Fragmentation as a population rate-changer: A field experiment
Links
- More information: Publisher Index Page (via DOI)
- Document: XML
- Open Access Version: USGS Accepted Manuscript
- Download citation as: RIS | Dublin Core
INTRODUCTION
Continued scientific study of any phenomenon is expected to lead to increased understanding and predictive ability. However, the effects of habitat fragmentation on single-species populations and communities have been studied for more than half a century, yet ecologists cannot even agree on whether such effects generally tend to be beneficial or deleterious for affected populations. Early reviews (Dooley and Bowers 1998, Debinski and Holt 2000, McGarigal and Cushman 2002) noted that results of previous experimental studies of fragmentation mechanisms and effects were characterized by inconsistencies and failed to provide clear insights. More recent reviews have not provided clarification, with some concluding strong evidence of negative effects on biodiversity (Haddad et al. 2015, Fletcher et al. 2018) and others concluding small, positive effects (Fahrig et al. 2017, 2019). Yet others (Dooley and Bowers 1998, Betts et al. 2014) suggest that fragmentation responses may be species- and system-specific, perhaps defying generalization, but warn that such a perspective may lead to regional and local descriptions that are disconnected from general theory.
Here we develop some simple predictions about the effects of fragmentation and then test them with a field experiment. This experiment is focused on single-species populations rather than on ecological communities. In addition, we are interested in fragmentation per se, and not in the reductions in area that frequently accompany fragmentation. However, our fragmentation treatment did result in some reduction in grid area (see below). Many other fragmentation studies have developed experimental landscapes with multiple patches of different sizes and distances from one another (e.g., see Diffendorfer et al. 1995; Dooley and Bowers 1996, 1998; Schweiger et al. 2000; and reviews cited above). In contrast, we focused on the simplest of fragmented landscapes: replicate 2-patch systems identical in size and interpatch distance.
The varied interpretations of previous fragmentation research results do not lead to clear predictions about effects. Many factors ranging from interspecific interactions (predation, competition), to shifts in disturbance regimes, to nutrient cycling are potentially affected by fragmentation and have been proposed as possible mechanisms underlying effects on focal populations (e.g., Debinski and Holt 2000, Fischer and Lindenmayer 2007, Haddad et al. 2015). Rather than assembling a list of such factors to use in making predictions, we developed simple, conditional predictions about fragmentation effects based solely on dispersal. All changes in population size are attributable to the four vital rates, survival, reproduction, emigration and immigration, with the latter two rates jointly referred to as dispersal. An important effect of fragmentation on any population entails changes (reductions) in rates of dispersal, so a natural approach to considering fragmentation effects is to focus on the consequences of such changes. Investigation of causal factors via their effects on vital rates, as opposed to state variables, carries multiple inferential advantages (e.g., Yackulic et al. 2015, Nichols and Cooch 2025).
Theoretical explorations of fragmentation effects have emphasized the importance of dispersal parameters in their modeling efforts (e.g., Fahrig and Paloheimo 1988, Kareiva 1990). The concept of “landscape connectivity” highlights dispersal of animals from one patch to another and refers to the degree to which such movement is facilitated or impeded (Taylor et al. 1993) by landscape features such as fragmentation. The importance of immigration and emigration to population dynamics has been noted repeatedly (Krebs et al. 1973, Abramsky and Tracey 1979, Connor et al. 1983, Nichols and Pollock 1990, Lieury et al 2015ab, Vasudev et al. 2015, Fletcher et al 2016, Millon et al 2019) and is emphasized in discussions of metapopulations and related concepts such as source-sink dynamics (Pulliam 1988) and patch contributions (Runge et al. 2006, Sanderlin et al. 2012).
We begin by describing the field methods for fragmenting our experimental landscapes and sampling meadow vole populations. We then describe our analytic methods for estimating meadow vole demographic parameters and fragmentation effects based on our before-after, control-impact (BACI; e.g., Stewart-Oaten and Bence 2001) study design. We introduce expressions for use in predicting demographic effects of changes in dispersal. We develop predictions about effects of fragmentation via computations with pre-treatment population data on the relative effects of dispersal on population gains and losses. Finally, we present results and assess the degree to which predictions were supported.
METHODS
Field methods
We established 4 replicate pairs of grids during late summer, 1991. Each grid began as a 7 x 15 rectangle of trapping stations with 7.6 m between each station. Each replicate (set of 2 grids) was trapped every 8 weeks (primary periods) from 23 September 1991 to 24 August 1993, producing 12 primary sampling periods. Trapping sessions extended for 5 consecutive days (secondary periods) for the first 8 primary periods and for just 2 consecutive days for the final 4 primary periods. The reasoning for this change was the relatively small bias in abundance estimates obtained using a 2-sample model (because of high capture probabilities) as compared with a mixture model (Norris and Pollock 1996, Pledger 2000) that required >2 sample occasions and permitted heterogeneous capture probabilities. The substantial extra field effort required for the mixture model was simply not needed. All grids were established in old field habitat that had been maintained by periodic mowing at Patuxent Wildlife Research Center, Laurel, Maryland, USA.
The 2 grids in each pair were in close spatial proximity to each other and were always trapped on the same days. Our general schedule was to trap 1 grid pair 1 week, take the next week for data entry, and then trap the next grid pair the following week, etc. One grid was randomly selected to be the treatment grid and the other the control grid in each pair. Both grids in each pair were sampled during primary sampling periods 1-4, the pre-treatment phase of the experiment. Just prior to sampling in primary period 5, the treatment grid in each pair was fragmented by plowing and discing 7.6 m around the perimeter and through the middle of the grid, dividing it into two 7 x 7 squares of trapping stations (Fig. 1). Approximately 3.8 m (half the distance between traps) of habitat was left between the outer trapping stations and the edge of the plowed strip on fragmented grids. The grids were plowed about every 6 weeks to maintain the strips of bare ground creating the fragmented habitats. This plowing schedule prevented the establishment of even low levels of vegetative cover. The maintenance of fragmented habitats continued for the remainder of the study during primary sampling periods 5-12. This treatment was designed to reduce, but not eliminate, movement between fragments.
The fragmentation treatment was intended to focus on patch isolation and reduction of movement between patches, and not so much on reduction of habitat area that frequently accompanies fragmentation. The plowing of the central strip on fragmented grids resulted in an area of good habitat that was 7% smaller than that on control grids. In addition, we recognize that some animals with range centers outside the control grids are exposed to trapping effort on the grids, so the plowed periphery may have reduced the area of the fragmented grids even more. Nevertheless, we expected that the relatively small habitat loss in our study would not have substantive effects. This belief was based on experience with a previous fragmentation study on our own system (Coffman et al. 2001), and on fragmentation studies such as that of Wolff et al. (1997) in which a 70% reduction in habitat on fragmented plots did not adversely affect Microtus canicaudus population size (also see Dooley and Bowers 1998), and reduced movement was the “most obvious effect of habitat fragmentation”. In addition, we conducted an analysis (see HYPOTHESES AND PREDICTIONS), results of which supported our belief that population-dynamic effects of loss of habitat were relatively small. In summary, we recognize that our estimated treatment effects result from both changes in movement and habitat loss. However, we expect habitat loss to be a minor contributor (also see DISCUSSION) and thus based our predictions on changes in movement.
A single Sherman live-trap made of aluminum and covered by a piece of plywood was placed at each trap station. Traps were prebaited (baited and locked open) 3–4 d before each primary trapping session. Traps contained cotton and were baited with whole rolled oats. The daily trapping schedule involved setting traps in the evening, checking them in the morning and closing them during the day. Newly captured animals were marked with individually numbered Monel fingerling tags in their ears, and identification numbers of recaptures were recorded. The captured animal’s trap location, sex, and reproductive condition were recorded as well. For females, size of nipples (small, medium, or large), perforation of vagina (yes or no), and obvious pregnancies were recorded. For males, we recorded whether or not the testes were descended. Each animal was weighed to the nearest gram on a Pesola 100-g scale and its weight was recorded.
Data-analytic methods
Capture-recapture models (Williams et al. 2002, Lebreton et al. 2009, Seber and Scofield 2019) were used to draw inferences about focal demographic parameters during pre- and post-treatment periods. Specifically, the temporal symmetry model developed by Pradel (1996) was used to estimate rates of apparent survival, recruitment and population growth for adult animals (g, Krebs et al. 1969). The multistate modeling approach of Brownie et al. (1993) was used to estimate rates of adult movement. The estimation of these parameters was implemented in program MARK (White and Burnham 1999), and we used the following notation:
Δt
interval (in months) separating sampling occasions t and t +1.
apparent survival rate; probability that an animal (grid g, age a [(J = juvenile, A = adult], sex s [F = female, M = male]) in the sampled population (the area exposed to traps) in primary sampling occasion t was still in the sampled population in primary sampling occasion t+1.
apparent monthly survival rate; probability that an animal on grid g of age a and sex s survives a month during the period Δt.
monthly recruitment rate; expected number of new adult animals on grid g of sex s in the sampled population at sampling occasion t+1 per adult of sex s in the sampled population at occasion t, scaled to a 1-month period.
adult movement rate; probability that an adult animal of sex s on half 1 of grid g in sampling occasion t is in half 2 of grid g in sampling occasion t+1, given that it is alive in the sampled population at occasion t+1. In all models we imposed the constraint = .
population size; number of animals of age a, sex s in the sampled population of grid g at sampling occasion t.
monthly rate of population increase for adults of sex s on grid g between occasions t and t+1;
We conducted 5 sets of capture-recapture analyses. The first set was focused on selecting appropriate models for temporary emigration and capture probabilities, and all subsequent analyses then used the models selected for these processes. The second set of models focused on treatment effects on adult meadow voles for survival, recruitment, and population growth. The third set of models focused on treatment effects for rates of adult movement. In all 3 of these sets of analyses, each pair of grids was modeled separately, because of the different locations of the pairs and the different days on which the pairs were trapped. Our primary approach for assessing treatment effects was to estimate mean effect size (MES, see below).
