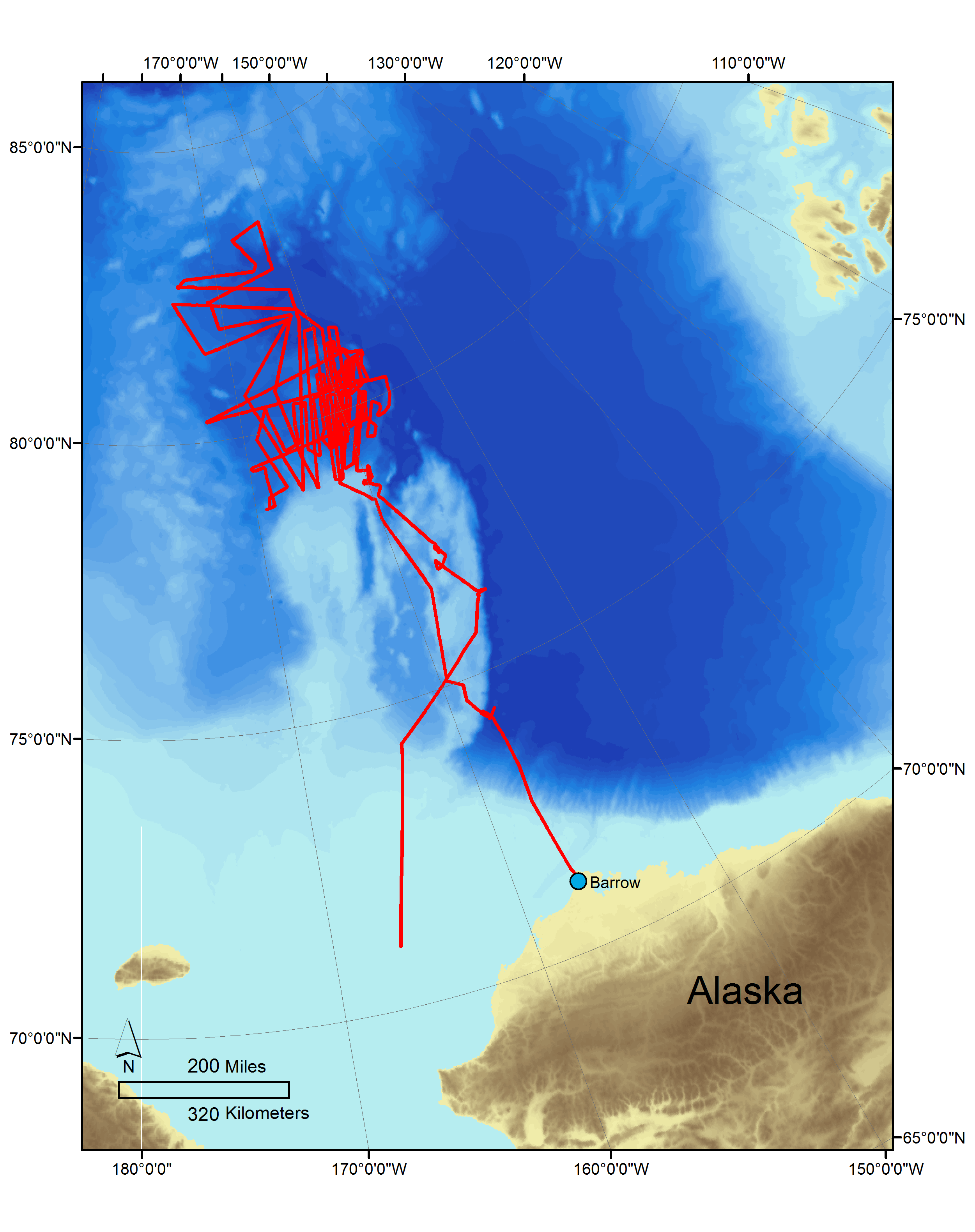
Arctic Ocean
The Canada Basin in the Arctic Ocean (fig. 1) (1,524,664 km2 ) has a fairly constant temperature of 0 degrees Celsius (°C), and supports some of the most productive marine areas in the world. Its cold waters absorb carbon dioxide more rapidly than warmer seawater. An increase in temperature of approximately 1.8 degrees Fahrenheit (°F), over the past 150 years, has increased the melting of Arctic ice. Until recently, the perennial ice cover has prohibited significant equilibration of CO2 with the atmosphere, creating a polar mixed layer that has lower partial pressure of CO2 (pCO2) levels than those found in the atmosphere. Over the last three decades, the retreat of summertime sea ice cover has exposed shelf waters to the atmosphere and allowed additional absorption of atmospheric CO2. The combination of these processes accelerates the rate at which pH and carbonate mineral saturation state decrease (Bates and others, 2006). Collecting waters in the Canadian and Makarov Basins have been the focus of the ocean acidification team during recent Healy cruises (2010, 2011, and 2012). Recent data from 2010 and 2011 cruises (Robbins and others, 2013b and Robbins and others, 2013c) on the Healy have shown that 20% of the Canada Basin in the Arctic is undersaturated with respect to aragonite (Robbins and others, 2013a ; also see Olaffson and others, 2009; Yamamoto-Kawai and others, 2009; Chierici, and Fransson, 2009) when ice melt is at its largest extent. These data corroborate models that project that the entire Arctic Ocean will become undersaturated with respect to carbonate minerals in the next decade (Orr and others, 2005; Steinacher and others 2009a,b; Popova and others, 2014).
