Thiaminase I activity is high in grass and silver carp, but negligible in bighead and black carp
Links
- More information: Publisher Index Page (via DOI)
- Document: XML
- Open Access Version: USGS Accepted Manuscript
- Download citation as: RIS | Dublin Core
Introduction
Produced by certain plants, fungi, and bacteria, thiamine (vitamin B1) is a dietary requirement for all multicellular animals (Tylicki et al., 2018). An organism deficient in thiamine can develop neurological deficits, cardiac dysfunction, and numerous other physiological outcomes in thiamine deficient individuals (Dhir et al., 2019; Harder et al., 2018). Populations of fish and wildlife have experienced declines in abundance and, in certain cases, complete reproductive failures over large geographic areas from thiamine deficiency (Balk et al., 2016; Sutherland et al., 2018). Collapse of lake trout (Salvelinus namaycush) populations in the Great Lakes of North America (Brown et al., 2005b) and loss of Atlantic salmon (Salmo salar) spawning runs in Baltic Sea tributaries in Sweden and Finland (Majaneva et al., 2020) are attributed at least partly to thiamine deficiency. Thiamine deficiency research has been reinvigorated with recent discoveries of thiamine deficiency in populations of Chinook salmon (Oncorhynchus tshawytscha) from the Central Valley of California (Mantua et al., 2021; Mantua et al., 2025) and steelhead trout (O. mykiss) in Oregon (Reed et al., 2023). Thiamine deficiency is now recognized as one of the top emerging issues affecting global biodiversity (Sutherland et al., 2018), and it has been an issue in the Great Lakes for decades.
Lake trout declines in the Great Lakes have been of management concern since the 1950s, and these populations are actively managed to ensure persistence (Hansen and Bronte, 2019; Krueger and Ebener, 2004). Recruitment failures of lake trout have been linked to thiamine deficiency since the 1990s (Brown et al., 2005a; Fisher et al., 1995). Development of thiamine deficiency in lake trout has been connected to consumption of large quantities of prey that have high activity levels of thiaminase (Honeyfield et al., 2005), an enzyme that degrades thiamine in the gut of predators. High thiaminase activity has been documented in invasive species such as alewife (Alosa pseudoharengus) and rainbow smelt (Osmerus mordax) (Tillitt et al., 2005). Thiaminase is produced de novo (Richter et al., 2023) by roughly half of the 400 fishes that have been assessed either for sequence homology or by thiaminase activity assays, representing a wide range of life histories (Rowland et al., 2023). There is a strong correlation between salmonine diets rich in invasive alewife and development of thiamine deficiency (Fitzsimons et al., 2010; Ladago et al., 2020).
This connection between invasive fishes and thiamine deficiency in predators is worth exploring further, especially considering the Great Lakes region is particularly vulnerable to aquatic invasive species introduction and establishment (Escobar et al., 2018). Invasive carp are a concern due to the potential economic and environmental damage if they become established in the Laurentian Great Lakes (Chapman et al., 2021). Invasive carp— consisting of four species including bighead carp (Hypophthalmichthys nobilis), black carp (Mylopharyngodon piceus), grass carp (Ctenopharyngodon idella), and silver carp (H. molitrix) — have established reproductive populations within the Mississippi River basin with grass carp present in some of the Great Lakes, including documented reproduction in the Sandusky and Maumee rivers of western Lake Erie (Chapman et al., 2021).
The presence of invasive carp in diets of fish predators likely varies by season. Small-bodied young-of-year and juvenile fish are small enough for most gape-limited predators, but the rapid growth of carp will make them too large within a year. Anderson et al. (2021) found that combined bighead carp and silver carp comprised roughly 15% of diet samples for shortnose gar (Lepisosteus platostomus), white bass (Morone chrysops), black crappie (Pomoxis nigromaculatus), channel catfish (Ictalurus punctatus), smallmouth bass (Micropterus dolomieu), largemouth bass (Micropterus nigricans), white crappie (Pomoxis annularis), yellow perch (Perca flavescens), flathead catfish (Pylodictis olivaris), freshwater drum (Aplodinotus grunniens), and yellow bass (Morone mississippiensis) in the La Grange reach of the Illinois River, Illinois, during August to September, but were much less commonly in diets in later months. Wolf and Phelps (2017) observed less significant contribution of bighead carp and silver carp to native fish predators’ diets in segments of the Mississippi River sampled in the field and among laboratory experiments. However, laboratory results for the selectivity of bighead carp and silver carp by largemouth bass from Sanft et al. (2018) suggested higher selection for bighead carp than for native prey. Beyond studies listed here, there is little data to assess the potential of Great Lakes piscine predators such as salmonines on consumption of or selection for invasive carp, though some interaction is inevitable. In addition to piscine predators, avian or mammalian predators may also interact with and consume invasive carp depending on opportunity and population size.
Common carp Cyprinus carpio (Greig and Gnaedinger, 1971), crucian carp Carassius carassius (Wistbacka et al., 2009), and silver carp (Wolfe et al., 2023) all have well-documented thiaminase activity. However, the thiaminase activity in grass carp, black carp, and bighead carp is still unknown. Multiple studies have documented large variation in thiaminase activity between species and within a species (Mantua et al., 2025; Tillitt et al., 2005; Wistbacka and Bylund, 2008), but the function and causes of thiaminase upregulation in fishes is still unknown (Rowland et al., 2023). There are some indications that thiaminase activity may increase with body size in field-collected alewife (Tillitt et al., 2005), disease exposure (Wistbacka et al., 2009), diet quality (Lepak et al., 2013), and when field-caught fishes are brought into the laboratory (Lepak et al., 2008; Wistbacka and Bylund, 2008). Testing thiaminase activity in laboratory-raised fishes may help resolve some of this variation because it disentangles some of the environmental and physiological variation. Laboratory fishes have controlled diet and water conditions, as well as few disease and predation stressors. If thiaminase functions to salvage thiamine as it does in bacteria (Jenkins et al., 2007; Sannino et al., 2018), body condition and growth may correlate with thiaminase activity.