The fourth set of analyses focused on treatment effects on juvenile (22g, Krebs et al. 1969) survival. Data were segregated by sex and grid as for the adult analyses. However, numbers of juvenile captures were relatively small and unable to support sex-, time-, and grid-specific modeling. Instead, juvenile survival was constrained to be the same for males and females. Combining data from all control grids, we estimated one juvenile survival for all sampling occasions during the pre-treatment period and a different survival for all occasions during the post-treatment period. Similarly, for each of these periods we estimated juvenile survival for fragmented grids. A single MES was used to assess treatment effects.
The fifth set of analyses used pre-treatment capture-recapture data and estimates from the previous 4 sets of analyses to make predictions about fragmentation effects. We used both adult and juvenile capture-recapture data for these efforts. We summarize the modeling for the second, third and fourth sets of analyses below. The first and fifth sets are described in Appendices A and D, respectively.
Apparent survival rate, recruitment rate and population growth rate
We used a robust design model (Pollock 1982) with apparent survival rate () and recruitment rate () as the focal parameters (Pradel 1996). Population growth rate was estimated as a derived parameter, = . Capture probabilities were modeled based on results of the prior modeling step as for primary periods 1-8, where p is capture probability, modeled as an occasion-specific mixture (mx) model with occasion-specific mixture parameter (; see Appendix A). Capture probability for primary periods 9-12 was modeled as occasion-specific and equal for the 2 secondary periods, without heterogeneity, .
We fit 3 different models for apparent survival rates and recruitment rates. The most general model was Strt*t+sx , ftrt*t+sx with occasion-specific (t) estimates of monthly survival and recruitment that differed by grid treatment (trt) and additively by sex (sx). A more specific model, Strt*per+sx, ftrt*t+sx , modeled recruitment as occasion-specific but depicted apparent survival as period-specific, where “period” refers to pre- and post-treatment periods. This model included a treatment x period interaction and an additive sex effect. Similarly, we modeled survival as occasion-specific but recruitment as period-specific, Strt*t+sx , ftrt*per+sx. These latter two models represented attempts to reduce the number of parameters within each treatment period of the study, but model selection provided strong evidence for the most general model with both survival and recruitment occasion-specific. MES estimates were thus computed using arithmetic means of time-specific estimates for all three parameters, survival, recruitment, and population growth. Estimates of survival and recruitment came directly from model Strt*t+sx, ftrt*t+sx. Monthly population growth rates for adults were computed as: = .
Movement rate
The previous modeling of temporary emigration and capture probability (Appendix A), and the subsequent modeling of survival, recruitment, and population growth, all treated each 7 x 15 trap station grid as a single unit for the purposes of estimation. In contrast, inferences about adult movement rate were based on multistate modeling (Arnason 1972, Brownie et al. 1993, Schwarz et al. 1993, Lebreton et al. 2009) of each grid as a 2-location system, with grid half 1 comprised of trap rows 1-7 and grid half 2 comprised of rows 9-15. Probability of moving from grid half 1 at occasion t, to grid half 2 at occasion t +1, conditional on the animal being alive and on the grid at occasion t+1 () was set equal to the probability of moving from half 2 to half 1 on the same grid; i.e., . Capture probability was modeled the same for the 2 grid halves and followed the same model structure as for the previous modeling. Apparent survival rates on the two halves of each grid were set equal () and allowed to vary by primary trapping occasion and sex Strt*t+sx, the same as for the above survival- recruitment modeling. The following two movement models were fit to the data, trt*t+sx and trt*per+sx, and the model with period-specific movement parameters was selected. Approaches to inference about treatment effects on movement were the same as for apparent survival and recruitment (see below), except that there was no computation of occasion-specific means, as we used the estimates for each period in the analyses.
Juvenile survival rate
We used a Pollock (1981) 2-age model to estimate juvenile survival rate with the same structure on adult survival as described above. However, because of limited numbers of juveniles, we modeled their survival differently. Sex of juveniles was recorded and used in the modeling, but juvenile survival was set equal for males and females, based on both the data limitations and our expectation of few behavioral differences between the sexes for juveniles. Instead of allowing juvenile survival to be occasion-specific, we constrained survival to be constant within each period, pre- and post-treatment. Finally, we constrained juvenile survival within each period to one value for all control grids and a different value for all fragmented grids. This approach produced a single MES for all grids combined.
Treatment effect inferences
Treatment effects for rates of movement, survival, recruitment, and population growth were estimated using a general BACI approach and focused on differences in mean parameters for the 2 periods (pre-treatment and post-treatment) on treatment versus control grids. denotes any of the focal parameters listed above for treatment grid j (trtj = 1 for control, 2 for treatment) and period i (peri =1 for pre-treatment; 2 for post-treatment). The BACI approach led us to estimate mean effect size (MES) for any parameter θ as the difference between pre- and post-treatment values between treatment and control grids:
We computed means of time-specific estimates for the pre- and post-treatment periods and used these in conjunction with equation 2 to estimate for apparent adult survival rate, recruitment rate, and population growth rate. For movement, we instead used the period-specific rate estimates rather than means of time-specific estimates. For each of these 4 focal parameters for adults, var ( was estimated using the variance-covariance matrix for the 4 of equation 2. As a means of quantifying the magnitude of the estimated MES relative to its standard error, we computed z statistics for each grid pair x sex combination as . We also computed an overall z statistic as , where 8 is the number of grid x sex combinations. For those who favor significance tests, the z statistics are distributed as normal (0,1) under the null hypothesis that the mean effect is 0.
We obtained four estimates of juvenile survival : control, pre-treatment (; control, post-treatment (; fragmented, pre-treatment (;; fragmented, post-treatment (;. Instead of estimating MES for each sex-grid combination as we did for adult parameters, we estimated a single MES (and associated across all grids and both sexes to test for an effect of fragmentation on juvenile survival.
MES for movement probability was based on ln( (where the per subscript refers to period, pre-treatment or post-treatment). We used the ln transformation to reflect proportional treatment differences. This was appropriate for movement because some of these estimates were small, and our predicted treatment effects are based on proportional changes in immigration and permanent emigration.
HYPOTHESES AND PREDICTIONS
Our primary hypotheses focused on the effect of habitat fragmentation on 5 parameters of the meadow vole populations that were estimated directly via the capture-recapture modeling: adult movement rate, apparent survival rate for adults and juveniles, recruitment rate, and adult population growth rate. We focused our treatment on movement reductions associated with fragmentation but recognize that our treatment produced some habitat loss as well.
Our predictions first focus on the magnitude of potential reductions in adult movement. We then develop expressions for changes in apparent survival rate of adults and juveniles, recruitment rate, and adult population growth rate expected to accompany changes in movement. These expressions require decomposition of the survival and recruitment rates estimated via capture-recapture into components that do, and do not, include movement. Predictions about fragmentation-induced change in these rate parameters are based on our hypotheses about the magnitudes of these decomposed survival and recruitment rates, some of which are based on the literature and previous work with this system, and others of which are based on ad hoc analyses of pre-treatment data.
Movement Rate
We expected a decrease in movement probability following fragmentation. This expectation was based on small mammal literature as well as experience with our own system (Coffman et al. 2001). Movement rate is a difficult parameter to estimate, and our prior work on this system produced imprecise estimates. Thus, we predicted that fragmentation based on our specific treatment could produce reductions in adult movement ranging from 20% to 80%.
Apparent Survival Rate
The complement of apparent survival rate (1- ) as estimated using the Pradel (1996) temporal symmetry model includes individuals that die between successive sampling occasions and those that permanently emigrate from the population between sampling occasions. We predicted that fragmentation should reduce the rate of permanent emigration, producing an increase in apparent survival. Predictions about the degree to which survival should increase require decomposition of apparent survival into 2 multiplicative components, one associated with mortality and one with movement (Appendix C). These components were estimated (Appendix D) and used to compute predicted changes associated with fragmentation (Appendix E). The estimated loss rate associated with mortality for males was approximately 8x larger than the loss rate attributable to permanent emigration (Appendix E). Thus, based on these ad hoc estimates, reductions in permanent emigration by 80%, 50% and 20% were predicted to increase adult survival by only about 0.01 to 0.02 (1% to 3%) based on equation E1.
As noted in the Discussion of Appendix D, we believe that the component of adult loss (1-) associated with permanent emigration was underestimated to a large degree. If losses from movement and death were roughly equal, then we would predict that fragmentation would produce increases in adult survival of 0.03 to 0.11 (4% to 16%).
We based predictions for juvenile survival on reasoning similar to that for adults. However, juvenile data were inadequate for use of our approach for decomposing adult survival (Appendix D), so we simply assumed the same proportion of losses attributable to permanent emigration as for adults. These small estimated losses to emigration led to very small predicted increases in juvenile survival associated with fragmentation. However, if losses from permanent emigration and mortality are approximately equal, then we expect fragmentation to produce larger increases in juvenile survival (0.03 to 0.11).