The Arctic Ocean is a key area in which expanded time series studies of coastal and open ocean seawater chemistry will provide critical information to scientists and decision-makers for monitoring the progress of ocean acidification and place it in context with other global climate change studies (Robbins and others, 2010; Robbins and others, 2013c, AMAP, 2013).
From August 25 to September 27, 2012, the United States Coast Guard Cutter (USCGC) Healy was part of an Extended Continental Shelf Project to determine the limits of the extended continental shelf in the Arctic. On a non-interference basis, a USGS ocean acidification team participated in the cruise to collect baseline water data in the Arctic. The collection of data extended from coastal waters near Barrow, Alaska, to 83°32'N., -175°36'W., and southward back to coastal waters near Barrow and on to Dutch Harbor, Alaska. As a consequence, a number of hypotheses were tested and questions asked associated with ocean acidification, including:
During the cruise, underway continuous and discrete water samples were collected, and discrete water samples were collected at stations to document the carbonate chemistry of the Arctic waters and quantify the saturation state of seawater with respect to calcium carbonate. These data are critical for providing baseline information in areas where no data have existed prior and will also be used to test existing models and predict future trends.
During the cruise, Healy’s instrumentation continuously recorded temperature, salinity, dissolved oxygen, fluorescence, and pCO2 of surface water. Collaborating with the University of South Florida (USF), the U.S. Geological Survey also used a flow-through Multiparameter Inorganic Carbon Analyzer (MICA) (Liu and others, 2006, Wang and others, 2007). The MICA collected continuous measurements every 2 minutes (min) of pCO2, pH, and total carbon (TCO2) in the seawater. Because of a computer failure, the MICA was operable only 3 days of this cruise. Discrete water samples were collected for ship-board measurement of pH and total alkalinity (TA). Water samples were also collected for TA, TCO2, nutrient, isotope, and elemental analyses to be analyzed at a land-side laboratory. Temperature, salinity, dissolved oxygen, and fluorescence were collected during vertical casts of a Niskin rosette. Discrete depth-profile water samples for TA, TCO2, nutrient, isotopes, and elemental analyses were taken during the Niskin casts and analyzed land-side. Additionally, waters from the Niskin casts were filtered onboard to obtain micro-organismal and microbiological community data, plus particulate organic carbon and particulate inorganic carbon data, which are not reported here.
Suggested Citation: Robbins, L.L., Wynn, Jonathan, Knorr, P.O., Onac, Bogdan., Lisle, John, McMullen, Kate, Yates, K.K., Byrne, R.H., and Liu, Xuewu, 2014, USGS Arctic Ocean carbon cruise 2012: Field activity L-01-12-AR to collect carbon data in the Arctic Ocean, August-September 2012: U.S. Geological Survey Data Series 862, 1 CD, https://dx.doi.org/10.3133/ds862.
Bellerby, Richard., Browman, H.I., Sumaila, U.R., coordinating authors, 2013, AMAP assessment 2013: Arctic Ocean acidification: Oslo, Norway, Arctic Monitoring and Assessment Programme (AMAP), 99 pp., accessed October 9, 2014, available at http://www.amap.no/documents/doc/AMAP-Assessment-2013-Arctic-Ocean-Acidification/881.
Assayag, Nelly, Rivé, Karine, Ader, Magali, Jézéquel, Didier, and Agrinier, Pierre, 2006, Improved method for isotopic and quantitative analysis of dissolved inorganic carbon in natural water samples: Rapid Communications in Mass Spectrometry, v. 20, no. 15, p. 2243-2251, accessed October 9, 2014, available at https://dx.doi.org/10.1002/rcm.2585.