We tested the thiaminase activity in four species of invasive carp from cultured fish stocks (Fig 1). Based on the prevalence of thiaminase activity in cypriniform fishes, we hypothesized all four species would have measurable thiaminase activity but were unsure how or if they would differ in their activity levels. We also hypothesized that thiaminase activity might differ among fishes raised in indoor culture compared to outdoor ponds, and size and body condition would relate to thiaminase activity.
Methods
The fish tissue was taken from fish residing in the U.S. Geological Survey Columbia Environmental Research Center (CERC) cultures and had not been used for any other testing. All fish used for tissue collection were originally produced through hormone-induced spawning of wild caught adult fish and raised in the CERC facility until their time of euthanasia. All four species from indoor fish cultures were fed a combination of brine shrimp nauplii and commercially available fish food pellets (Otohime Tokyo, Japan). Bighead carp and silver carp also received supplemental powdered Spirulina and Chlorella algae (Bulkfoods.com, Toledo, Ohio, USA). Fish from indoor cultures were raised in flow-through tanks supplied by aerated well water ranging from 16 °C to 19 °C, depending on seasonality. Indoor fish cultures were fed to satiation twice daily. Two species, grass and silver carp, had outdoor pond cultures in addition to the indoor cultures. Outdoor fish cultures fed on a natural diet from earthen ponds, located at the CERC, with a supplemental feeding of a commercially available fish food (Rangen Aquaculture, Wilbur-Ellis Nutrition, Buhl, Idaho, USA). Fish in the outdoor ponds were exposed to normal seasonal fluctuations in temperature. All bighead, silver, and grass carp were 9 months of age at the time of collection. Black carp used in this study were 20 months of age at time of collection. Individual fish were selected by haphazard sampling from the pool of fish using seine nets in ponds or by dipnet for indoor tanks of CERC fish culture to avoid size or behavioral bias. All fish were of visibly good health and body condition. Fish tissue used for this study was taken from fish residing in the CERC cultures and had not been used for any other testing. All fish used for tissue collection were originally produced through hormone-induced spawning of wild caught adult fish and raised in the CERC facility until their time of euthanasia. Fish were opportunistically analyzed after being euthanized for normal culture reasons (e.g., to control population density as fish grew) under standard operating procedures at CERC. We collected two batches of fish: one in November 2023, and a second batch in April and May 2024.
Thiaminase activity measurements
We selected juvenile fish to represent the most likely size range of consumption by predators. All fishes were euthanized, weighed to the nearest 0.1 g, measured to the nearest mm for both total length (TL) and standard length (SL), and then flash frozen before being placed into a -70 °C freezer until each fish could be homogenized to a whole body fine powder on dry ice. Thiaminase I activity was measured using a 4-NTP assay on homogenated samples following the methods of Hanes et al. (2007) and Kraft et al. (2014). Briefly, homogenate (0.5 g wet weight) was suspended in phosphate buffer (1.25 mL of 100 mM, pH 6.5), bead beat to lyse cells and centrifuged at 4 °C for 10 min at 16,000 x g. A 750 μL aliquot of each supernatant was transferred to 0.8 mL Pierce centrifuge columns containing 30 μm pore size polyethylene filters in new 1.5 mL centrifuge tubes and centrifuged a second time for 5 min at 5,000 x g at 4 °C to remove any particulates. Filtrates were placed on ice and immediately assayed for thiaminase activity using Tris (2-carboxyethyl) phosphine hydrochloride (TCEP, 10 mM, pH 6.9) buffer with 4-NTP (80 µM), both with and without thiamine (400 µM), in 96-well microtiter plates. Wells of the microtiter plate were filled with 97 μL of solution using a multi-channel pipette; three wells with the solution without thiamine and three with solution with additional thiamine. Enzymatic reactions were initiated by the addition of 3 μL of sample supernatant to each well. Readings of absorbance at 411 nm were taken every minute over 60 minutes total at 37 °C in a Synergy HT Multi-Detection Microplate Reader (BioTek Instruments, Winooski, VT). Activity was calculated by linear regression of the change in absorbance over time, corrected for non-thiaminase activity by the difference in slopes (with and without thiamine added), and converted to concentration using the molar extinction coefficient for 4-NTP (13,650 M-1 cm-1). We did not correct thiaminase activity for protein concentrations, so activity measurements are reported as nmol/min/g-sample wet weight.
All assays were performed in triplicate. Solution blanks and positive control samples (Paenibacillus thiaminolyticus supernatant) were run with each plate of samples as a QA/QC check. The positive control (P. thiaminolyticus) is a known thiaminase-producing bacterium isolated from alewife gut (Honeyfield et al., 2002). The bacterium was grown in 50 mL of terrific broth at 37 °C set to shake at 225 revolutions per minute for 24 hours or until optical density was approximately 1. Cultures were centrifuged at 5000 x g for 60 minutes at 4 °C, and then the supernatant was filtered through 10 kD molecular weight cutoff filters (Fisher Scientific, Waltham, MA, USA) rinsed with sterile phosphate-buffered saline (PBS). The original volume was concentrated to 750 µL of P. thiaminolyticus filtered supernatant and was brought up to 1 mL volume with 60% sterile glycerol stock and stored at -20 °C. Across all plates run for this study, the average activity of P. thiaminolyticus supernatant control samples was 480 nmol/g/min (± 26 standard deviation ( stdev)) and the coefficient of variation was 5%.