Recruitment Rate
Recruitment () includes new individuals that have been produced in the population via reproduction and immigrants from outside the population (Nichols and Pollock 1990). Fragmentation is expected to reduce the immigration component of recruitment. However, the reproductive component of recruitment includes both reproductive rate (juveniles produced per adult individual) and survival of those juveniles to recruit to the adult population. We expect the apparent survival of juveniles to be increased by fragmentation (reduced permanent emigration, see above). Thus, fragmentation is expected to decrease one component of recruitment rate (immigration) and increase another component (reproductive recruitment). Predictions about even the direction of fragmentation effects thus require decomposing recruitment rate into its components (Appendix C), estimating those components (Appendix D), and then considering effects of reduced movement (Appendix E).
Our ad hoc estimators for recruitment components indicated that the mean proportional contribution of reproduction to recruitment during the pre-treatment period for most grids was only about 5-10%. As a result, decreased permanent emigration associated with fragmentation should result in small increases in overall recruitment (<0.03) even with an 80% reduction in movement. In contrast, reductions in immigration by 80%, 50% and 20% should result in overall recruitment decreases ranging from 0.27 to 0.06 (equation E2) or 73% to 18%.
However, as noted in Appendix D, we believe that (1) the component of juvenile loss associated with movement was substantially underestimated, and that (2) the component of recruitment attributable to immigration was substantially overestimated. Our previous work with this specific Microtus system (Nichols and Pollock 1990, Coffman et al. 2003) showed evidence of similar rates for the two components of recruitment, reproduction and immigration. In the absence of other information, we used this similarity of rates for our predictions of increases in reproductive recruitment of 1-3%, decreases in recruitment from immigration of 3-13%, and decreases in overall recruitment of 2-10%.
Population Growth Rate
Population growth rate in this system is the sum of rates of apparent survival and recruitment of adult voles (Equation 1). As noted above, fragmentation is expected to increase apparent survival of adults and young (a component of recruitment) and decrease immigration (also a component of recruitment). Fragmentation effects on population growth rate could thus be either positive or negative, depending on the specific effects on adult survival, juvenile survival, and immigration, as well as on values of the components of recruitment and survival not affected by movement. Equation E4 was used to predict changes in population growth rate that would accompany different proportional reductions in movement associated with permanent emigration of adults and juveniles, and immigration. However, as noted above, we have little information for predictions about changes in movement associated with fragmentation, so for purposes of predicting effects on population growth, we made the simplifying assumption that proportional changes in movement are the same for apparent survival of adults and juveniles, and for immigration. We then used the decomposed parameter estimates for the pre-treatment period with equation E4 to predict the change in population growth rate expected to accompany different proportional decreases in movement. Losses and gains associated with reductions in movement of 80%, 50%, and 20% led to predicted reductions in population growth rate of about 0.23, 0.14 and 0.06. However, based on our revised predictions for adult survival and recruitment, we believe that our best predictions for changes in population growth rate in response to fragmentation are a modest increase (0.01-0.03).
Summary
We had initially hoped to develop specific predictions about the effects of fragmentation on our 5 focal parameters: adult rate of movement, apparent adult survival rate, apparent juvenile survival rate, recruitment rate, and adult population growth rate. Predictions about effects on survival, recruitment and population growth are dependent on predictions about fragmentation effects on movement. However, we had no basis for precise predictions about movement, so had to consider a wide range of possibilities.
We decomposed focal parameters to separate components that reflected movement into or out of the population (Appendix C), allowing us to develop expressions for changes in parameters expected to accompany specified changes in movement rates (Appendix E). Because of the importance of movement changes to fragmentation effects, we believe that the decomposition of commonly estimated parameters into components that isolate movement contributions should be useful to other investigations. We used ad hoc approaches to estimate these decomposed parameters using pre-treatment data (Appendix D) and then used the estimates to make predictions (Appendix E). However, potential problems with the estimators caused us to also consider other approaches to making predictions.
RESULTS
A total of 3185 meadow vole individuals were captured a total of 14059 times on the four pairs of study grids at Patuxent Wildlife Research Center, Laurel, Maryland, September 23, 1991 – August 24, 1993 (Table 1). Adult data were adequate for fitting all of the models required for our BACI analyses. As anticipated, we had to model juvenile data with parameters held constant across sexes, across grids within each of the 2 treatments, and across sets of sampling occasions within the pre-treatment period and the post-treatment period.
Table 1.
Total numbers of meadow vole individuals and captures on the 4 pairs of study grids at Patuxent Wildlife Research Center, Laurel, Maryland, September 23, 1991 – August 24, 1993.Our BACI analyses were focused on 5 parameters, rates of adult movement, adult survival, juvenile survival, recruitment, and adult population growth. Estimates of these parameters are summarized and plotted in Appendix F. Population size was estimated, but we selected population growth rate for our tests of fragmentation effects. Meadow voles are known to undergo strong fluctuations in population size, with peaks at the latitude of our study site occurring every 2 or 3 years (e.g., Krebs et al. 1969, 1973). The first 9 months of our study were designated as pre-treatment, and estimates of population growth rate on most grids during this period were indicative of the increase phase of the population “cycle”. The next 9-10 months included the end of the increase phase and the entirety of the peak phase for these populations. During the remaining 4-5 months, the populations were in the decline phase. Seasonality is also likely to be an important influence on meadow vole population dynamics in Maryland. By focusing on time-specific differences in rate parameters between paired treatment and control grids, the BACI analyses and resulting estimates of mean effect size provided appropriate tests of fragmentation effects in the face of this natural variation.
Capture Probability and Temporary Emigration
The initial modeling step (Appendix A) led us to model capture probability with additive effects of time (sampling occasion) and individual heterogeneity. We modeled the heterogeneity with a 2-point finite mixture model (Norris and Pollock 1996, Pledger 2000). As noted in the description of field sampling, we trapped for 5 consecutive days in each primary sampling occasion for the first 8 such occasions. The finite mixture modeling of heterogeneity was thus used for the initial 8 primary occasions. Each of the last 4 primary occasions included only 2 days of trapping, so capture probability was modeled as a function of primary occasion only. Analyses based on Kendall et al. (1997) did not provide substantive evidence to include temporary emigration in our models. Modeling decisions from this step were carried through all subsequent modeling.
Movement Rate
Movement analyses were based on model trt*per+sx. Estimates of period-specific movement for the 8 grids ranged from 0.01 to 0.10 for females, and from 0.02 to 0.20 for males (Table F1). The estimated MES [ln( are all negative as predicted, indicating less movement in the post-treatment period for fragmented grids than for paired control grids (Fig. 2). One-tailed probabilities associated with the MES statistics were <0.10 for all grid x sex combinations, <0.05 for 4 of the 8 combinations, and <0.01 overall (z). The reductions in movement post-treatment were 60% to 85% larger for fragmented grids than for controls.
Apparent Adult Survival Rate
Inferences were based on model Strt*t+sx, with a treatment by sampling occasion interaction and an additive sex effect. Mean monthly estimates of apparent survival rates for the pre- and post-treatment periods across the 8 grids ranged from 0.68 to 0.86 for females and 0.61 to 0.83 for males (Table F2). The estimated MES ( are all positive as predicted (Fig. 3). Indeed, mean survival estimates were smaller during the post-treatment period than the pre-treatment period for every control grid, but survival estimates increased from the pre-treatment period to the post-treatment period for every treated grid. One-tailed probabilities for MES statistics were <0.02 for all grid x sex combinations.
Apparent Juvenile Survival Rate
Inferences were based on a model for which juvenile male and female survival rates were constrained to be equal, and survival rates were constrained to be equal for all 4 control grids across all sampling occasions within each of the 2 periods, pre-treatment and post-treatment, as were survival rates for all 4 fragmented grids. The 4 resulting mean monthly estimates of apparent juvenile survival were smaller than most adult survival estimates, as expected (Table F3). MES () = 0.17, with SE[MES ()]=0.13, and the MES z = 1.29, with 1-tailed probability of about 0.10, providing evidence supporting our prediction of increased juvenile survival following fragmentation.
Recruitment Rate
Recruitment rates were based on model ftrt*t+sx, with a treatment by sampling occasion interaction and an additive sex effect. Mean monthly estimates of recruitment rates for the pre- and post-treatment periods across the 8 grids ranged from 0.17 to 0.41 for females and 0.19 to 0.51 for males (Table F4). The estimated MES () were not consistent across grid pairs, being positive for both males and females on grid pairs 1 and 3 and negative on grid pairs 2 and 4 (Fig.4). None of the MES statistics provided much evidence favoring our prediction of lower recruitment to the fragmented grids. Grid pair 4 males and females had 1-tailed probabilities supporting this prediction of approximately 0.15, and probabilities for all other grids were higher. The probabilities for the statistics computed over all 4 grid pairs were >0.96 for both males and females, running counter to our predictions
These overall recruitment rates provide the basis for our inferences about fragmentation effects. However, we also used the methods developed to decompose recruitment rates into components associated with in situ reproduction and immigration (Appendices C, D) to gain additional insight about fragmentation effects. Specifically, we had already computed point estimates of the two recruitment components for the pre-treatment period, as we used these as a basis for our predictions. Thus, we also computed point estimates of reproductive and immigration recruitment for the post-treatment period and used the means to compute MES statistics for the 2 components. All of the point estimates for in situ reproduction MES were positive and thus consistent with our prediction that decreased permanent emigration of juveniles would increase apparent juvenile survival, following fragmentation (Table 2). The point estimates for immigration MES were positive for grid pair 1 and negative for all other grid pairs. We had predicted that fragmentation would produce large negative effects on immigration, but not all estimates were consistent with this prediction.