Bates, N.R., Moran, S.B., Hansell, D.A., and Mathis, J.T., 2006, An increasing
CO2 sink in the
Caldeira, Ken, and Wickett, M.E., 2003, Oceanography-Anthropogenic carbon and ocean pH: Nature, v. 425, no. 6956, p. 365, accessed October 9, 2014, available at https://dx.doi.org/10.1038/425365a.
Chayes, Dale, Roberts, Steve, and Bolmer, Tom, 2010, HLY 1002 data description summary: Seattle, Washington, United States Coast Guard (USCG), Science Operations, 74 p., accessed October 9, 2014, available at http://www.icefloe.net/files/HLY1002_Data_Synopsis.pdf 8.03MB. [Report is also included.]
Chierici, M. and Fransson, A., 2009, Calcium carbonate saturation in the surface water of the Arctic Ocean: undersaturation in freshwater influenced shelves, Biogeosciences, v. 6, no. 11, 2421-2431., accessed October 9, 2014, available at https://dx.doi.org/10.5194/bg-6-2421-2009.].
Dickson, A.G., 1990, Standard potential of the reaction AgCl(s) + .5H2(g) = Ag(s) + HCl(aq) and the standard acidity constant of the ion HSO4- in synthetic sea water from 273.15 to 318.15 K: The Journal of Chemical Thermodynamics, v. 22, no. 2, p. 113-127., accessed October 9, 2014, available at https://dx.doi.org/10.1016/0021-9614(90)90074-z.]
Dickson, A.G., Sabine, C.L., and Christian, J.R., eds., 2007, Guide to best practices for ocean CO2 measurements: Sidney, British Columbia, Canada, North Pacific Marine Science Organization, PICES Special Publication no. 3, IOCCP report no.8. variously paged.
Epstein, S., and Mayeda, T., 1953, Variations of O18 content of waters from natural sources: Geochemica et Cosmochimica Acta, v. 4, no. 5, p. 213-224. [Also available at https://dx.doi.org/10.1016/0016-7037(53)90051-9.
Gattuso, J.-P., Hansson, Lina, and the EPOCA Consortium, 2009, European Project on Ocean Acidification (EPOCA)—Objectives, products, and scientific highlights, with a section on Modeling ocean acidification in the Arctic Ocean (box 5), by Steinacher, Marco, Joos, Fortunat, and Frölicher, T.L.: Oceanography, v. 22, no. 4, p. 198–199, accessed October 9, 2014, available at https://dx.doi.org/10.5670/oceanog.2009.108.
Ho, D.T., Law, C.S., Smith, M.J., Schlosser, Peter, Harvey, Mike, and Hill, Peter, 2006, Measurements of air-sea gas exchange at high wind speeds in the Southern Ocean: Implications for global parameterizations: Geophysical Research Letters, v. 33, no. 16, 6 p., accessed October 9, 2014, available at https://dx.doi.org/10.1029/2006GL026817].
Liu, Xuewu, Patsavas, M.C., and Byrne, R.H., 2011, Purification and characterization of meta-cresol purple for spectrophotometric seawater pH measurements: Environmental Science and Technology, v. 45, no. 11, p. 4862–4868, accessed October 9, 2014, available at https://dx.doi.org/10.1021/es200665d.
Liu, Xuewu, Wang, Z.A. Byrne, R.H. Kaltenbacher, E.A. and Bernstein, R.E., 2006, Spectrophotometric measurements of pH in-situ: Laboratory and field evaluations of instrumental performance: Environmental Science and Technology, v. 40, no. 16, p. 5036-5044, accessed October 9, 2014, available at https://dx.doi.org/10.1021/es0601843.].
Lueker, T.J., Dickson, A.G., and Keeling, C.D., 2000, Ocean pCO2 calculated from dissolved inorganic carbon, alkalinity, and equations from K1 and K2 - Validation based on laboratory measurements of CO2 in gas and seawater at equilibrium: Marine Chemistry, v. 70, nos. 1-3, p. 105-119, accessed October 9, 2014, available at https://dx.doi.org/10.1016/s0304-4203(00)00022-0.].
Olafsson, J., Olafsdottir, S.R., Benoit-Cattin, A., Danielsen, M., Arnarson, T.S., and Takahashi, T., 2009, Rate of
Orr, J.C., Fabry, V.J., Aumont, O., Bopp, L., Doney, S.C., Feely, R.A., Gnanadesikan, A., Gruber, N., Ishida, A., Joos, and others, , 2005, Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms: Nature, v. 437, no. 7059, p. 681–686, accessed October 9, 2014, available at https://dx.doi.org/10.1038/nature04095
Popova, E.E., Yool,A., Aksenov, Y., Coward, A.C., and Anderson, T. R., 2014, Regional variability of acidification in the Arctic: a sea of contrasts, Biogeosciences, v. 11, no. 2, p. 293-308., accessed October 9, 2014, available at https://dx.doi.org/10.5194/bg-11-293-2014.]