Statistics
All statistical analyses were completed in R v4.5.1 (R Core Team, 2025). Enzyme activity data are often highly variable due to enzyme production being up- or down-regulated for specific functions. There are often non-detections in the data. We accounted for this using functions specifically designed for datasets that include censored (i.e., below detection limit) data. We assessed differences among species using censored ANOVA fit with the cenanova function in the ‘NADA2’ package (Julian and Helsel, 2024) with a log-link function and 10,000 permutations. This function performs a parametric maximum likelihood test of differences among group means including censored data, followed by a parametric Tukey’s multiple comparison test.
For testing between culture- and pond-sourced fishes, we used the cenpermanova function in the ‘NADA2’ package (Julian and Helsel, 2024). Permutation tests are more robust to small or uneven group sizes, and the cenpermanova function allows for testing differences between two group means that include censored data in a non-parametric way. We tested the two species with both indoor and outdoor/pond cultures, silver and grass carp, for differences in thiaminase activity between pond and indoor culture sources as separate species-specific models. We fit the models separately because there was little overlap in thiaminase activity of grass and silver carp. We ran all cenpermanova models with 10,000 permutations.
We assessed whether fish body condition (K, eq 1) or size (standard length in mm) would relate to thiaminase activity.
Where w = mass (g), SL = standard length (mm), and 100,000 is a conversion factor for metric units.
Because the four invasive carps are from three different genera, we fit separate models for each species to examine effects of body length or body condition on thiaminase activity. We fit Bayesian linear models fit in the ‘rstanarm’ package (Goodrich et al., 2024) exploring how body length or condition affected the ln-transformed thiaminase activity for all values above the limit of quantification. For each model, we ran four chains for 20,000 iterations and discarded the first 5,000 posterior samples as warm-up, leaving 60,000 posterior samples for analysis. We assessed fit by examining traceplots for convergence and using leave-one-out cross-validation using function ‘loo’ in the R package ‘rstan’ (Stan Development Team, 2024). All data are available in a data release (Rowland et al., 2025).
Results
Differences among species
Overall, we assessed 145 individual carp for thiaminase activity (Table 1) including bighead carp from indoor culture (n = 7), black carp from indoor culture (n = 38), grass carp from both indoor culture (n = 37) and outdoor pond culture (n = 13), and silver carp from indoor culture (n = 37) and outdoor pond culture (n = 13). Censored ANOVA indicated that there were statistical differences among species (d.f. = 3, Χ2= 170.8, p < 0.0001; Fig 2). The model estimated the mean thiaminase activity was highest in grass carp (60.1 nmol/g/min), followed by silver carp (14.7 nmol/g/min), bighead carp (2.7 nmol/g/min), and lowest in black carp (2.2 nmol/g/min). Tukey contrasts with lognormal censored data indicated that grass carp had statistically higher thiaminase activity than silver carp (z = 8.626, p < 0.001), bighead carp (z = 8.430, p < 0.001), and black carp (z = 16.613, p < 0.001). Silver carp had statistically higher thiaminase activity than bighead carp (z = 4.207, p < 0.001) and black carp (z = 9.121, p < 0.001). There were no differences between bighead and black carp (z = 0.948, p = 0.768). Notably, no grass carp samples, 8% of silver carp, and 14% of bighead carp samples were below detection limits (Table 1), whereas the majority (~66%) of black carp fell below assay detection limits.
Differences between indoor and outdoor cultures
Censored permutation ANOVAs suggested there were no differences between fish sourced from outdoor rearing ponds or indoor rearing spaces for either grass carp (meanpond = 65.9, meanindoors = 58.8; p = 0.235 to 0.235) or silver carp (meanpond = 14.9, meanindoors = 14.7; p = 0.851 to 0.880).
Relationship between thiaminase activity and fish body metrics
Bayesian linear models indicated that there were no relationships between thiaminase activity and standard length of bighead or grass carp (Fig. 3a,c; Table 2). Black carp and silver carp had statistically negative relationships between standard length and thiaminase activity (Table 2, Fig 3b,d). All 95% credible intervals strongly overlapped zero for the relationship between carp body condition and thiaminase activity (Fig 4, Table 2), suggesting that body condition does not predict thiaminase activity in juvenile bighead carp, black carp, grass carp, and silver carp.
Discussion
Predators consuming large amounts of prey fishes with high thiaminase activity can develop thiamine deficiency complex (Honeyfield et al. 2005). Thiamine deficiency can result in juvenile mortality of up to 90% in Great Lakes salmon (Harder et al., 2020; Ketola et al., 2000). Furthermore, recent work has documented significant decreases in cardiac health of adult salmon with high thiaminase diets (Adeli et al., 2025; Baker et al., 2023). The very first documentation that predator diets rich in carp can cause thiamine deficiency was in the 1930s with common carp fed to farm-raised silver foxes (Vulpes vulpes) (Ender and Helgebostad, 1939). In the intervening years, thiaminase activity has been documented across multiple species of carp (Rowland et al., 2023; Wistbacka et al., 2009; Wolfe et al., 2023). Our hypothesis that the previously untested invasive carp species including grass carp, bighead carp, and black carp would have thiaminase activity was supported. While bighead carp and black carp had very low thiaminase activity levels, grass carp had very high thiaminase activity and silver carp confirmed previous observations of measurable thiaminase activity (Wolfe et al., 2023). Contrary to our hypotheses, we found no differences in thiaminase activity between indoor and outdoor cultures despite very different environmental conditions and diets and no relationship between thiaminase activity and body condition. Two species (grass and black carp) had a weak but negative relationship between body size and thiaminase activity. The data collected here suggested species-specific differences outweigh environmental conditions (including temperature, diet, and UV radiation differences between indoor and outdoor cultures), length, and condition for explaining variation in thiaminase activity measurements among juvenile invasive carp.
What affects thiaminase activity in carp?
