Protocols for Collecting and Processing Macroinvertebrates from the Benthos and Water Column in Depressional Wetlands
Links
- Document: Report (4.02 MB pdf) , HTML , XML
- Download citation as: RIS | Dublin Core
Acknowledgments
All authors have made substantial contributions to this work. These protocols have been adapted and improved over the past 15 years. We thank Adam Janke, Alan Afton, Mark Hanson, Ray Norgaard, David Mushet, Allen Rutherford, and Carl Isaacson for their contributions to protocol development.
These protocols were funded by the Minnesota Environment and Natural Resources Trust Fund, the U.S. Geological Survey, and the Minnesota Department of Natural Resources.
This report was kindly peer reviewed and improved by Brian Tangen and Jennifer Sauer from the U.S. Geological Survey.
Abstract
Freshwater aquatic macroinvertebrates are key links in food webs and nutrient cycles, and thus often serve as biological indicators of ecosystem health. Macroinvertebrate investigations in research and monitoring require consistent and reliable field and laboratory procedures. Comprehensive standard operating procedures for sampling macroinvertebrates from depressional wetlands, which can range from riverine floodplain lakes to wetlands of any size and hydrologic regime, remain relatively sparse. This report provides step-by-step protocols for efficient use of time and resources while collecting and processing aquatic macroinvertebrate samples; for example, a single wetland can typically be field surveyed in less than 1 hour, and the samples can be processed in the laboratory in less than 2 hours. Samples can be collected from inside a motorboat or canoe or while wading. This procedures manual describes dip netting to collect macroinvertebrates from the wetland bottom and water column separately to facilitate investigations of habitat use by species occupying different areas of the wetland. This report also provides descriptive supplemental materials and data sheets to assist with the preparation of survey maps, the acquisition of field and laboratory equipment, and the calculation of macroinvertebrate densities from the wetland bottom and water column. These procedures can be applied to most macroinvertebrate species and communities that inhabit a variety of wetland sizes and types. Uses and applications can range from elementary and secondary environmental education to rigorous scientific evaluations of community abundance, diversity, distribution, or species-habitat relations.
Introduction
The purpose of this standard operating procedure (SOP) is to outline and describe step-by-step instructions for efficiently collecting, preserving, and processing aquatic macroinvertebrate samples, and for subsequently calculating species counts and densities. Although several related field and laboratory manuals are available (Barbour and others, 1999; Moulton and others, 2002; DiFranco, 2014; Minnesota Pollution Control Agency, 2014), this SOP was developed to provide comprehensive and adaptable procedures that can quantify macroinvertebrates in depressional wetlands. Depressional wetlands form in topographic depressions with closed contours on at least three sides that collect standing water for various hydroperiods. Depressional wetlands occur globally across all biomes in a variety of wetland types, such as but not limited to vernal pools, freshwater marshes, semipermanent wetlands, permanent wetlands, riverine floodplain wetlands, and shallow lakes.
Related SOPs often target wadable streams and rivers with little organic matter in the benthos (the bottom of the aquatic system), resulting in methods that are commonly ineffective in systems with standing water and soft, organic sediments. Other SOPs describe passive sampling techniques, such as passive activity traps suspended in the wetland overnight (Hanson and others, 2000). Activity traps provide relative abundance estimates and minimize sample debris, but also restrict quantifying macroinvertebrate abundance and increase sample time and travel costs, and can be biased by time of day, weather, water temperature, and predators. Some procedures detail intensive, active sampling techniques that collect large quantities of organic matter (for example, using dredge samplers or extensive dip-netting surveys), which require substantial resources for sample preservation and laboratory processing.
Several factors should be considered before the selection and use of an SOP, including the type of data required (that is, quantitative versus qualitative), the project’s purpose (for example, environmental education versus rigorous research), the project’s scope (for example, sampling one system intensively versus less intensive sampling for multisystem comparison), and the availability of resources (such as time, personnel, storage space for samples, and preservation chemicals). For further guidance on selecting procedures for macroinvertebrate investigations, see Turner and Trexler (1997), Anderson and others (2013), and Meyer and others (2013); however, this SOP is highly adaptable for the user’s needs regarding all data types, project purpose and scope, and limited resources. As an example, the SOP has been effectively applied to rigorous scientific research on invertebrate and habitat associations (Larson and others, 2022). Many steps in this SOP instruct the user to choose between options to best suit their circumstances.
This SOP improves upon several published procedures for the sampling of amphipods and other macroinvertebrates in depressional wetlands of the United States (Anteau, 2006; Anteau and Afton, 2008; Anteau and others, 2011; Janke, 2016). Revisions were derived from the experiences of the authors, who trialed the various methods within several hundred depressional wetland systems throughout the Prairie Pothole Region of the United States. Major modifications from previous studies that were adopted within this SOP include (1) an optional stratified random sampling design to capture near-shore and offshore habitats, (2) instructions for locating parallel sample transects instead of radial transects to evenly disperse survey stations throughout the wetland, (3) instructions for differing sweep net techniques for improved quantification of invertebrates, (4) an optional vertical sweep to capture and quantify macroinvertebrates occupying the water column only, and (5) instructions for sampling a smaller and standardized benthos area (0.5 meter [m]) to increase reproducibility of sample technique, as well as substantially reduce the amount of organic material to store and preserve.
Field Protocols
This section of the SOP describes processes for selecting survey stations, sampling aquatic macroinvertebrates in wetlands, and preserving samples containing specimens and wetland debris. For field supply information, see appendixes 1 and 2. Descriptive data sheets and step-by-step protocols coinciding with field processes are provided in appendixes 3–5.
Selecting Survey Stations
At each wetland, samples are collected from several stratified-random survey stations along transects within near-shore zones (less than [<] 5 m) and likely open-water zones (<50 m from shore). This stratified random sampling design is optimal if the wetland is greater than (>) 1 hectare and >1.5 m deep because there will be zones of vegetation and open water to target. If wetlands are small with little change in water depth or vegetation community, samples could simply be taken randomly, ignoring stratum designations.