Prosser, S.J., and Scrimgeour, C.M., 1995, High-precision determination of 2H/1H in H2 and H2O by continuous-flow isotope ratio mass spectrometry: Analytical Chemistry, v. 67, p. 1992-1997.
Raven, J.A., and others, 2005, Ocean acidification due to increasing atmospheric carbon dioxide: London, UK, The Royal Society, 60 p., accessed May 20, 2014, at http://royalsociety.org/policy/publications/2005/ocean-acidification/.
Robbins, L.L., Yates, K.K., Feely, Richard., and Fabry, Victoria., 2010, Monitoring and assessment of ocean acidification in the Arctic Ocean: A scoping paper: U.S. Geological Survey Open-File Report 2010-1227, 4 p., accessed May 20, 2014 at https://pubs.usgs.gov/of/2010/1227/.
Robbins L.L., Wynn J.G., Lisle J.T., Yates K.K., Knorr P.O., Byrne, R.H., Liu, Xuewu, Patsavas, M.C., Azetsu-Scott, Kumiko, and Takahashi, Taro., 2013a, Baseline monitoring of the western Arctic Ocean estimates 20% of Canadian Basin surface waters are undersaturated with respect to aragonite: PLoS ONE v. 8, no. 9, : e73796, 15p., accessed October 9, 2014, available at https://dx.doi.org/10.1371/journal.pone.0073796.]
Robbins, L.L., Yates, K.K., Gove, M.D., Knorr, P.O., Wynn, Jonathan, Byrne, R.H., and Liu, Xuewu, 2013b, USGS Arctic Ocean carbon cruise 2010: Field activity H-03-10-AR to collect carbon data in the Arctic Ocean, August - September 2010: U.S. Geological Survey Data Series 741, 1 CD. [Also available at https://pubs.usgs.gov/ds/741.]
Robbins, L.L., Yates, K.K., Knorr, P.O., Wynn, Jonathan, Lisle, John, Buczkowski, Brian, Moore, Barbara, Mayer, Larry, Armstrong, Andrew, Byrne, R.H., and Liu, Xuewu, 2013c, USGS Arctic Ocean carbon cruise 2011: Field activity H-01-11-AR to collect carbon data in the Arctic Ocean, August - September 2011: U.S. Geological Survey Data Series 748, 1 CD. [Also available at https://pubs.usgs.gov/ds/748.]
Ruttiman, J., 2006, Oceanography-Sick seas: Nature, v. 442, no. 7106, p. 978–980, accessed October 9, 2014, available at https://dx.doi.org/10.1038/442978a.].
Sabine, C.L., Feely, R.A., Gruber, N., Key, R.m., Lee, Kitack., Bullister, J.L., Wanninkhof, Rik., Wong, C.S., Wallace, D.W.R., Tillbrook, Bronte., Millero, F.J., Peng, T.-H., Kozyr, Alexander, Ono, Tsueno, and Rios, A.F., 2004, The oceanic sink for anthropogenic CO2: Science, v. 305, no. 5682, p. 367-371., accessed October 9, 2014, available at https://dx.doi.org/10.1126/science.1097403.]
Steinacher, M., Joos, F., Frölicher, T.L., Plattner, G.-K., and Doney, S.C.,
2009b, Imminent ocean acidification in the
Wang, A.W., Liu, X., Byrne, R.H., Wanninkhof, R., Bernstein, R.E., Kaltenbacher, E.A., and Patten, J., 2007, Simultaneous spectrophotometric flow-through measurements of pH, carbon dioxide fugacity, and total inorganic carbon in seawater: Analytica Chimica Acta, v. 596, no. 1, p. 23-26., accessed October 9, 2014, available at https://dx.doi.org/10.1016/j.aca.2007.05.048.]
Yamamoto-Kawai, Michiyo., McLaughlin, F.A., Carmack, E.C., Nishino, Shigeto,
and Shimada, Koji, 2009, Aragonite undersaturation in the
Yao, Wenshang, and Byrne, R.H., 1998, Simplified seawater alkalinity analysis: Use of linear array spectrometers: Deep Sea Research Part I - Oceanographic Research Papers,, v. 45, no. 8, p. 1383-1392, accessed October 9, 2014, available at https://dx.doi.org/10.1016/S0967-0637(98)00018-1.]