Scientists do not fully know why some fishes produce thiaminase (Rowland et al., 2023), nor why there is such wide variation in activity even among the same species collected at the same location on the same day (Wistbacka et al., 2002). Multiple researchers have noted rapid elevation of thiaminase activity in laboratory studies over short periods of time (Lepak et al., 2008; Wistbacka and Bylund, 2008), presumably as a stress response, although the reasons for this are unresolved. In bacteria, thiaminase is a thiamine salvage pathway (Sannino et al., 2018) but this has not been demonstrated in fishes. There is strong evidence of elevated thiaminase in fish exposed to microbial pathogens (Wistbacka et al., 2009), and seasonal variation has been documented for at least some species (Tillitt et al., 2005). The very high levels of thiaminase activity we measured in grass carp juveniles did not differ between indoor and outdoor cultures, suggesting thiaminase activity was not related to diet, stress, or temperature, similar to previous experimental work on clupeids (Lepak et al., 2013). For black carp and silver carp, we found a slight negative relationship between total length and thiaminase activity, opposite of field observations of a positive correlation between alewife size and thiaminase activity in summer and fall, but not spring (Tillitt et al., 2005). However, limiting our samples to only juveniles within a narrow range of sizes may have limited our ability to detect trends. Additionally, all our study animals had body condition > 1, indicating that they had no food stress. Future research could explore patterns in the field and assess how thiaminase may vary within grass and silver carp across seasons, ages, and body condition/starvation indices. Additionally, although fish can produce thiaminase de novo (Richter et al., 2023), it is possible that the very low thiaminase activity seen in bighead and black carp could represent the microbiota contribution of thiaminase (Honeyfield et al., 2002) compared to grass and silver carp that are producing their own thiaminase. The thiaminase I-producing bacterium P. thiaminolyticus has been observed as a small component of the gut microbiome of alewife in the Great Lakes (Richter et al., 2012), and P. thiaminolyticus and/or other thiaminase I-producing bacteria could be present in other fish as well. The relative contributions of the microbiome vs. de novo production is an open question.
How might thiaminase affect predators?
We found juveniles of a size that gape-limited predators could consume had consistent thiaminase activity. Potential negative effects of consuming thiaminase-producing fishes such as invasive carp are dependent upon both the thiaminase activity at time of consumption, which can vary within a species (Lepak et al., 2013), and the proportion of the diet containing high thiaminase activity prey (Honeyfield et al., 2005). In the Great Lakes, there is strong evidence that alewife thiaminase is linked to lake trout declines (Fitzsimons et al., 2010; Ladago et al., 2020; Riley et al., 2011). Piscivores with diets transitioning to a larger reliance on grass carp or silver carp could be at risk for developing thiamine deficiency complex and the symptoms that follow (Harder et al., 2018).
Comparisons with previous work are complicated by differences between the radiometric assay that directly measures thiamine degradation and allows flexibility in pH (Tillitt et al., 2005; Zajicek et al., 2009) compared to the present study assay that uses 4-NTP as the nucleophile and restricts pH to 6.9 (Hanes et al., 2007). However, a recent advance in thiaminase activity assays found thiamine degradation and 4-NTP assays yielded similar results (Porter and Pinger, 2025), so we will compare with these caveats in mind. If we generously assume the current assay represents optimum conditions for carp thiaminase, the thiaminase activity of grass carp is some of the highest reported in Great Lakes food webs, and the activity could be even higher if the pH optimum of grass carp thiaminase differs significantly from 6.9 as it does for some species (Zajicek et al., 2009). Alewife activity measured using the radiometric assay found 4 nmol/g/min (Tillitt et al., 2005) is enough to induce thiamine deficiency, Wolfe et al. (2023) using the 4-NTP assay found a mean thiaminase activity in adult silver carp viscera of 9 nmol/g/min, which was lower than the mean whole-body thiaminase activity we measured in juveniles of approximately 15 nmol/g/min. Average grass carp thiaminase activity of 60 nmol/g/min in the present study is on par with northern anchovy (Engraulis mordax) thiaminase activity (avg = 48 nmol/g/min) linked to thiamine deficiency in Pacific Chinook salmon (Mantua et al., 2025). However, there is a lot of uncertainty surrounding which predators may feed on invasive carp if they become established (Robinson et al., 2021). Anderson et al. (2021) found bighead carp and silver carp common within the diet of native predatory fish in the Illinois River, though this changed throughout the season. Rapid growth rates of invasive carp may facilitate a shift in diet to other available forage fish species as invasive carp grow to exceed the mouth gape of predatory fish. Regardless, juvenile silver and grass carp have high enough thiaminase activity to cause thiamine deficiency complex if they are a large part of a predator’s diet. Alewife comprising 35% or more of the diet of lake trout was enough to cause thiamine-dependent fry mortality (Honeyfield et al., 2005).
Thiaminase activity has been documented in other invasive forage fish species within the Great Lakes, including alewife and rainbow smelt (Tillitt et al., 2005). Great Lakes salmonines consuming grass and silver carp may be at additional risk for developing thiamine deficiency if they switch to consuming grass or silver carp. The potential for developing thiamine deficiency by consuming thiaminase-containing invasive carp extends beyond predatory fishes to other piscine predators such as waterbirds and mammals. Thiamine deficiency in European common eider (Somateria mollissima) has been linked massive population declines in recent years (Mörner et al., 2017). Waterbirds of the Great Lakes region like double-crested cormorant (Nannopterum auritum), mergansers (Mergus spp.), bald eagles (Haliaeetus leucocephalus), osprey (Pandion haliaetus), or great blue heron (Ardea herodias) are all piscivorous (Hebert and Morrison, 2003) and may be susceptible to thiamine deficiency induced by high thiaminase prey. Bald eagles may also scavenge potentially larger-bodied invasive carp carcasses or directly predate on living carp. Mammalian predators, like the river otter (Lontra canadensis) may also consume carp as part of their diet (Raesly, 2001). The timing of predation and subsequent physiological effects of a high thiaminase diet would depend on prey size and predator- prey behaviors at the time of co-occurrence. Migratory avian predators for example may not co-occur with invasive carp among habitats at the same time of year as prey size facilitates predation.