Following methodology from Anteau and Afton (2008) and Anteau and others (2011), macroinvertebrates are collected from eight locations dispersed throughout the wetland (fig. 1); however, the SOP user can choose the number of survey stations for their work dependent on wetland size or study objectives. Survey stations are predetermined to ensure randomization of selection. Stations use the emergent vegetation zone as the spatial reference for survey point spacing along transects, but details in appendix 3 describe solutions when emergent cover is lacking or covering the wetland. The stations are uniquely identified and mapped using ArcMap software (Esri, Redlands, California) and programmed into a Global Positioning System device for field navigation. This selection process can be completed manually using step-by-step instructions (appendix 3).
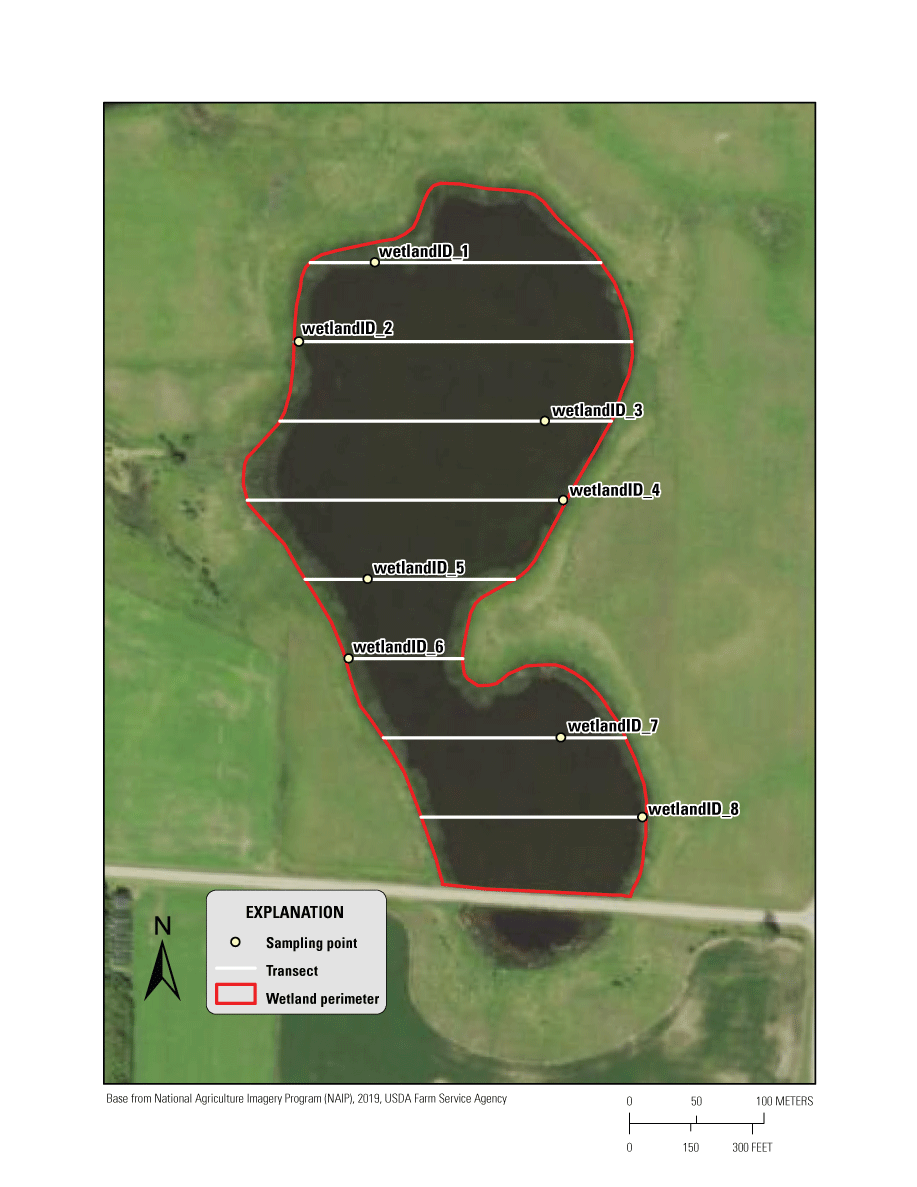
An example of a macroinvertebrate survey using a stratified random sampling design for depressional wetlands. Survey points (uniquely labeled with wetland name and survey number; for example, “wetlandID_1”) fall along the transects within a stratum.
Invertebrate Sampling
Most surveys should be completed from inside a boat because depressional wetlands frequently contain soft benthic zones where sediments are easily disturbed and depths that cannot be sampled via wading. Wetlands with hard bottoms and shallow depths (less than or equal to 1 m) throughout could be sampled via wading if care is taken to not disturb sediments before collecting samples. Samples are collected using D-frame dip nets constructed with a mesh that is small enough to contain macroinvertebrates but large enough to allow some organic matter and sediment to filter out. Depending on the body size of targeted macroinvertebrates, suitable mesh size can range from 500 to 1,200 micrometers. Sampling depths should be limited to the length of the dip-net handle to reduce error to invertebrate quantification from unknown sweep length. Handle extensions could be added to the dip net to sample deeper waters.
Samples from the benthic and water column zones can be collected and stored separately, which requires sweeps using two nets at each survey station. Both nets should be the same size (handle length of at least 1.5-m and frame dimensions of about a 0.3-m width and 0.3-m height), but the water column net (also referred to as the “modified” net) contains a frame bent at a 90-degree angle relative to the net handle (figs. 2 and 3). This modification allows for the collection of macroinvertebrates from a vertical path extending throughout the entire water column zone. Both net handles must be marked with depth increments so that water depth can be measured while sampling. Samples from each survey station can be stored in separate containers or composited into one bottle, depending on whether the research objectives are at the survey station level or the wetland level. Samples should be immediately preserved with 95-percent ethanol to minimize predation of specimens and decay during storage.

Technician collecting a macroinvertebrate sample from the water column using a modified dip net where the frame was bent at a 90-degree angle relative to the net handle.