Adult Population Growth Rate
Mean monthly adult population growth rate was estimated as a derived parameter, = , based on a general Pradel (1996) model (Strt*t+sx, ftrt*t+sx) in which both survival and recruitment were modeled with a treatment by sampling occasion interaction and an additive sex effect. Mean monthly estimates of population growth rate for the pre- and post-treatment periods across the 8 grids ranged from 0.93 to 1.20 for females and 0.90 to 1.22 for males (Table F5). The MES ) were all positive (Fig. 5). Based on our revised prediction of fragmentation producing small positive changes in population growth rate, probabilities associated with MES statistics were <0.03 for males and females of grid pairs 1-3, and <0.08 for both sexes on grid pair 4. Thus, our prediction of positive changes in population growth was supported, but the MES ) were generally larger than we predicted.
DISCUSSION
Reviews cited in the INTRODUCTION support the conclusion that there is no clear scientific consensus on either the effects of fragmentation on animal populations and communities or on the various mechanisms that produce those effects. Given this uncertainty, we conducted a replicated population-level experiment to investigate effects of one form of fragmentation on meadow vole population dynamics. We identified a reduction in dispersal as the most basic predicted effect of fragmentation and then based our predictions about population effects entirely on hypotheses about how reduced dispersal affects the rate parameters underlying population dynamics. We do not claim that the various other mechanisms proposed to underlie fragmentation effects are not relevant. Instead, we simply view predicted effects of reduced dispersal on rate parameters as a parsimonious starting point.
We conducted a 2-year capture-recapture study on 4 pairs of replicate meadow vole populations. No manipulations were implemented for the initial 7 months, followed by habitat fragmentation on one member of each grid pair and subsequent trapping for 17 months. We used a BACI-type analysis that focused on pre- and post-treatment differences for grids that were and were not fragmented. Specifically, we computed estimates of MES, effectively asking whether pre- vs. post-treatment period differences depended on treatment. Our capture-recapture analyses provided estimates of five rate parameters of primary interest: adult movement, adult and juvenile survival, recruitment, and adult population growth.
Movement Rate
Our most basic prediction for fragmented grids was a reduction in movement. Although we developed ad hoc approaches to directly estimate and test for movement in and out of our systems (Appendix D), our experimental design permitted a more robust approach. Specifically, we directly estimated rates of adult movement between the 2 grid halves of each grid, before and after fragmentation. Estimated changes in movement between pre- and post-treatment periods on treated and control grids showed post-treatment decreases in movement on fragmented grids with movement probabilities only 15% to 40% that of control grids. The results thus supported our predictions, although the magnitude of the estimated effect varied among grids. Much of the variation in MES estimates is likely associated with low precision of movement estimates. When movement is a relatively rare event as in our system (most estimates < 0.10), it is simply difficult to estimate, and estimates tend to have low precision.
The degree to which movement is reduced by fragmentation clearly depends not only on the identity of the affected species but also the nature of fragmentation. For example, many studies of small mammals have created fragments via mowing (e.g., Dooley and Bowers 1998, Schweiger et al. 2000) whereas our study used plowing to produce bare ground around fragments. The width of inhospitable habitat (bare ground) in our study was 7.6m, and this has been variable within and among studies as well. In our study the inferences about between-patch movement were between patches of roughly equivalent quality, as determined via selection and assessed by pre-treatment sampling, whereas this has not necessarily been true of some other studies. Thus, beyond the basic prediction of fragmentation producing reductions of movement, among- and within-study variation in various components of fragmentation do not permit specific predictions or comparisons based on other work.
Additional potential sources of variation in rates of movement that increase difficulty in making specific predictions and comparisons include sex, age, and the direction of movement (i.e., into vs. out of the focal population). For example, average movement rates of male meadow voles in our study were 1.5–3 times larger than those of females, both before and after fragmentation. The reduced numbers of juvenile captures relative to adults precluded accurate inferences about potential age-specificity of movement. In addition, it may be that age- and sex-specific rates of immigration vs. emigration differ, depending on the relative fitness gains associated with movements in vs. out of the focal population. Such gains are likely to be a function of focal population density, phase of meadow vole population fluctuations (e.g., Krebs et al. 1969, 1973), distance of focal populations to potential sources of emigrants, etc.
Apparent Adult Survival Rate
We predicted that fragmentation would decrease permanent emigration, and thus increase apparent adult survival. Our results provided strong evidence supporting this prediction for both males and females. Males showed somewhat lower survival rates than females, also consistent with our predictions. The complement of apparent survival includes two components, mortality and permanent emigration, only one of which is expected to necessarily be affected by reduced movement. Our efforts to estimate these two components suggested that 88-90% of adult losses were associated with mortality (Appendix D). The small fraction of estimated losses attributable to permanent emigration led to a reduction in the predicted influence of reduced movement on apparent survival.
However, we noted (Appendix D) that we had concerns about our approach to inference for components of apparent survival. Specifically, we are attempting to estimate the upper asymptote of the relationship between apparent survival and grid area/perimeter ratio based on data from three points near the lower end of the relationship (i.e., grids with small area/perimeter, see Appendices C, D). We suspect that permanent emigration is a more important component of loss than indicated by our estimates. Indeed, Krebs et al. (1973) estimated that up to 69% of female meadow vole losses in an Indiana population resulted from permanent emigration, and estimates by Gaines et al. (1979) approached 100% for Microtus ochrogaster in Kansas. However, estimates from both of these studies were based on movements of marked animals into “vacant” habitats created by removing animals, and may not apply to movements into existing vole populations. Because accurate prediction of fragmentation effects requires separate estimates of losses attributable to movement and mortality, it may be useful to include larger grid sizes with the approach developed in Appendices C and D, or to consider other approaches for accomplishing this decomposition (e.g., Jackson 1939, Manly 1985, Powell et al. 2000, Schaub and Royle 2014).
Apparent Juvenile Survival Rate
Our inferences about apparent juvenile survival were not as strong as those for adult survival, primarily because of much smaller numbers of juvenile captures. As predicted, estimated juvenile survival was smaller than that estimated for adults. Also following predictions, the positive MES provided evidence that juvenile survival rate increased in response to the fragmentation treatments, contributing to increased reproductive recruitment.
We were not able to estimate decomposed apparent survival parameters for juveniles in the same way as for adults, because of sample size issues. We thus borrowed information from our adult analyses, assuming a similar fraction of juvenile and adult losses to permanent emigration. Therefore, uncertainty remains about how to predict the magnitude of the fragmentation effect on juvenile survival.
Recruitment Rate
Recruitment rate estimates were not consistent with our predictions that fragmentation should reduce recruitment. Two pairs of grids showed negative MES estimates, as predicted, and the other two pairs showed positive estimates. Recruitment can be viewed as the sum of two additive components, reproductive recruitment and immigration. Fragmentation is expected to increase reproductive recruitment by decreasing permanent emigration of juveniles, thereby increasing apparent juvenile survival. Our MES estimate for juvenile survival provided evidence that apparent juvenile survival indeed increased. Fragmentation is also expected to reduce immigration. We predicted that reductions in immigration would dominate the recruitment effects, leading to an overall decrease in recruitment, but this did not occur.
MES based on point estimates of recruitment components were consistently positive for reproductive recruitment as predicted, indicating an increase in this recruitment component with fragmentation. MES estimates for immigration were negative for three of the four grid pairs and generally smaller in absolute value than those for reproductive recruitment, indicative of smaller effects. The magnitudes of these admittedly ad hoc MES estimates ran counter to our prediction that substantial decreases in immigration would dominate effects of fragmentation on recruitment.
The unexpected results for recruitment may be a result of overly simplistic predictions. For example, our predictions were based on the idea that fragmentation effects should be similar for movement into, and out of, a population. However, this hypothesized symmetry of movement may not be true. For example, all of our study grids were set out in relatively good, old field habitat. It is plausible that immigration by dispersers to a favorable habitat might not be affected as much by barriers to movement as emigration from such habitat. We partially tested this hypothesis by creation of very good habitat in two subsequent experiments, one involving density reduction and the other food supplementation, but conclusions await the results of our analyses.
Adult Population Growth Rate
Results provided strong evidence that fragmentation led to increases in population growth rate. The direction of the result was predicted, but we expected smaller MES than we observed. The larger MES are consistent with the absence of substantive declines in recruitment that we expected to accompany fragmentation. Reduction of permanent emigration on fragmented grids appears to have increased rates of apparent adult and juvenile survival, the latter leading to increased reproductive recruitment. Fragmentation did not appear to reduce immigration to the extent that we had predicted, so immigration effects did not counterbalance effects on permanent emigration.