Dietary thiaminase can affect piscivore health directly. Experimental dietary thiaminase has been associated with reduced cardiac performance (Baker et al., 2023), decreased body condition and lower swimming performance (Houde et al., 2015), and negative effects on growth, survival, and food conversion efficiency (Therrien et al., 2025) in salmonines. High-thiaminase rainbow smelt was correlated with heart irregularities such as increases in ventricular mass, reductions in ventricular fluid content, and changes to the allometry of myocardia (Adeli et al., 2025). It is interesting that some predators seem to be selecting for bighead carp (Anderson et al., 2021), a species with low thiaminase activity. More research is needed to understand which invasive carp are a likely forage base to help us understand whether the very high thiaminase activities observed in grass carp and silver carp are a risk to aquatic and avian predators. Additional data on whether predators may preferentially avoid species with high thiaminase could be useful.
Importantly, enzymes are denatured with heat and suboptimal pHs. Silver carp in previous work had no detectable thiaminase activity when cooked (Wolfe et al., 2023). There is no risk for human consumption of thiaminase positive fishes if the enzyme is denatured. Research suggests carps are nutritious and a sought-after food source elsewhere in the world (Li et al., 2021). The risk of consuming thiaminase positive fishes only applies to raw fish, so harvesting invasive carp for human consumption and species control (Varble and Secchi, 2013) presents no risk to consumers of cooked carp even in species with high thiaminase activity.
Why does this matter within the Great Lakes?
The Laurentian Great Lakes are fragile ecosystems that are vulnerable to aquatic invasive species (Sterner et al., 2017). Losses from commercial and sportfishing due to aquatic invasive species are estimated to cost several hundred million dollars per year (Rothlisberger et al., 2012). Among the most damaging invasive species in the Great Lakes, two thiaminase-positive species, alewife and grass carp, were ranked by Lower et al. (2024) among the top ten species for most significant negative environmental and socio-economic effects. The current study adds an axis for grass carp; their extremely high thiaminase activity has potential to negatively affect predator health and demography. Our data suggest that silver and grass carp consumed in large quantities may put piscivorous fishes, birds, and mammals at risk of developing thiamine deficiency.
Tables
Table 1.
Summary data of the average (avg) and standard deviations (stdev) of length, mass, and body condition for the four species tested for thiaminase activity. All fishes were juveniles < 160 mm total length with body conditions above 1.Table 2.
Linear Bayesian model output including posterior median, 95% credible intervals (CRI), and effective sample size (ESS) for how standard length (in mm) and body condition (K) might predict thiaminase activity (nmol/g/min) in bighead, black, grass, and silver carps.Figures

We tested juvenile Silver Carp Hypophthalmichthys molitrix (top left), Grass Carp Ctenopharyngodon idella (top right), Bighead Carp Hypophthalmichthys nobilis (bottom left), and Black Carp Mylopharyngodon piceus (bottom right) of about 80–100 millimeters total length for thiaminase activity. Photo credit U.S. Geological Survey, Columbia Environmental Research Center..
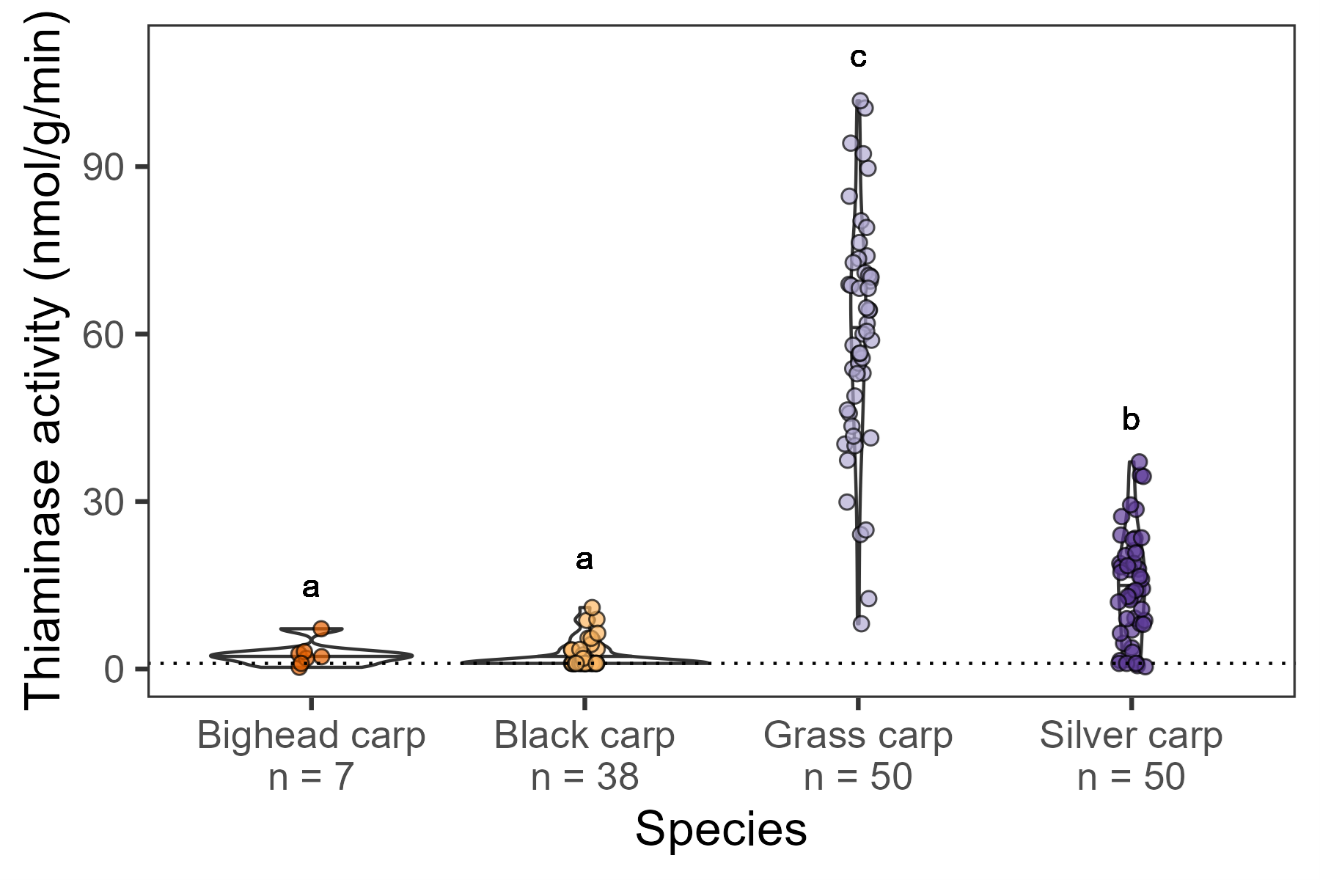
Each thiaminase activity assay result is represented as a dot overplotted on a violin plot to show the spread and density trends of the data. A sample was categorized as below the detection limit if the estimated slope of each assay minus 3x the standard error of the slope was less than 0 (roughly around 1 nmol/g wet mass/min, plotted as a dotted horizontal line). Letters above indicate statistically different results as found using a censored ANOVA to account for non-detects.