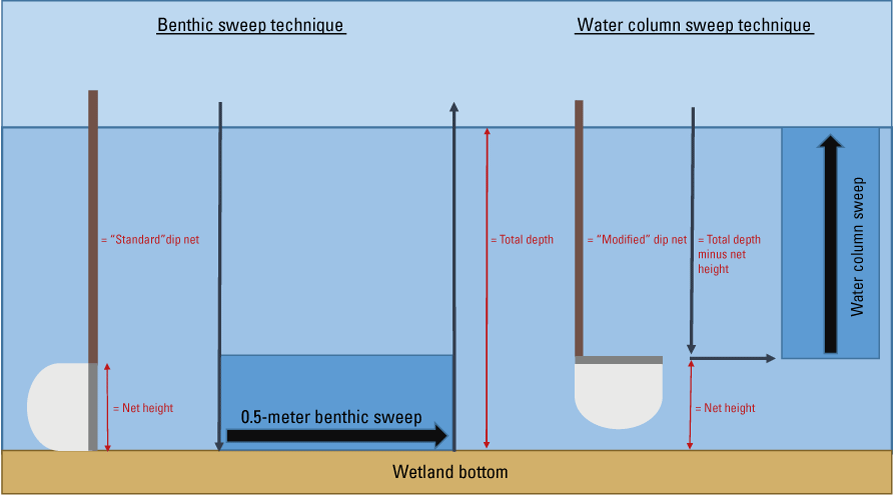
Benthic and water column sweeping techniques using dip nets in depressional wetlands. In a benthic sweep, the net is plunged vertically into the top layers of wetland benthos, swept for a standard 0.5-meter distance, and then pulled up vertically to sample only the benthos. In a water column sweep, a modified dip net (where the net is angled 90 degrees from the handle) is plunged vertically to the benthos, minus the distance of the net height, and slowly towed vertically to the surface to capture free-floating macroinvertebrates within the water column.
Step-by-step procedures coinciding with the field sampling and preservation techniques listed previously are described in appendix 4. Field personnel should complete two data sheets while at the wetland: (1) a site information data sheet for key information about the wetland and (2) a macroinvertebrate sampling data sheet to be filled out while sampling at each survey station (appendix 5).
Laboratory Protocols
This section of the SOP describes the process for organizing samples, decanting samples to prevent decay, picking macroinvertebrates, and preserving specimens for long-term (>1 year) archiving. Laboratory personnel should consult the laboratory supply list (appendix 6) and complete three data sheets to assist with these processes (appendix 7).
Sample Organization and Preservation During Storage
Upon returning to the laboratory, check field data sheets to ensure all sample bottles are accounted for. Then, record all samples on the macroinvertebrate sample inventory log (appendix 7). All samples should be stored in a flammable safety cabinet because the ethanol used for preservation is flammable. Organize sample bottles in the cabinet according to the date they were collected to ensure the oldest samples are processed first.
To prevent decomposition, samples must be monitored and occasionally decanted by carefully removing old ethanol and refreshing the sample with new 95-percent ethanol. All samples should be inspected and decanted (only if necessary) within 72 hours of field collection and every 2–3 weeks thereafter until processed. First, put on gloves and safety goggles, carefully open the sample bottle, and waft the sample’s smells towards your nose. If the sample smells like alcohol, no decanting is required. If the sample smells slightly organic, it must be decanted. If the smell of decaying organic matter overtakes the smell of alcohol, the sample may be compromised. If decanting is needed, carefully decant the ethanol through a 500-micrometer sieve held over a labeled ethanol waste container. Backwash the sieve over the sample bottle with 95-percent ethanol to collect any organisms or debris on the sieve. Inspect the sieve with a 3× magnifying light to ensure no macroinvertebrates remain. Pour fresh 95-percent ethanol into the sample bottle following a 1 part sample to 2 parts ethanol ratio. Label the bottle with decant date and fill out the macroinvertebrate sample decanting form (appendix 7) for organization.
Sample Picking
Macroinvertebrates are ideally “picked” from the sample containing debris under good lighting, ventilation, and microscopy in the laboratory to maximize detection of all species. Alternatively, macroinvertebrates could be processed at the wetland in sunlight with a hand magnifier, especially for environmental education applications or rapid assessments of select species. The samples could be picked in entirety or subsampled, depending on user preference for time allotted per sample. For example, if benthic samples or water column samples are large and have a picking time greater than 2 hours, macroinvertebrates could be quantitatively subsampled using a randomized fixed count of 25 percent of the entire sample volume or a fixed number of macroinvertebrate specimens per sample (determined by the SOP user). A minimum of 25 percent of the sample volume must be picked to increase the likelihood that the subsample is representative of the overall sample.
Guidance for building a subsampling apparatus is provided in appendix 8, and all subsampling and picking is completed using detailed instructions described in appendix 9 and depicted in figure 4A–D. Macroinvertebrates can be identified and quantified during the picking process or preserved in 70-percent ethanol and processed later.
Identifying Macroinvertebrates
Invertebrates may be identified to any level (family, genus, or species) using an appropriate microscope and identification key. If desired, body length measurements can be collected simultaneously for the approximation of organism biomass, as described in Benke and others (1999). All identification and length data should be recorded in the macroinvertebrate identification data sheet (appendix 5).

The four subsampling steps in processing macroinvertebrates in the laboratory. A, the composited invertebrate sample, which has been evenly mixed and poured over a squared subsampling apparatus (see appendix 8 for details on building this apparatus); B, a gridded frame overlaying the subsampling apparatus to divide the sample into four equal parts; C, one grid (or 25 percent of the sample) processed in entirety for macroinvertebrates. The user may choose to sample a minimum of one grid or as many as four grids, depending on time and research objectives; D, a part of contents from one grid, which are placed in a white tray under high lighting and 3× magnification for picking invertebrates from the detritus.
Calculating Macroinvertebrate Densities
Invertebrate counts (the number of individuals) and volumetric densities (the number of individuals per cubic meter) can be calculated for the benthos and (or) water column because samples are collected using a dip net with known net and sweep areas. These calculations can be applied in Microsoft Excel, R, or similar software. The steps to calculate volumetric and areal densities for the benthos and water column are shown in figure 5.