Although our fragmentation treatment was designed to produce reduced movement of meadow voles, we noted that it also resulted in some habitat loss. We believed this loss to be so small as to not have substantive effects on the fragmented vole populations. Much larger habitat losses in other Microtus fragmentation experiments have not led to negative effects on population size or growth (Wolff et al. 1997, Dooley and Bowers 1998), so we based our predictions solely on movement reductions. A subsequent analysis provided some support for this decision (Appendix B). In addition, we note that our overall result of increased population growth rate on treated grids does not support a hypothesis of substantial population reductions associated with habitat loss.
Other Microtus Fragmentation Studies
This study was focused on a general question about effects of fragmentation, but inferences are especially relevant to previous work on Microtus population dynamics. Some of the first specific indications of the importance of dispersal to Microtus population ecology were labeled the “fence effect” (e.g., Krebs et al. 1969, 1973; Boonstra and Krebs 1977; Tamarin et al. 1984, Ostfeld 1994), in which populations that were fenced (preventing movement in and out) showed high apparent survival rates and attained very high densities, eventually followed by overexploitation of resources, destruction of habitat, and population decline. Krebs et al. (1973) concluded that dispersal must play a key role in Microtus population regulation (also see Lidicker 1962, Boonstra and Krebs 1977, Abramsky and Tracy 1979).
Prohibition of all dispersal (e.g., via fencing) to or from a patch can be viewed as 1 endpoint of a fragmentation continuum, with free movement throughout a population the other endpoint. The plowing treatment in our study was designed to reduce, but not eliminate, movement, producing a scenario between these 2 endpoints. The reduced movement probabilities that accompanied fragmentation in our study are consistent with other results for Microtus (e.g., Diffendorfer et al. 1995, Wolff et al. 1997, Coffman et al. 2001). Although not a central question for our study, our finding of higher rates of movement for adult males than females is consistent with other studies of dispersal in Microtus (e.g., Wolff et al. 1997, Andreassen et al. 1998, Aars and Ims 1999, Coffman et al. 2001).
Our finding that fragmented grids showed higher population growth rates than control grids followed our predictions and is consistent with at least 2 other experimental studies of fragmentation effects on Microtus (Wolff et al. 1997, Dooley and Bowers 1998). Interestingly, investigators in these 2 studies viewed their results as unexpected. We found strong evidence of increased apparent survival on fragmented grids, but Dooley and Bowers (1998), who also studied meadow voles, found no evidence of increased adult survival on their fragmented grids. Thus, they speculated that the increased population growth on fragmented grids could be attributed to increased reproductive rate (Dooley and Bowers 1998). Our decomposition of adult recruitment rate provided evidence of an increased contribution of in situ reproduction on the fragmented grids, consistent with increased apparent survival of juveniles.
Our finding of higher apparent survival rates for females than males is consistent with findings of many others (e.g., Krebs et al. 1969, Tamarin 1977a, Coffman et al. 2001). Our estimates of the relative importance of emigration and death to total losses suggested that death was much more important, although we question these results. The proportion of losses attributable to dispersal in other Microtus studies are quite variable, ranging from 6% to nearly 100%, and often greater for males than females. (Myers and Krebs 1971, Krebs et al. 1973, Tamarin 1977b, Gaines et al. 1979). This substantial variation, coupled with the various methods on which these estimates are based, leave substantial uncertainty about the decomposition of Microtus losses.
Conclusions
The persistent uncertainty about effects of fragmentation on natural populations and communities, despite hundreds of previous experimental and observational studies, led us to use a simple experimental approach with predictions based on a single causal effect. We focused on population dynamics of a single species in replicate, 2-patch systems. Although many factors have been cited as contributing causal mechanisms for fragmentation effects, some of which may have been relevant to our system, we focused on the most obvious (our opinion) mechanism of reduced movement in and out of patches. We developed predictions for effects of reduced movement on population vital rates and then used a BACI design to estimate fragmentation effect sizes for these rates. We claim no necessary generality to our results, but believe that basic approaches with straightforward predictions about simple systems offer advantages to eventual understanding.
Results provided strong evidence of reduced movement of both adult males and females on fragmented grids relative to that on controls. Our experimental approach to fragmentation thus reduced, but did not eliminate, movement, as predicted. The study also provided evidence supporting our prediction that fragmentation would produce increases in apparent survival rate for both adults and juveniles. We predicted that fragmentation would have positive effects on reproductive recruitment, negative effects on immigration, and negative effects on overall recruitment. However, our results for overall recruitment were ambiguous, leading to speculation that immigration to good habitat might not be reduced as much as emigration from it, because of the asymmetry in habitat quality. Future work directed at estimation of immigration and emigration should be useful in reducing the uncertainty surrounding our recruitment results.
Various fragmentation reviews have arrived at different conclusions about whether fragmentation effects are negative (Haddad et al. 2015, Fletcher et al. 2018) or positive (Fahrig 2017, Fahrig et al. 2019). We used the finite rate of population growth as our focal metric, viewing higher average growth rate as a positive effect. The study provided evidence that the effects of fragmentation on both apparent survival and recruitment combined to produce an overall positive effect on population growth rate. Although we predicted the direction of the effect, we thought it would be smaller than that which we estimated. This study certainly does not resolve the uncertainty surrounding fragmentation effects, but can be viewed as a contribution to the accumulated evidence on this topic (Nichols et al. 2019).
Our predictions were based explicitly on effects of reductions in movement on population vital rates; however, we note that our inferences about fragmentation incorporate other effects as well. Fragmentation-induced changes in amount of available habitat, predation, competition, edge effects, etc., could have contributed to the changes that we found in vital rates. Because we did not include such changes in our a priori hypotheses about this system, we made no effort to investigate them specifically.
We were pleased at the strength of evidence for some of our inferences about fragmentation effects, but these inferences are not necessarily general. They apply to our single-species study of simple 2-patch systems and are specific to meadow voles and to the nature of our fragmentation treatment (the strips of bare ground). Our study extended about 2 years, and we do not know the degree to which these effects may have persisted in the longer term. Of course, the generality of any experiment is limited by experimental design.
The process of trying to develop quantitative (as opposed to simply directional) predictions was useful in requiring hard thought (see Nicholson 1954) about underlying mechanisms. The decomposition of commonly estimated vital rates (Appendix C) allowed us to isolate movement components and to develop explicit expressions for changes in these rates, as well as overall population growth rate, as a function of changes in movement (Appendix E). We hope that these expressions are useful to others for predicting fragmentation effects. Our expression for changes in population growth rate demonstrates that fragmentation effects on movement alone can lead to either positive or negative changes in population growth. We focused on the direct effects of fragmentation on animal movement, but if fragmentation effects include other processes such as species interactions (e.g., predation or competition), these can be modeled via vital rate effects as well (e.g., Miller et al. 2012; Yackulic et al. 2014, 2018). We hope that evidence from fragmentation studies such as this will continue to accumulate and eventually lead to more definitive conclusions and generalizations about fragmentation effects.
FIGURE CAPTIONS
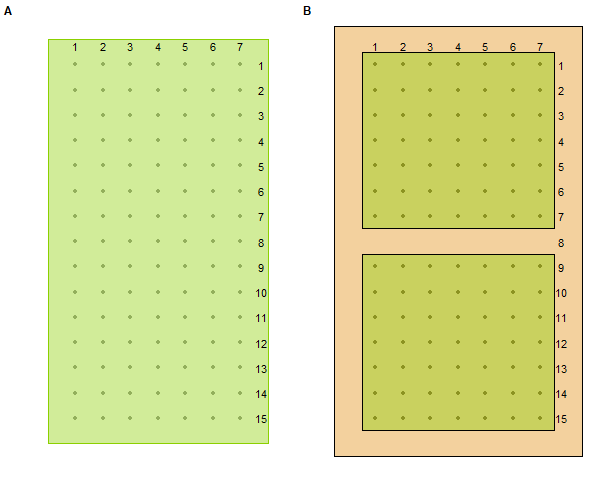
Basic grid design. A: continuous (unfragmented) grid. B: treated (fragmented) grid, where brown indicates bare ground. Distance between adjacent trap sites (dots) = width of brown strip (bare ground) = 7.6m.
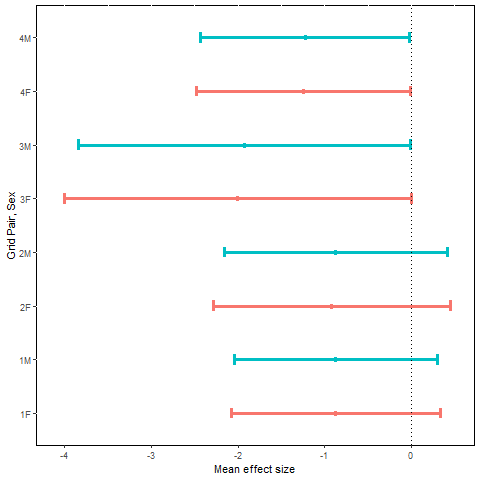
Mean effect size and approximate 95% confidence interval for the natural logarithm of estimated adult movement probability, MES [ln(, plotted by sex (F = female, M = male) and grid pair number.
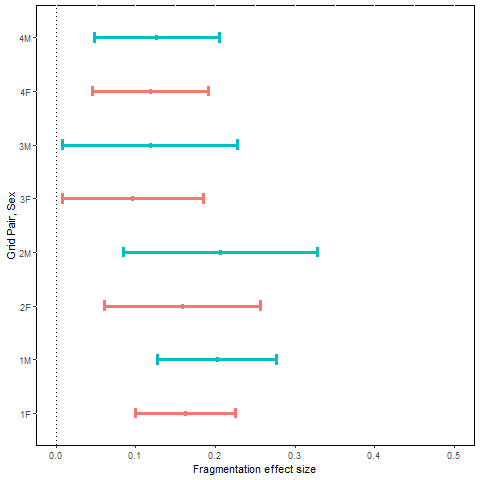
Mean effect size and approximate 95% confidence interval for estimated monthly adult survival probability, MES ( , plotted by sex (F = female, M = male) and grid pair number.