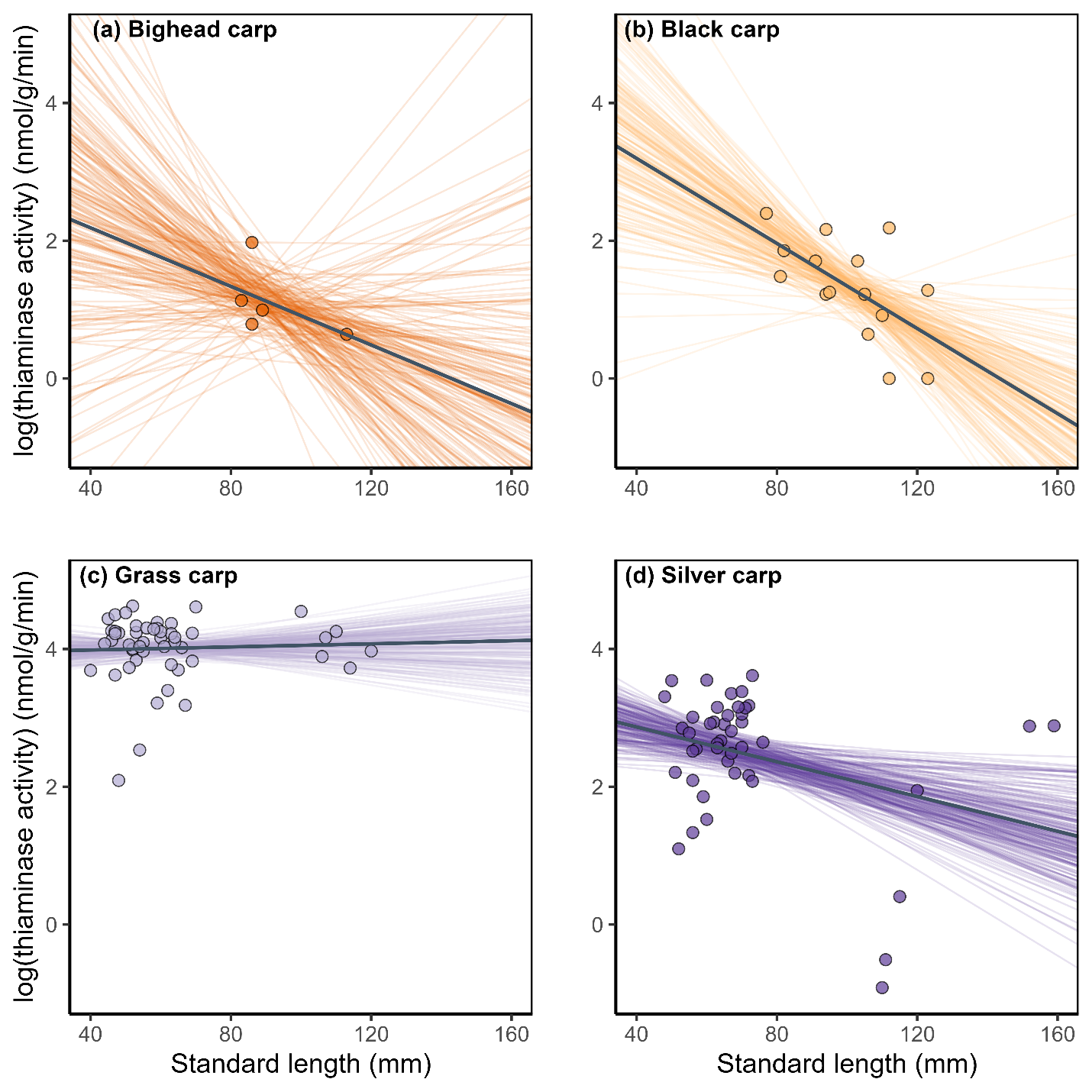
The linear relationship between juvenile carp standard length and thiaminase activity (nmol/g wet mass/min) for the four study species. The plots show 200 draws of the posterior of each model to show the uncertainty around the estimate of the line plus the median (solid line). There were no strong relationships between body length and thiaminase activity, but black carp and silver carp had 95% of the credible interval below zero, suggesting a negative relationship (Table 2). Only thiaminase activity levels above the limit of quantification were included.