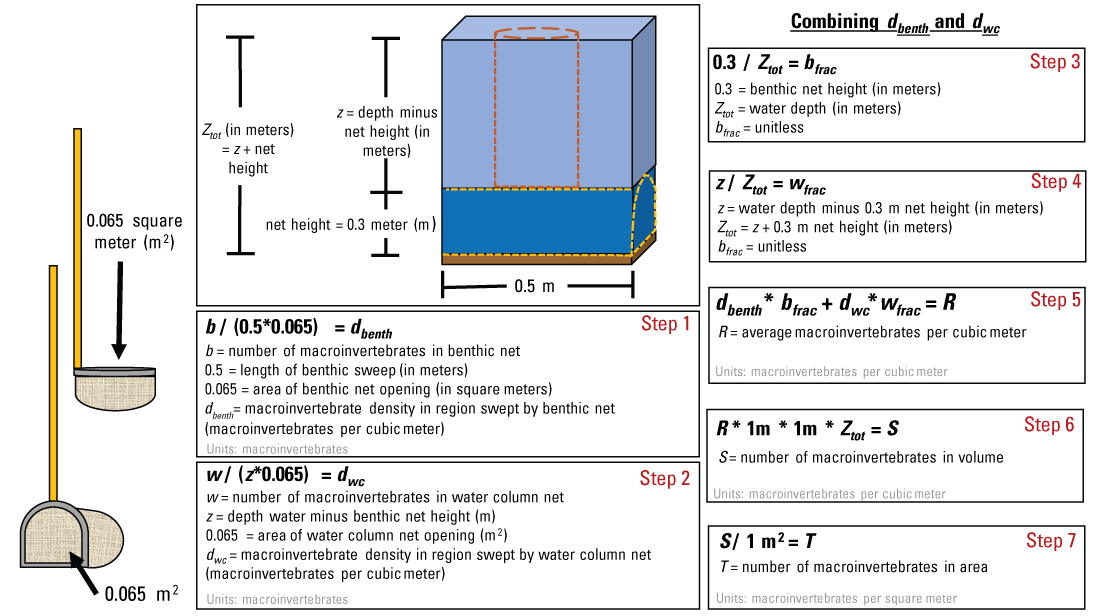
The steps for calculating benthic (dbenth) and water column (dwc) macroinvertebrate densities collected from dip nets in depressional wetlands. To calculate volumetric density, proceed from steps 1 to 6. To calculate areal density, proceed from steps 1 to 7.
Summary
Aquatic macroinvertebrate investigations are often integrated into wetland research and monitoring and require consistent and reliable field and laboratory procedures. This SOP was developed to meet a need for comprehensive and standardized procedures that can be applied to a wide range of depressional wetland types. These procedures improved upon previously published works and are designed to be descriptive but adaptable to accommodate a wide range of project objectives. Following these procedures, a wetland can typically be field surveyed at the wetland and processed in the laboratory in a matter of hours. The use of a two different dip nets allows for investigations within either the benthos or the water column zones. The supplemental materials assist with the preparation of field sampling, laboratory processing, and the calculation of macroinvertebrate abundances. Collectively, these procedures can be applied to a wide variety of macroinvertebrate investigations in both educational and research-based settings.
References Cited
Anderson, J.T., Zilli, F.L., Montalto, L., Marchese, M.R., McKinney, M., and Park, Y.L., 2013, Sampling and processing aquatic and terrestrial invertebrates in wetlands: Wetland Techniques, v. 2, p. 143–195. [Also available at https://doi.org/10.1007/978-94-007-6931-1_5.]
Anteau, M.J., and Afton, A.D., 2008, Amphipod densities and indices of wetland quality across the upper-Midwest, USA: Wetlands, v. 28, no. 1, p. 184–196. [Also available at https://doi.org/10.1672/07-53.1.]
Anteau, M.J., Afton, A.D., Anteau, A.C.E., and Moser, E.B., 2011, Fish and land use influence Gammarus lacustris and Hyalella azteca (Amphipoda) densities in large wetlands across the upper Midwest: Hydrobiologia, v. 664, no. 1, p. 69–80. [Also available at https://doi.org/10.1007/s10750-010-0583-2.]
Benke, A.C., Huryn, A.D., Smock, L.A., and Wallace, J.B., 1999, Length-mass relationships for freshwater macroinvertebrates in North America with particular reference to the southeastern United States: Freshwater Science, v. 18, no. 3, p. 308–343. [Also available at https://doi.org/10.2307/1468447.]
Hanson, M.A., Roy, C.C., Euliss, N.H., Zimmer, K.D., Riggs, M.R., and Butler, M.G., 2000, A surface-associated activity trap for capturing water-surface and aquatic invertebrates in wetlands: Wetlands, v. 20, no. 1, p. 205–212. [Also available at https://doi.org/10.1672/0277-5212(2000)020[0205:ASAATF]2.0.CO;2.]
Larson, D.M., DeJong, D., Anteau, M.J., Fitzpatrick, M., Keith, B., Schilling, E.G., and Thoele, B., 2022, High abundance of a single taxon (amphipods) predicts aquatic macrophyte biodiversity in prairie wetlands. Biodiversity and Conservation [original paper], 22 p., accessed February 2022 at https://doi.org/10.1007/s10531-022-02379-9.]
Meyer, M.D., Davis, C.A., and Bidwell, J.R., 2013, Assessment of two methods for sampling invertebrates in shallow vegetated wetlands: Wetlands, v. 33, no. 6, p. 1063–1073. [Also available at https://doi.org/10.1007/s13157-013-0462-5.]
Moulton, S.R., II, Kennen, J.G., Goldstein, R.M., and Hambrook, J.A., 2002, Revised protocols for sampling algal, invertebrate, and fish communities as part of the National Water-Quality Assessment Program: U.S. Geological Survey Open-File Report 2002–150. [Also available at https://doi.org/10.3133/ofr2002150.]
Turner, A.M., and Trexler, J.C., 1997, Sampling aquatic invertebrates from marshes—Evaluating the options: Journal of the North American Benthological Society, v. 16, no. 3, p. 694–709. [Also available at https://doi.org/10.2307/1468154.]
Glossary
- amphipod
Aquatic crustacean in the order Amphipoda.
- benthic/benthos
Refers to the bottom; for example, the benthic zone in a wetland system is at or near the sediment surface.
- decant
Slowly pour liquid from container without disturbing the underlying solids.
- macroinvertebrates
Animals lacking a backbone that can be seen with the naked eye, such as insects, mollusks, worms, and crustaceans.