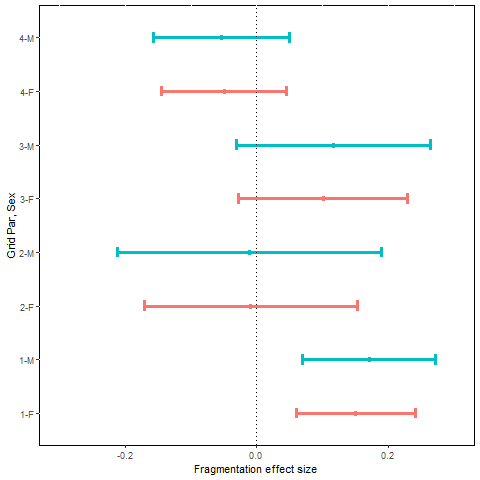
Mean effect size and approximate 95% confidence interval for estimated monthly recruitment rate, MES ( , plotted by sex (F = female, M = male) and grid pair number.
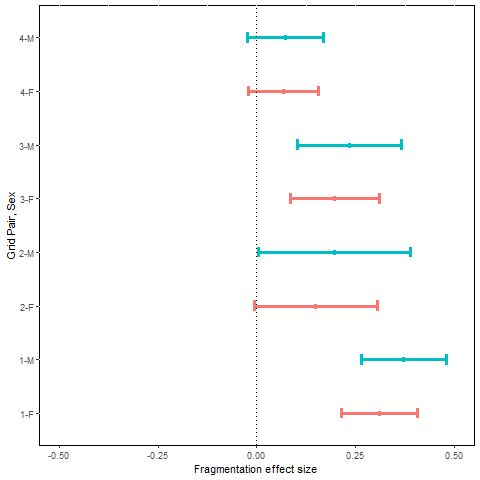
Mean effect size and approximate 95% confidence interval for estimated monthly rate of adult population growth, MES (, plotted by sex (F = female, M = male) and grid pair number.
ACKNOWLEDGMENTS
We thank Jeff Hostetler, Julien Martin, Divya Vasudev and 2 anonymous referees for providing constructive and useful comments on an earlier version of this manuscript. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Additional Information
CONFLICT OF INTEREST STATEMENT: The authors declare no conflicts of interest.
ORCID: James D. Nichols https://orcid.org/0000-0002-7631-2890
DATA AVAILABILITY STATEMENT: All capture-recapture data are available at: Nichols, J.D., Hines, J.E., Hinz, R.L., and Hinz, J., 2025, Meadow vole data for fragmentation study: U.S. Geological Survey data release, https://doi.org/10.5066/P13LNR4Q .
REFERENCES
Nichols, J.D., and E.G. Cooch. 2025. “Predictive Models are Indeed Useful for Causal Inference.” Ecology 106: e4517. https://doi.org/10.1002/ecy.4517.
Appendix A. Modeling Temporary Emigration and Capture Probabilities
Methods
The base model class for investigating temporary emigration and capture probabilities was the robust design (Pollock 1982, Kendall and Nichols 1995, Kendall et al. 1997). The open-population modeling component was that of the Cormack-Jolly-Seber model (Cormack 1964, Jolly 1965, Seber 1965). All modeling of capture probabilities and temporary emigration included a general sex x primary occasion model for monthly survival probability. Model selection for capture probabilities was based on Markovian temporary emigration models (sensu Kendall et al. 1997) in which emigration probabilities varied by occasion and sex. For primary sampling periods 1-8, each including 5 secondary periods, we explored models with all the following sources of variation for capture probability: sex, time (secondary sampling occasion), trap response (differing capture probabilities depending on whether the animal had been previously caught during this primary period) and heterogeneity (2-point mixture models of Norris and Pollock 1996 and Pledger 2000). Primary periods 9-12 included only 2 secondary occasions each, so for these periods, models considered sex and time, but not trap response or heterogeneity, as potential sources of variation in capture probability. We had no specific predictions for sampling occasion or individual heterogeneity, other than these sources of variation should be important to capture probability. We were uncertain about whether sex-specific variation in detection probability would exist, but if it did, we predicted greater capture probabilities for males (as males typically show more movement, e.g., Coffman et al. 2001). We predicted no behavioral trap response.
Model selection for temporary emigration models used a very general capture probability model structure that included all four sources of variation as additive effects. Temporary emigration modeling included Markovian, random, and no temporary emigration, as well as occasion-specificity. We also fit models that included non-zero temporary emigration for the control grids and pre-treatment period for treated grids, with post-treatment emigration constrained to 0. We had no specific predictions about existence or type of temporary emigration.
Model selection was based primarily on Akaike’s Information Criterion (AIC; Akaike 1973, Burnham and Anderson 2002), but also on considerations of consistency of estimates among the different grids and the existence of boundary estimates (estimates of 0 or 1). For example, if low-AIC models suggested sex-specific capture probabilities on only 2 grid pairs, and if the capture probability estimate was higher for males than females on one grid pair and lower for males on the other, then we would select a model with no sex-specificity of capture probabilities. We would view these overall results as not providing strong evidence of a sex effect. The models for capture probability and temporary emigration selected during this stage of the analysis were maintained throughout the subsequent modeling focused on treatment effects.
Results
The low-AIC models for capture probability all included additive effects of occasion (primary sample period) and individual heterogeneity. Model selection results for sex effects on capture probability were inconsistent across grid pairs. Estimates of capture probability were greater for females than males on one grid pair, greater for males on the other three, and differences were very small on all four grid pairs. Results for trap response were similarly inconsistent, with estimates indicating a trap-happy response on one grid pair, a trap-shy response on the other three, and being very small (negligible) on two grid pairs. We did not include sex or behavioral response effects on capture probability in subsequent modeling, instead using a time plus heterogeneity (2-point mixture, Pledger 2000) structure on capture probability for all four grid pairs. The capture probability notation for all modeling was , where the denotes the occasion-specific heterogeneity parameter specifying the 2-point mixture. For primary occasions 9-12, each characterized by only 2 secondary samples, capture probability was modeled as the same for secondary samples 1 and 2 but varying across primary occasions.
Model selection results for temporary emigration favored models with no temporary emigration for three of the four grid pairs. Only one grid pair showed some evidence of non-negligible temporary emigration, but nothing about the location of this grid pair suggested any reason for modeling it as different from the other three. All subsequent modeling was thus based on a model with no temporary emigration for all four grid pairs.
Appendix B. Treatment Effects of Habitat Loss
Methods
We reasoned that effects of habitat loss (e.g., animals departing the grid because of the sudden reduction in habitat) would be most readily seen immediately following the treatment, before subsequent effects of movement to or from the 2 remaining patches. Thus, we estimated for both treated and control grids the difference between population growth rate during the interval between primary periods 4 and 5 within which the treatment application occurred, and growth rate for the interval just before this application, . If habitat loss on the treated grids resulted in a substantial loss of animals, then we predicted that this difference, , should be smaller for treatment grids than for control grids, because losses attributable to reduced habitat should reduce on treatment grids.
Regardless of any effects of habitat loss, we also expected treatment to affect rates of movement. Normal rates of movement would be expected prior to the treatment application (plowing) followed by reduced movement after application. Treatment applications occurred just a few days before primary period 5, so movement reduction should not be very significant or important for , especially relative to movement occurring before treatment. For this reason, any consistent difference of between treatment and control grids should primarily reflect effects of habitat loss.
Results
We computed the difference, , for both sexes for all 4 grid pairs. On grid 1, this difference was smaller for both males and females on treatment grids, but for grids 2, 3 and 4 the difference was larger for males and females on treatment grids. This small analysis thus provided no consistent evidence that habitat loss produced an immediate reduction in population growth.
Conclusions
Despite our selection of a fragmentation treatment designed to reduce animal movement, we recognize that it produced some habitat loss as well. We believed that any effects of habitat loss would be very small, and our analysis here supported that belief. In retrospect, our overall results also support this belief (see main text DISCUSSION). Nonetheless, we recognize that our evidence of fragmentation effects on meadow vole population dynamics may result from mechanisms other than reduced movement, including habitat loss.
Appendix C. Generating Fragmentation Predictions
Apparent Survival
The complement of apparent adult survival rate as estimated by capture-recapture can be viewed as the result of 2 different processes, death and permanent emigration out of the study system. We conceptualize these as 2 competing risks (sensu Chiang 1968), such that when an animal dies on the study grid, it is no longer available for permanent emigration, and when an animal permanently emigrates, death on the grid is no longer possible. This perspective allows us to view apparent survival as the product of 2 rate parameters:
probability of an adult of sex s surviving death on grid g during any 1-month interval between occasions t and t+1, in the hypothetical absence of any permanent emigration.
probability of an adult of sex s not permanently emigrating from grid g during any 1-month interval between occasions t and t +1, in the hypothetical absence of any grid death.