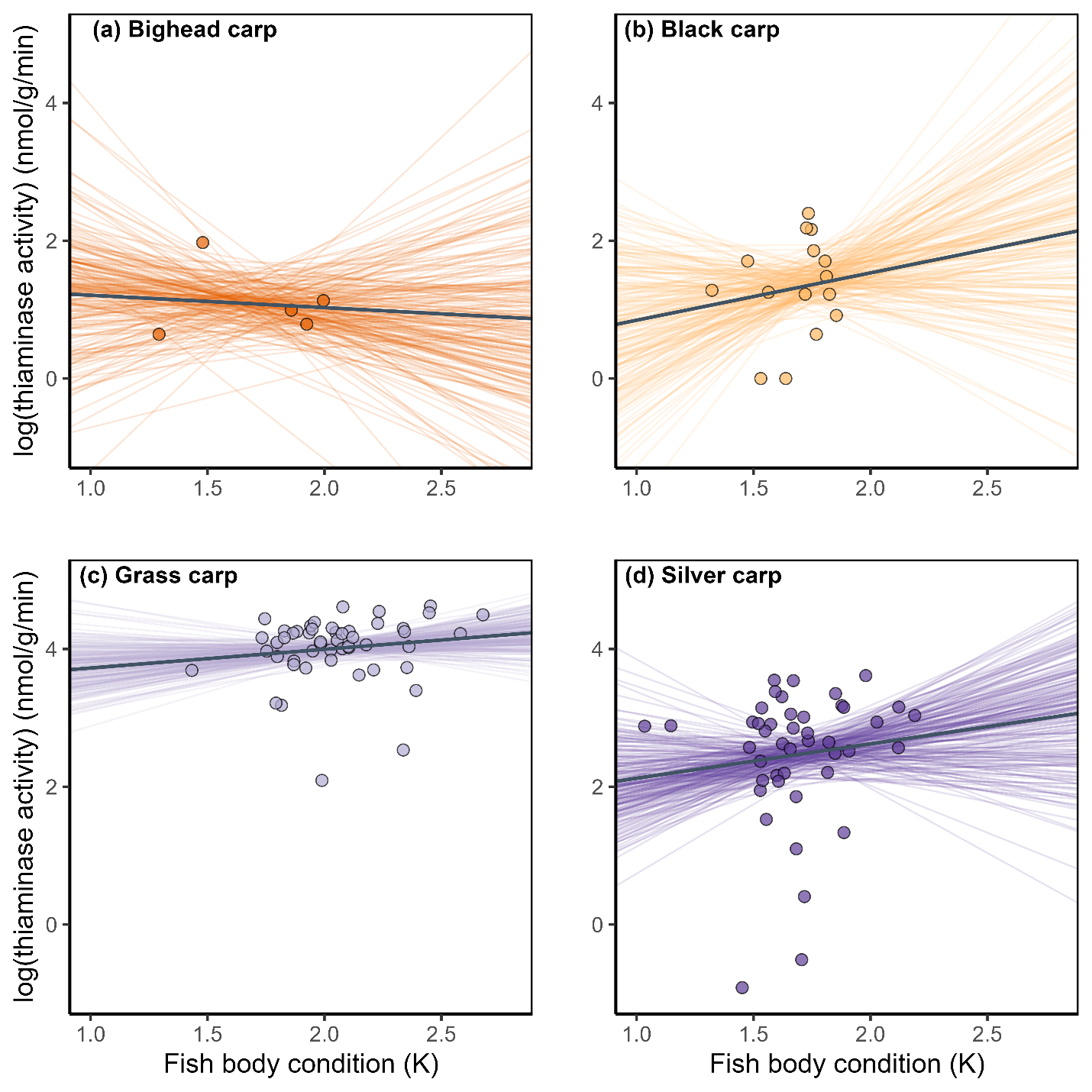
The linear relationship between juvenile carp body condition and thiaminase activity (nmol/g wet mass/min) for the four study species. The plots show 200 draws of the posterior of each model to show the uncertainty around the estimate of the line plus the median (solid line). All slope credible intervals overlapped 0 (Table 2), suggesting no relationship between body condition and thiaminase activity. Only thiaminase activity levels above the limit of quantification were included.
Acknowledgments
We thank the many people at U.S. Geological Survey Columbia Environmental Research Center who maintain the fish cultures. Erinn Scott provided invaluable help with sample processing. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Literature Cited
Anderson, C.A., Anderson, R.L., Wang, J., Gillespie, N., Lampo, E.G., McClelland, N.N., Solomon, L.E., Pendleton, R., Lamer, J.T., 2021. Juvenile silver carp and bighead carp as forage for predatory fish in the LaGrange Reach of the Illinois River. North American Journal of Fisheries Management 43, 164-175.
Balk, L., Hägerroth, P.-Å., Gustavsson, H., Sigg, L., Åkerman, G., Ruiz Muñoz, Y., Honeyfield, D.C., Tjärnlund, U., Oliveira, K., Ström, K., McCormick, S.D., Karlsson, S., Ström, M., van Manen, M., Berg, A.-L., Halldórsson, H.P., Strömquist, J., Collier, T.K., Börjeson, H., Mörner, T., Hansson, T., 2016. Widespread episodic thiamine deficiency in Northern Hemisphere wildlife. Scientific Reports 6, 38821.
Brown, S.B., Brown, L.R., Brown, M., Moore, K., Villella, M., Fitzsimons, J.D., Williston, B., Honeyfield, D.C., Hinterkopf, J.P., Tillitt, D.E., 2005a. Effectiveness of egg immersion in aqueous solutions of thiamine and thiamine analogs for reducing early mortality syndrome. Journal of Aquatic Animal Health 17, 106-112.