- picking
The process of removing macroinvertebrates from sample debris.
- sieve
Apparatus consisting of a wire mesh and solid frame that allows for the separation of smaller particles (for example, sediment) from larger particles (for example, macroinvertebrates).
- survey station
Predetermined location in wetland at which the macroinvertebrate sample is collected.
- sweep
A steady, smooth movement; in this report, referring to the movement of passing a dip net across the wetland bottom or through the water column to collect invertebrates.
- transect
Straight line extending across the lake or wetland surface, along which survey stations are positioned for collecting invertebrates.
- wetland
Topographic depression containing a wetland system.
Appendix 1. Field Supplies List
-
• 1 or 2 500–1,200-micrometer dip nets (see appendix 2 on acquiring a “modified” net, as shown in fig. 2)
-
• Field data sheets (appendix 5)
-
• 225–1,000-milliliter sample bottles (small bottles for individual survey station samples; large bottles for composite samples)
-
• Storage tub(s)
-
• 95-percent ethanol (for sample preservation)
-
• Internal labels
-
• External labels
-
• Clear packing tape
-
• Gloves
-
• Global Positioning System with preprogrammed survey station points
-
• Wetland map
-
• Clipboard
-
• Ethanol-resistant markers
-
• Pencils
-
• Flagging tape
-
• Waders
-
• U.S. Coast Guard-approved inherently buoyant personal flotation devices (life jackets)
-
• Canoe/sport boat/motor (if needed)
-
• Paddles
Appendix 2. Acquiring a Modified Dip Net
Appendix 3. Detailed Instructions for Generating Survey Stations
-
1. Using ArcMap software, extract or construct the open-water shapefile corresponding to the targeted study wetland. This shapefile may be extracted from existing publicly accessible wetlands data such as the National Wetlands Inventory data layer (U.S. Fish and Wildlife Service, 2020), or it may be created manually by constructing a polygon in reference to aerial imagery. For either method, select data or imagery collected as close to your sampling time as possible to ensure desktop-sourced wetland boundaries are representative of current conditions.
-
2. Navigate to the “create fishnet” tool (“Toolboxes” → “Data Management Tools” → “Sampling” → “Create Fishnet”). Using the measuring tool, determine whether or not the distance between the northern and southern shoreline is longer than the distance between the eastern and western shoreline; if longer, transects should run in an east–west direction (n rows = 9, n columns = 1, where n is number), and if shorter should run in a north–south direction (n rows = 1, n columns = 9).
-
3. Use the “clip” tool (“Toolboxes” → “Analysis Tools” → “Extract” → “Clip”) to clip the transects to the wetland outline.
-
4. Each transect will contain one survey station. The survey stations are in two strata: either less than 5 meters (m) of the innermost ring of emergent vegetation near shore or 50 m from the innermost ring of emergent vegetation (see example in fig. 1). If the wetland is small, it is possible to modify the strata according to proportional distances between the vegetation zone and wetland center. Use a random-selection technique (such as a coin flip or a random number generator) to select the starting transect and shoreline to alternate between strata.
-
5. Create survey point shapefiles by creating a new feature class for point features and manually placing them in the appropriate positions along the transects.
-
6. Assign a unique identifier to each survey station that includes the wetland name and station number.
-
7. Print a copy of the map, and load survey station shapefiles onto a Global Positioning System as waypoints, ensuring survey points are clearly labeled with a unique identifier on both.
Reference Cited
U.S. Fish and Wildlife Service, 2020, National Wetlands Inventory: U.S. Fish and Wildlife Service web page, accessed July 18, 2021, at https://www.fws.gov/wetlands/.
Appendix 4. Macroinvertebrate Sample Collection and Preservation Protocols
-
1. After loading the boat with necessary supplies (appendix 1), use the field Global Positioning System (GPS) device to navigate to a preprogrammed survey station.
-
2. Upon arriving at the survey station, prepare to take a benthic sweep. To collect the benthic sample, use the unmodified dip net. Plunge the net to the wetland bottom in a vertical position to avoid capturing invertebrates on the way down. Note the water depth by using depth increments marked on the handle and record it on the macroinvertebrate sampling data sheet (appendix 5). If water depth exceeds the length of the dip-net handle, either attach an extension to the handle or move along the transect toward shore until reaching an appropriate depth. If the GPS coordinates changed from the predetermined point because of depth or accessibility limitations, record the actual GPS coordinates for repeating samples or spatial analyses.
-
3. While keeping the net perpendicular to the wetland bottom, sweep horizontally for 0.5 meter (m), which can be referenced on your boat for improved accuracy. Skim the net along the top layer of sediment to the depth at which target organisms reside (generally a few centimeters deep) for 0.5 m. Take care to keep the handle vertical (that is, straight). Then, raise the net out of the water while keeping it in a vertical position to avoid capturing water column invertebrates.
-
4. Once the net emerges from the water, rotate it into a horizontal position. If vegetation and other organic debris are hanging over the net’s rim, place 50 percent of the vegetation/debris into the net and discard the rest. While keeping the dip-net rim out of the water, rinse sediment and fine organic matter from the sample by gently dipping the bell of the net in and out of the water, or by moving it from side to side slightly below the water’s surface. Splash the edges with water to flush all materials to the bottom, allow water to drain out, and place the contents into an appropriately labeled benthic sample bottle. Benthic samples can either be stored in separate bottles for each survey station or composited into the same bottle(s) for wetland-level estimates. Each bottle should contain an external and internal label. Internal labels must be written on an ethanol-resistant paper with an ethanol-resistant writing utensil (such as a graphite pencil), and external labels should be secured with clear packing tape. Immediately pour 95-percent ethanol into the bottle to an approximate volumetric ratio of 1 part sample to 2 parts ethanol.
-
5. An optional step for wetlands with sufficient water depth and depending on research objectives is to prepare to take a water column sweep. Shift the boat slightly (1–2 m) away from the disturbed area. To collect the water column sample, plunge the modified dip net to a depth equal to the total water depth minus the benthic net height. Shake out the net to ensure the mesh is free of the frame. Move the net sideways to undisturbed water and raise it vertically through the water column. Once the net emerges, while keeping the rim out of the water, splash the edges with water to flush all materials to the bottom and place the contents into an appropriately labeled water column sample bottle; like the benthic samples, water column samples can either be stored in separate bottles for each survey station or composited into the same bottle(s). Immediately pour 95-percent ethanol into the bottle to an approximate volumetric ratio of 1 part sample to 2 parts ethanol.