Apparent survival rate, as estimated via capture-recapture methods, is the product of these 2 parameters:
A reduction in movement should not affect , but should increase , and thus . Estimation of these components is described in Appendix D. We note that the same decomposition and associated arguments correspond to apparent juvenile survival as well.
Recruitment Rate
Monthly recruitment rate, , as estimated via Pradel’s (1996) temporal symmetry modeling, focuses on new adults of sex s and can be decomposed into additive components associated with in situ reproduction and immigration (Nichols and Pollock 1990):
monthly reproductive recruitment rate; expected number of new adult animals of sex s in the sampled population of grid g at sampling occasion t+1 that arise from local reproduction, per adult of sex s in the sampled population at occasion t, scaled to a 1-month interval.
monthly immigration rate; expected number of new adult immigrant animals of sex s in the sampled population of grid g at sampling occasion t+1, per adult of sex s in the sampled population at occasion t, scaled to a 1-month interval. The recruitment rate estimated in our capture-recapture modeling can thus be written as the sum of these 2 components:
isolating immigration rate ) as a parameter affected directly by fragmentation.
In order to isolate fragmentation effects on the other component of recruitment, we can further decompose reproductive recruitment, , into multiplicative components:
number of new young of sex s in the population of grid g at sampling occasion t per adult of sex s on grid g at t.
apparent monthly survival rate of juveniles; probability that a juvenile animal on grid g of sex s survives a month during the period Δt.
So we can write reproductive recruitment as:
As with adult survival, we can then decompose juvenile survival into multiplicative components for loss through mortality and permanent emigration:
probability of a juvenile of sex s surviving death on grid g during any 1-month interval between occasions t and t+1, in the hypothetical absence of any permanent emigration.
probability of a juvenile of sex s not permanently emigrating from grid g during any 1-month interval between occasions t and t +1, in the hypothetical absence of any grid death.
Reproductive recruitment can thus be written as:
The purpose of this decomposition is to isolate the effects of fragmentation on movement, , for the purpose of predicting effects on population growth rate. Estimation of these components is described in Appendix D.
Population Growth Rate
Based on the decomposition of rates of apparent survival and recruitment (C1-C4), we can rewrite monthly population growth rate as the sum of 3 terms:
The overall effect of fragmentation on population growth rate depends minimally on the relative effects of changes in dispersal on rates of adult permanent emigration (1-), juvenile permanent emigration (1-) and immigration (). In Appendix E, we use expression C5 to develop an overall expression for the change in growth rate expected to accompany specified decreases in movement.
Appendix D. Estimation of Decomposed Parameters
Inference Methods
Estimating recruitment components. Recruitment rate can be decomposed into components associated with in situ reproduction and immigration (Appendix C):
-
= monthly reproductive recruitment rate; expected number of new adult animals of sex s in the sampled population of grid g at sampling occasion t+1 that arise from local reproduction, per adult of sex s in the sampled population at occasion t, scaled to a 1-month interval.
-
= monthly immigration rate; expected number of new adult immigrant animals of sex s in the sampled population of grid g at sampling occasion t+1, per adult of sex s in the sampled population at occasion t, scaled to a 1-month interval.
The reason for this decomposition is that fragmentation is expected to affect the two recruitment components differently. Reduced dispersal should increase and decrease .
We estimated the 2 recruitment components using an approach similar to that outlined in Nichols and Pollock (1990). Inference about requires estimates of both apparent survival rate and abundance for juveniles, as well as abundance for adults. Numbers of juveniles captured were much smaller than adults, precluding estimation of juvenile survival separately for each grid pair. Thus, we used the approach described in the main text to estimate monthly juvenile survival rate for period i (pre-treatment or post-treatment) as , where J. denotes juveniles of both sexes and trt,i denotes all grids of a specific treatment in period i.
Juvenile abundance was estimated using capture probability modeling for closed populations for the entire juvenile data set (grids, occasions within periods, and sexes combined). All models for capture probability were based on the pre-treatment period (occasions 1-4) for all grids combined and the post-treatment period (5-12) separately for all treatment grids and all control grids. Models with and without heterogeneous capture probabilities and trap response were fit. The most appropriate model based on AIC and parameter estimates was a trap response model with no heterogeneity. The estimated initial capture probabilities from this model were then applied to the sex-, occasion-, and grid-specific numbers of juveniles caught to estimate juvenile abundance, .
Based on these survival and abundance estimates for juveniles, we can estimate monthly reproductive recruitment rate as:
Expression (D1) simply estimates monthly reproductive recruitment rate between sampling occasions t and as the total number of juveniles that survive from occasion t to become adults in the population 1 month later, divided by the number of adults present at occasion t.
Immigration rate can then be estimated (based on equation C2) as the difference between the monthly adult population growth rate between occasions t and t+1 and the sum of monthly adult survival rate and monthly reproductive recruitment rate over that same period:
Estimating survival components. The complement of apparent survival rate as estimated by capture-recapture is the result of 2 different processes, death and permanent emigration out of the study system. Based on a competing risk perspective (sensu Chiang 1968) we view apparent adult survival as the product of 2 rate parameters, , the monthly probability of surviving death on the grid between sampling occasions t and t +1 in the hypothetical absence of any permanent emigration, and , the monthly probability of not permanently emigrating from the grid during the interval t to t +1 in the hypothetical absence of any grid death. So . But because these risks are always operating simultaneously in our actual study system, inference about these rate parameters is not straightforward.
We obtained separate estimates of these 2 components ( using an ad hoc nested grid approach reminiscent of that used by Otis et al. (1978) to estimate population density. Each grid was viewed as 3 nested grids, the entire grid of 7 x 15 trapping stations, an interior grid of 5 x 13 stations, and an inner grid of 3 x 11 stations. We computed the area/perimeter ratio for each nested grid h (2.44 for the outer grid [h =3], 1.81 for the middle grid [h =2] and 1.18 for the inner grid [h =1]). We reasoned that the probability of an individual permanently emigrating should decrease as area/perimeter ratio increases, to the point where a very large grid (e.g., covering the state of Maryland) would have negligible (approaching 0) permanent emigration rates, as most individuals would reside far from the perimeter. We thus hypothesized the following relationship between average estimated survival and Ah/Ph:
For each sex we had 8 average adult survival estimates for each of the 3 nested grids using data from the pre-treatment period and used them to estimate and of D3. We then used this expression to compute an expected survival estimate for a very large Ah/Ph ratio (denote as Ah /Ph = 10000), viewing this estimate as approximating the probability of surviving death. So as the probability of permanent emigration becomes vanishingly small, we expect . We can then estimate the complement of permanent emigration for our grids as:
In order to compute we also require estimates of the permanent emigration component for the apparent survival of juveniles. However, we were unable to use the same approach followed for adults, because of the smaller numbers of juveniles marked and recaptured. We thus used an ad hoc approach and assumed that the proportion of losses attributable to permanent emigration was similar for juveniles and adults, i.e.,
leading to the estimators:Survival component estimates were computed using (D5, D6) for each grid, and predictions were then based on means over all 8 grids.
Results
These efforts to decompose estimates of survival and recruitment into components that did, and did not, include emigration and immigration were admittedly ad hoc. In addition, we were forced to combine grids, sexes and sampling occasions within pre- and post-treatment periods in order to obtain adequate sample sizes for some estimates. We also had to assume equal ratios of mortality to permanent emigration probabilities for adults and juveniles. Thus, we did not try to compute variances for these estimates, as we did not believe we could do so reliably. Similarly, we did not use the estimates for our primary inferences about treatment effects. Instead, we restricted use of these estimates of decomposed parameters to development of predictions and to post hoc explanations for our primary results.
Survival components. The nested sub-grid approach using data from all 8 grids yielded estimates for that were not much larger than overall survival (Table D1). Use of these estimates with overall survival estimated for each grid separately (Eq. D4) produced mean estimates for the probability of not permanently emigrating that were very close to 1. These estimates indicate that mortality dominated rates of loss and was much more important to apparent survival than permanent emigration over the period of estimation.
Table D1.
Estimated monthly probabilities of surviving death (), permanent emigration (), and both sources of loss ( ) for adult female and male meadow voles on the 8 experimental grids, during the pretreatment sampling occasions. Also presented are estimates of the average proportion of total losses attributable to permanent emigration (far right column).Table D2.
Estimated monthly probabilities of surviving death (), permanent emigration (), and both sources of loss ( ) for juvenile meadow voles (sexes combined) on the 8 experimental grids, during the pretreatment sampling occasions.Recruitment components. Recruitment rate associated with reproduction was estimated for each sex, grid, and sampling occasion separately. Mean estimates over sampling occasions and grids were small for both females and males (Table D3). The resulting estimates were then used with overall recruitment estimates for each sex, grid, and sampling occasion to estimate recruitment rate from immigration, . The fraction of recruitment attributable to immigration was estimated to be approximately 90% for both males and females (Table D3).
Table D3.
Estimated monthly recruitment rates attributable to in situ reproduction () and immigration () for female and male meadow voles on the 8 experimental grids, during the pretreatment sampling occasions. Also presented are estimates of the overall (both sources) recruitment rate and the fraction of overall recruitment attributable to immigration.Discussion
Our estimators of these component parameters that comprise recruitment and apparent adult and juvenile survival were ad hoc, and we did not know how likely they were to perform well. We did not even attempt to compute variances for these estimates. Instead, we used them solely for the purpose of making ball-park predictions for the effects of fragmentation on adult survival, recruitment, and population growth rate.