Escobar, L.E., Mallez, S., McCartney, M., Lee, C., Zielinski, D.P., Ghosal, R., Bajer, P.G., Wagner, C., Nash, B., Tomamichel, M., Venturelli, P., Mathai, P.P., Kokotovich, A., Escobar-Dodero, J., Phelps, N.B.D., 2018. Aquatic Invasive Species in the Great Lakes Region: An Overview. Reviews in Fisheries Science & Aquaculture 26, 121-138.
Goodrich, B., Gabry, J., Ali, I., Brilleman, S., 2024. rstanarm: Bayesian applied regression modeling via Stan, R package version 2.32.1, https://mc-stan.org/rstanarm.
Greig, R., Gnaedinger, R., 1971. Occurrence of thiaminase in some common aquatic animals of the United States and Canada. US Department of Commerce, National Oceanic and Atmospheric Administration. Special scientific report--fisheries; no. 631 https://repository.library.noaa.gov/view/noaa/9031
Julian, P., Helsel, D., 2024. NADA2: Data Analysis for Censored Environmental Data, R package version 1.1.6, https://CRAN.R-project.org/package=NADA2.
Ladago, B.J., Futia, M.H., Ardren, W.R., Honeyfield, D.C., Kelsey, K.P., Kozel, C.L., Riley, S.C., Rinchard, J., Tillitt, D.E., Zajicek, J.L., Marsden, J.E., 2020. Thiamine concentrations in lake trout and Atlantic salmon eggs during 14 years following the invasion of alewife in Lake Champlain. Journal of Great Lakes Research 46, 1340-1348.
Mantua, N., Johnson, R., Field, J., Lindley, S., Williams, T., Todgham, A., Fangue, N., Jeffres, C., Bell, H., Cocherell, D., 2021. Mechanisms, impacts, and mitigation for thiamine deficiency and early life stage mortality in California's Central Valley Chinook Salmon, in: Commission, N.P.A.F. (Ed.), Technical Report, pp. 92-93. https://doi.org/10.23849/npafctr17/92.93
Mantua, N.J., Bell, H., Todgham, A.E., Daniels, M.E., Rinchard, J., Ludwig, J.M., Field, J.C., Lindley, S.T., Rowland, F.E., Richter, C.A., Walters, D., Finney, B., Distajo, H.A.R., Tillitt, D., Honeyfield, D.C., Lipscomb, T., Kwak, K., Kindopp, J., Cocherell, D., Ward, A., Williams, T.H., Harding, J., Fangue, N.A., Jeffres, C., Ruiz-Cooley, R.I., Litvin, S.Y., Foott, S., Adkison, M., Kormos, B., Harte, P., Colwell, F., Suffridge, C.P., Shannon, K.C., Cranford, A., Ambrose, C., Reed, A., Johnson, R.C., 2025b. Widespread thiamine deficiency in California salmon linked to an anchovy-dominated marine prey base. Proceedings of the National Academy of Sciences 122, e2426011122.
R Core Team, 2025. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria, https://www.R-project.org/.
Richter, Catherine A., Evans, Allison N., Wright-Osment, Maureen K., Zajicek, James L., Heppell, Scott A., Riley, Stephen C., Krueger, Charles C., Tillitt, Donald E., 2012. Paenibacillus thiaminolyticus is not the cause of thiamine deficiency impeding lake trout (Salvelinus namaycush) recruitment in the Great Lakes. Canadian Journal of Fisheries and Aquatic Sciences 69, 1056-1064.
Rowland, F.E., Scott, E.L., Byrd, C.G., Kroboth, P.T., 2025. Thiaminase I activity measurements in juvenile black carp, bighead carp, grass carp, and silver carp from lab cultures, U.S. Geological Survey data release, https://doi.org/10.5066/P14QEDAL.
Stan Development Team, 2024. RStan: the R interface to Stan, R package version 2.32.6, https://mc-stan.org/.
Therrien, C.A., Baker, P.M., Garner, S.R., Wolfe, P.C., Matesic, S., Edwards, K.A., Wilson, C.C., Swanson, H.K., Neff, B.D., 2025. The effect of diet-derived thiaminase on survival, growth, and liver transketolase activity in two strains of lake trout (Salvelinus namaycush). Journal of Great Lakes Research 51, 102689.
Additional Information
CRediT author Statement: Freya Rowland: Conceptualization, Methodology, Investigation, Formal Analysis, Data Curation, Writing – Original Draft, Writing – Reviewing and Editing. Curt Byrd: Conceptualization, Methodology, Investigation, Writing – Reviewing and Editing. Patrick Kroboth: Conceptualization, Methodology, Writing – Reviewing and Editing.
Data Availability Statement: Data are publicly available at https://doi.org/10.5066/P14QEDAL
Authors
Disclaimers
Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Although this information product, for the most part, is in the public domain, it also may contain copyrighted materials as noted in the text. Permission to reproduce copyrighted items must be secured from the copyright owner.
Suggested Citation
Rowland, F.E., Byrd, C.G., and Kroboth, P., 2026, Thiaminase I activity is high in grass and silver carp, but negligible in bighead and black carp: Journal of Great Lakes Research, v. 52, no. 1, 102751, 8 p., https://doi.org/10.1016/j.jglr.2026.102751.
| Publication type | Article |
|---|---|
| Publication Subtype | Journal Article |
| Title | Thiaminase I activity is high in grass and silver carp, but negligible in bighead and black carp |
| Series title | Journal of Great Lakes Research |
| DOI | 10.1016/j.jglr.2026.102751 |
| Volume | 52 |
| Issue | 1 |
| Publication Date | February 05, 2026 |
| Year Published | 2026 |
| Language | English |
| Publisher | Elsevier |
| Contributing office(s) | Columbia Environmental Research Center |
| Description | 102751, 8 p. |