-
6. Repeat steps 2–5 for each survey station and ensure both field data sheets (appendix 5) have been completed before leaving the wetland.
Appendix 5. Field Data Sheets
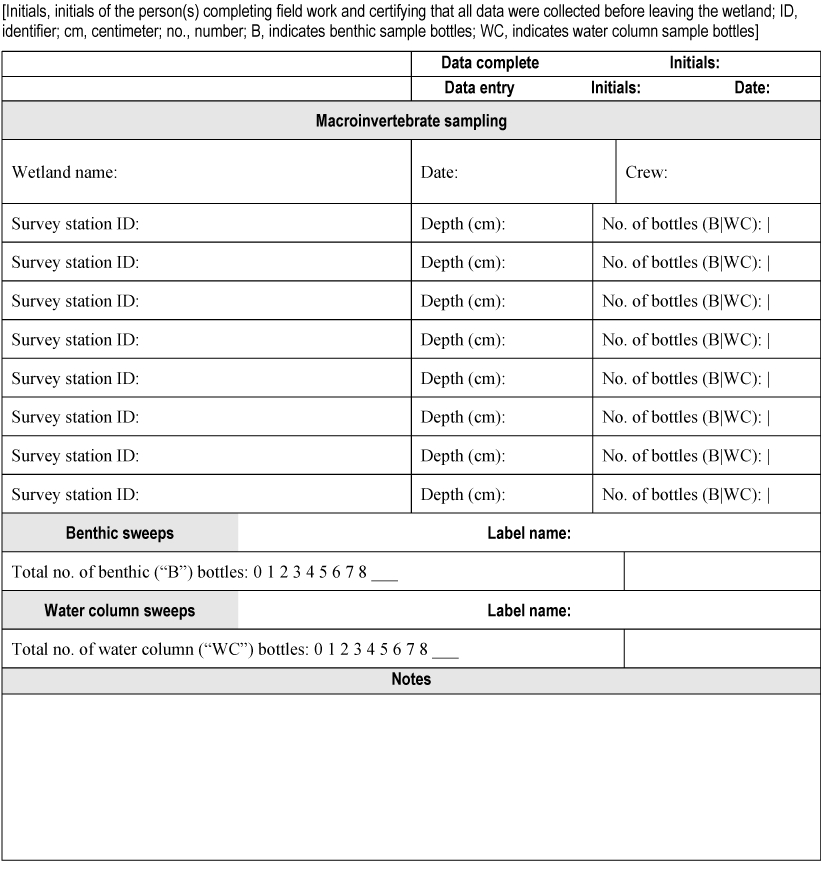
Data sheet for macroinvertebrate sampling.

Data sheet for site information.
Appendix 6. Laboratory Supply List
-
• Macroinvertebrate laboratory data sheets (appendix 7)
-
• Standardized subsampling apparatus with 500-micrometer sieve and gridded frame (appendix 8)
-
• White plastic or enamel pan for sorting
-
• Small 500-micrometer sieve for decanting ethanol
-
• 3× lighted magnifier
-
• Forceps
-
• Stainless steel spatula
-
• Ethanol waste container
-
• Funnel
-
• Gloves
-
• Specimen vials with screw caps (for example, 20-milliliter scintillation vials or 125-milliliter Nalgene bottles)
-
• Sample labels
-
• Ethanol-resistant pen
-
• 70-percent ethanol for long-term preservation of specimens (after picked from sediments and organic matter)
-
• Wash bottles with tap water
-
• Hand tally counter
-
• Scissors
-
• Spray bottle
Appendix 7. Laboratory Data Sheets
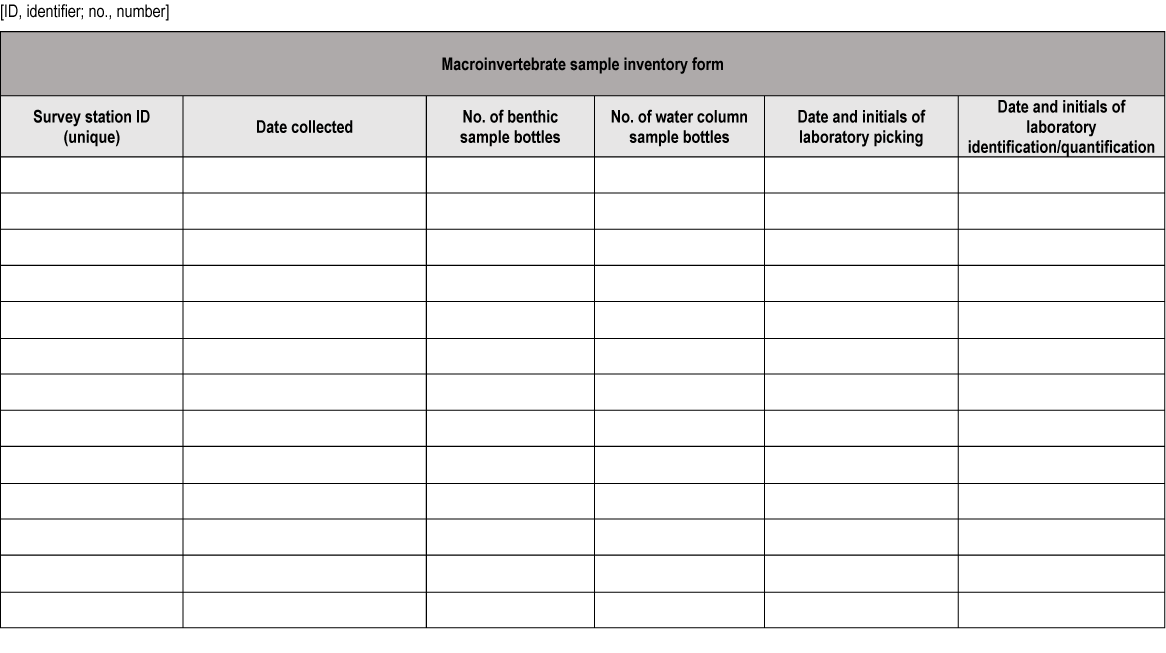
Data sheet for macroinvertebrate sample inventory.