We have a number of concerns about the estimates presented in Tables D1, D2, and D3, and will briefly note a couple of these here. Our decomposition of apparent survival rates of adults and young used the nested grid approach described above. Although we view this approach as conceptually sound, we also realize that the variation among nested grids in area/perimeter was relatively small (because we had no very large grids) and focused on one tail of the area/perimeter distribution. We are thus estimating the upper asymptote of a relationship based on 3 values at the opposite end of the survival vs. area/perimeter plot. In addition, it may also be that the relationship between dispersal and area/perimeter ratio differs for short-distance versus long-distance dispersers, and our analysis likely focused on variation in short-distance dispersal.
Our approach to estimating recruitment components may have underestimated the reproductive component and overestimated immigration. The reproductive component was based on juvenile animals present on the grids during one week and still present as adult recruits approximately 8 weeks later. All animals classified as juveniles in one primary sampling occasion and recaptured in the next sampling occasion had transitioned to adult weight. However, it is likely that some juveniles not of trappable size in one primary occasion could grow into adults by the next primary occasion, and thus appear in our estimates as immigrants. If this is a common occurrence, then it would lead to underestimation of reproductive recruitment and overestimation of immigration.
In sum, we suspect that adult and juvenile permanent emigration have been underestimated and immigration has been overestimated. These biases will likely translate to predicted changes in population growth rate that are too small (Appendix E), causing us to consider other information for prediction as well.
Appendix E. Predicting Effects of Fragmentation on Population Growth Rate
Appendix F. Summaries and Plots of Key Demographic Parameter Estimates for Fragmented and Control Meadow Vole Populations at Patuxent Wildlife Research Center, September 1991 – August 1993.
Table F1.
Estimated movement probabilities and associated standard errors, for adult female (F) and male (M) meadow voles during pre-treatment and post-treatment periods on both fragmented (f) and control (c) grids.Table F2.
Average estimated monthly apparent survival probabilities and associated standard errors, for adult female (F) and male (M) meadow voles during pre-treatment and post-treatment periods on both fragmented (f) and control (c) grids.Table F4.
Average estimated monthly recruitment rates and associated standard errors, for adult female (F) and male (M) meadow voles during pre-treatment and post-treatment periods on both fragmented (f) and control (c) grids.Table F5.
Average estimated monthly rates of population growth and associated standard errors, for adult female (F) and male (M) meadow voles during pre-treatment and post-treatment periods on both fragmented (f) and control (c) grids.Figure Captions
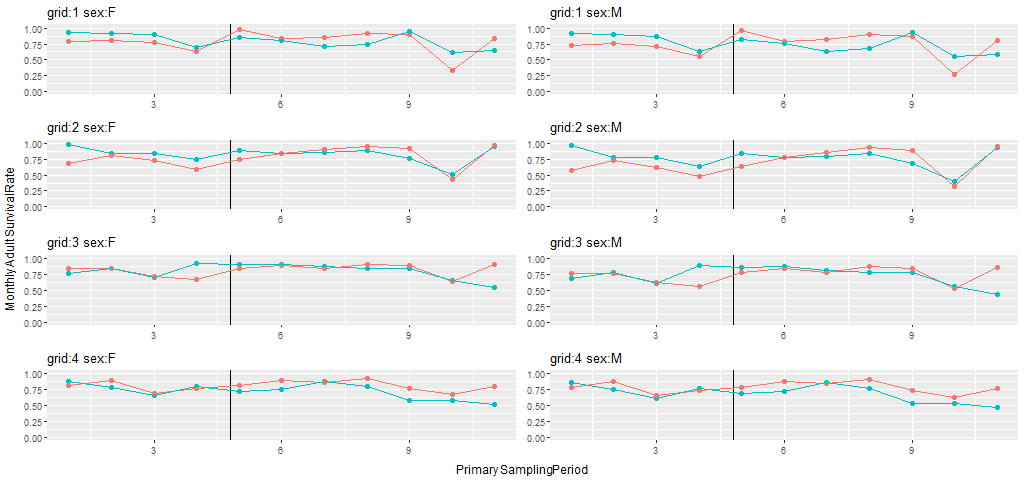
Point estimates of monthly apparent adult survival rates for female (F) and male (M) meadow voles on fragmented (red plots) and control (blue plots) grids at Patuxent Wildlife Research Center, September 23, 1991 – August 24, 1993. The black vertical line indicates time of the treatment. Male and female plots are based on an additive model and show the same fluctuations with fairly similar values.
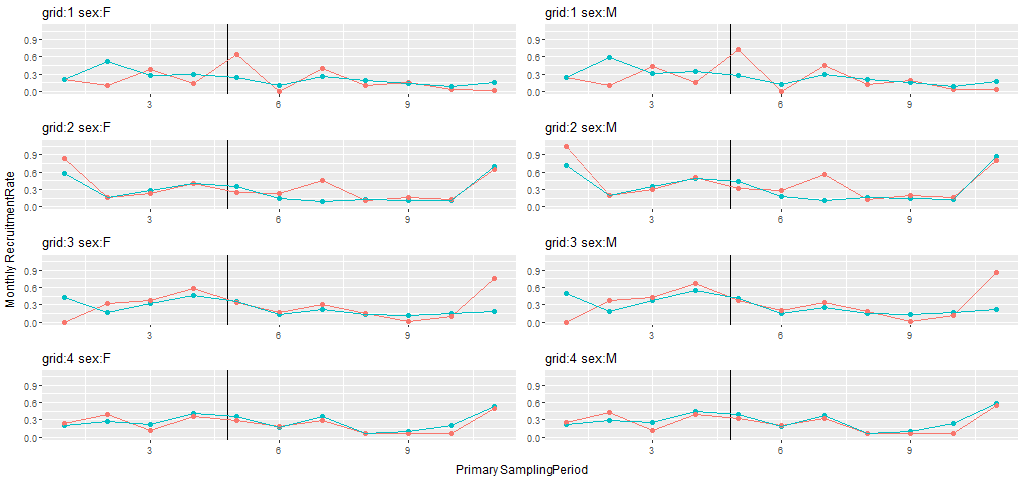
Point estimates of monthly recruitment rates for female (F) and male (M) meadow voles on fragmented (red plots) and control (blue plots) grids at Patuxent Wildlife Research Center, September 23, 1991 – August 24, 1993. The black vertical line indicates time of the treatment. Male and female plots are based on an additive model and show the same fluctuations with fairly similar values.
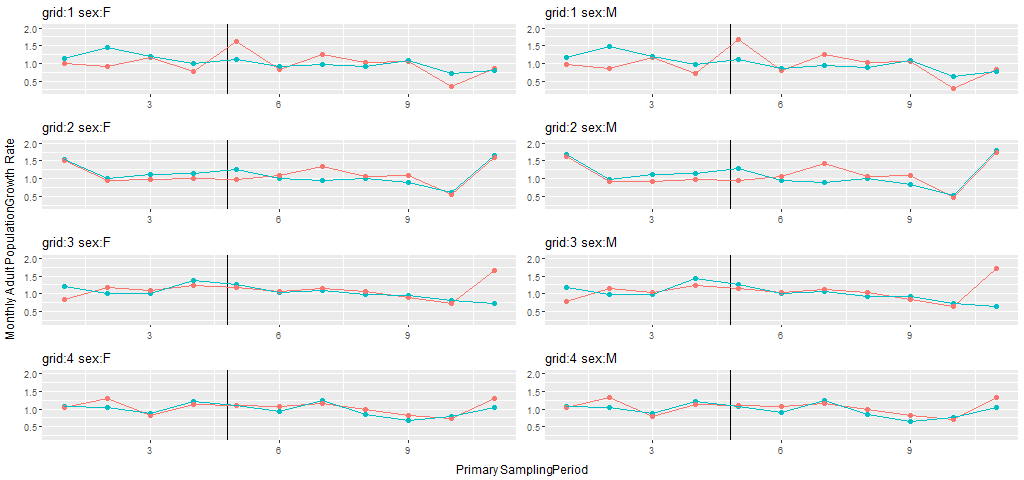
Point estimates of monthly adult population growth rates for female (F) and male (M) meadow voles on fragmented (red plots) and control (blue plots) grids at Patuxent Wildlife Research Center, September 23, 1991 – August 24, 1993. The black vertical line indicates time of the treatment. Male and female plots are based on an additive model and show the same fluctuations with fairly similar values.
Authors
Disclaimers
Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Although this information product, for the most part, is in the public domain, it also may contain copyrighted materials as noted in the text. Permission to reproduce copyrighted items must be secured from the copyright owner.
Suggested Citation
Nichols, J.D., Hines, J.E., Hinz, R.L., and Hinz, J., 2026, Fragmentation as a population rate-changer: A field experiment: Ecology, v. 107, no. 3, e70327, 19 p., https://doi.org/10.1002/ecy.70327.
| Publication type | Article |
|---|---|
| Publication Subtype | Journal Article |
| Title | Fragmentation as a population rate-changer: A field experiment |
| Series title | Ecology |
| DOI | 10.1002/ecy.70327 |
| Volume | 107 |
| Issue | 3 |
| Publication Date | March 05, 2026 |
| Year Published | 2026 |
| Language | English |
| Publisher | Ecological Society of America |
| Contributing office(s) | Eastern Ecological Science Center |
| Description | e70327, 19 p. |