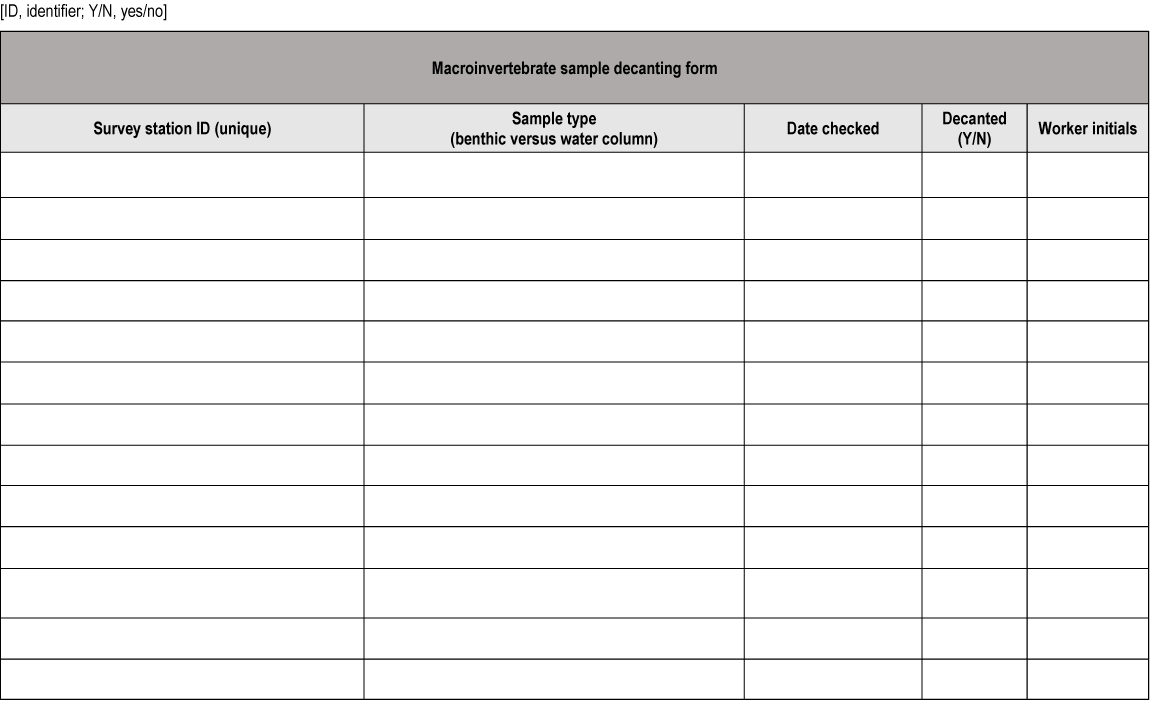
Data sheet for macroinvertebrate sample decanting.
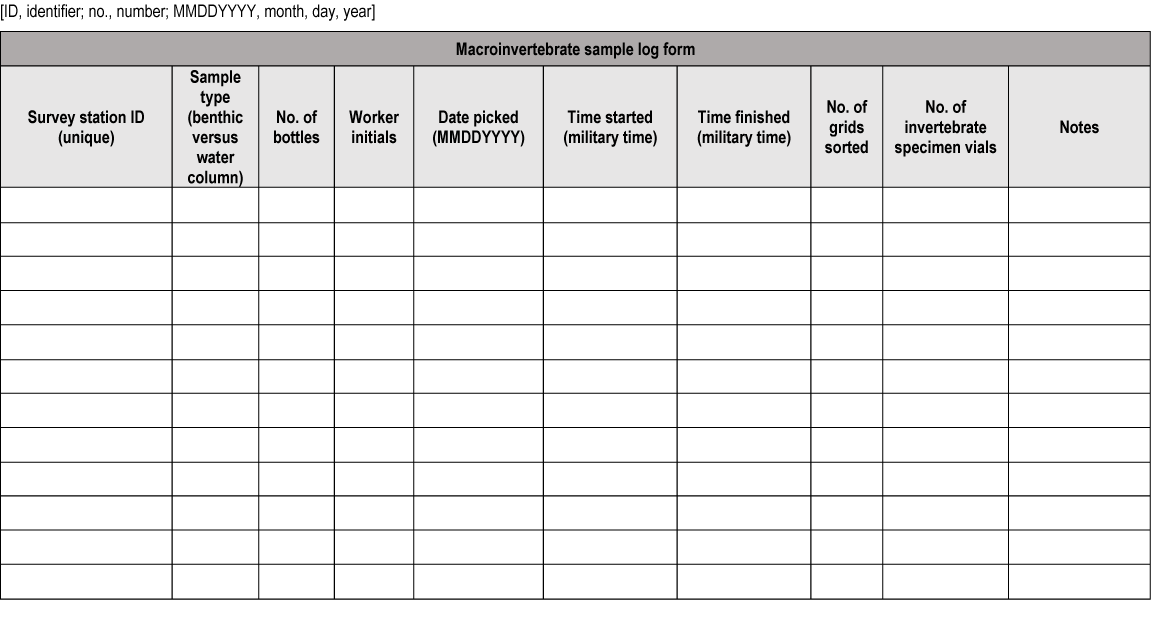
Data sheet for macroinvertebrate sample logging.
Appendix 8. Directions for Building a Subsampling Apparatus
-
1. Supplies
-
2. Construction of sieve
The sieve is composed of a stainless-steel mesh secured with staples between two wooden frames; this provides depth above and below the mesh to contain the sample and allow water to rinse through. Wood should be treated with a sealer to prevent water damage. The sieve shown in figure 8.1A–C measures about 29 cm long by 34 cm wide by 7.5 cm high.
-
3. Construction of grid
The overlying, removable grid is composed of a wooden frame containing a simple 2 x 2 wire grid to define the subsampling areas. The grid frame shown in figure 8.1D–F measures about 33 cm long by 38 cm wide.

The equipment and process for constructing the subsampling sieve (A–C) and overlying gridded frame (D–F). A, subsampling sieve; B, stainless steel mesh; C, wooden frames that secure the mesh; D, overlying removable grid; E, wire of removable grid; F, wire of removable grid secured to wooden frame. Descriptive laboratory processing protocols are provided in appendix 9.
Appendix 9. Detailed Instructions for Macroinvertebrate Picking
-
1. Put on gloves and safety goggles, open the sample bottle, and carefully decant ethanol from the sample by pouring the fluid through a small 500-micrometer sieve held inside a funnel over a labeled ethanol waste container. Repeat this step for the remaining bottles of the same sample. Thoroughly backwash/rinse the small sieve into the sample bottle to remove any organisms or debris that may have been collected on the sieve mesh while decanting.
-
2. If the contents of one sample are stored in more than one sample bottle, pour the contents of all bottles into a single appropriately sized tub. Rinse the inside of the sample bottles over the tub using water to collect any organisms or debris remaining inside. Pull the internal label out of the sample using forceps. Rinse the label over the tub using a water wash bottle, and set the label aside. Add enough water to the tub to completely cover all sample materials and thoroughly but gently mix them with a spoon or spatula (fig. 4A).
-
3. Once the sample has been mixed, pour its contents onto the subsampling sieve, which contains 500-micrometer stainless steel mesh. Next, gently rinse the sample with water to remove remaining ethanol and fine sediment. Rinse any large rocks or organic materials (whole leaves, twigs, macrophyte mats, and so on) over the sieve, visually inspect them under a lighted 3× microscope to remove remaining attached macroinvertebrates and discard them.
-
4. Place the subsampling sieve inside a tub filled with water to a level slightly above that of the sample materials; this allows the macroinvertebrates and debris to become slightly suspended in the water while remaining contained within the sieve frame. Use a spoon, spatula, or long forceps to gently spread the sample material over the sieve mesh as evenly as possible. Once the sample is evenly dispersed across the mesh, lift the sieve out of the water-filled container and place the gridded subsampling frame (four grids) over the sieve’s frame (fig. 4B).
-
5. Record the sample identifier information from the sample labels on a macroinvertebrate sample log form (appendix 7).
-
6. Use a random number generator to determine the order in which grids will be processed from the subsampling apparatus. Using a plastic spoon, spatula, paintbrush, and scissors (if needed to cut through vegetation), remove all materials (organisms and debris) from the randomly selected grid and place them into a separate container (fig. 4C). Any organism that is lying over a line separating two grids is considered to be on the grid containing its head. If it is not possible to determine the location of the head, the organism is in the grid containing most of its body. Cover the remaining sample in the apparatus with the tub lid or tin foil to prevent the desiccation of remaining macroinvertebrates, and occasionally mist the sample with water to keep it moist.
-
7. To maximize the visibility of macroinvertebrates mixed with sample debris, pick the subsampled contents gradually. Transfer 2–3 heaping tablespoons of subsampled materials into a shallow white tray partially filled with water (fig. 4D). Visually inspect the contents once with the naked eye and then again with the 3× lighted magnifier while using forceps to gently remove all macroinvertebrates. Place all macroinvertebrates or select taxonomic groups directly into prelabeled vials with 70-percent ethanol; a single vial may be filled as much as halfway with specimens. Keep track of total individuals collected by using a hand tally counter. Once the tray’s contents have been thoroughly examined and all macroinvertebrates have been removed, remaining materials in the tray may be discarded. Repeat these steps until the subsampled grid’s contents have been processed entirely.
-
8. Once processed, if the cumulative analysis of the first grid (25 percent of the sample) yields a representative quantity of macroinvertebrates (this may vary based on project objectives and needs), then subsampling is complete. In some cases, project investigators may wish to incorporate an additional subsampling target, such as a minimum number of macroinvertebrates (for example, 400 macroinvertebrates per composited, wetland-level sample) to ensure representativeness. If so, the grids must be processed, one at a time, until the target is reached. Once you begin processing a grid, that grid must be picked entirely to ensure macroinvertebrate counts are scaled up properly. Record the total number of grids processed and total number of specimen vials used in the macroinvertebrate sample log form (appendix 7) and label the vials with the following information: contents, sample identifier, and number of grids processed.
-
9. Once picking is completed and all data have been recorded in the macroinvertebrate sample log form (appendix 7), remaining sample debris may be discarded, and sample bottles can be cleaned. Pour excess water into the sink with a strainer to prevent debris from entering the drain. Dispose of ethanol according to local regulations.
Conversion Factors
International System of Units to U.S. customary units
For more information about this publication, contact:
Director, USGS Northern Prairie Wildlife Research Center
8711 37th Street Southeast
Jamestown, ND 58401
701–253–5500
For additional information, visit: https://www.usgs.gov/centers/npwrc
Publishing support provided by the
Rolla and Sacramento Publishing Service Centers
Disclaimers
Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Although this information product, for the most part, is in the public domain, it also may contain copyrighted materials as noted in the text. Permission to reproduce copyrighted items must be secured from the copyright owner.
Suggested Citation
Keith, B.R., Carleen, J.D., Larson, D.M., Anteau, M.J., and Fitzpatrick, M.J., 2022, Protocols for collecting and processing macroinvertebrates from the benthos and water column in depressional wetlands: U.S. Geological Survey Open-File Report 2022–1029, 22 p., https://doi.org/10.3133/ofr20221029.
ISSN: 2331-1258 (online)
| Publication type | Report |
|---|---|
| Publication Subtype | USGS Numbered Series |
| Title | Protocols for collecting and processing macroinvertebrates from the benthos and water column in depressional wetlands |
| Series title | Open-File Report |
| Series number | 2022-1029 |
| DOI | 10.3133/ofr20221029 |
| Publication Date | April 27, 2022 |
| Year Published | 2022 |
| Language | English |
| Publisher | U.S. Geological Survey |
| Publisher location | Reston, VA |
| Contributing office(s) | Northern Prairie Wildlife Research Center, Upper Midwest Environmental Sciences Center |
| Description | vi, 22 p. |
| Online Only (Y/N) | Y |


