Sampling and Analysis Plan for the Water-Quality Monitoring Program in Lake Koocanusa and Upper Kootenai River, Montana, Water Years 2022–23
Links
- Document: Report (3.2 MB pdf) , HTML , XML
- NGMDB Index Page: National Geologic Map Database Index Page (html)
- Download citation as: RIS | Dublin Core
Abstract
The U.S. Geological Survey, in cooperation with the U.S. Environmental Protection Agency, collected water-quality samples and environmental data in Lake Koocanusa (also known as “Koocanusa Reservoir”), the Kootenai River, and the Tobacco River during water years 2022–23. The transboundary Lake Koocanusa is in southeastern British Columbia, Canada, and northwestern Montana, United States. It was formed by constructing Libby Dam on the Kootenai River 26 kilometers upstream from Libby, Montana. One of the lake sites and the Kootenai River site, in the Libby Dam tailwater (the outflow of the lake flow into the Kootenai River), were equipped with automated, high-frequency ServoSipper water samplers. At the lake site, these samplers were mounted to pontoon platforms during the summer, and a submersible ServoSipper sipper was deployed with ice buoys during the winter. Samples were automatically collected from multiple depths. At the Kootenai River site, these samplers were housed in the gage house. In water year 2022, discrete water-quality samples were collected every 4–6 weeks, year round, at all four lake sites in the Kootenai River between April and November. In water year 2023, discrete water-quality samples were collected at three lake sites and the Kootenai and Tobacco River sites every 4–6 weeks. The goal of this project was to collect multidepth, high-frequency vertical and temporal water-quality samples and data to understand the limnological and biological processes that control variations and trends in selenium concentrations and loads throughout Lake Koocanusa and in the Libby Dam tailwater at the southern end of the lake. This sampling and analysis plan documents the organization, sampling and data-collection scheme and design, pre- and post-collection processes, and quality-assurance and quality-control procedures of the Koocanusa/Kootenai water-quality monitoring program during water years 2022–23.
Introduction
Selenium in the 145-kilometer (km) long transboundary Lake Koocanusa (also known as “Koocanusa Reservoir”) in southeastern British Columbia, Canada, and northwestern Montana, United States (fig. 1), has been detected at concentrations above State and Federal water-quality and aquatic-life standards (Montana Department of Environmental Quality, 2020a). The lake is within the international Kootenai (spelled “Kootenay” in Canada) River watershed, which contains critical habitat for native fish species and supports a fishery inclusive of westslope cutthroat trout (Oncorhynchus clarkii lewisi), burbot (Lota lota), and bull trout (Salvelinus confluentus). Site-specific selenium standards for surface water (the dissolved fraction) and fish tissue were established for the lake in 2020 (Montana Department of Environmental Quality, 2020b). The site-specific selenium standards were developed from modeling the bioaccumulation of selenium in fish tissue (Presser and Naftz, 2020) in accordance with the U.S. Environmental Protection Agency (EPA) guidance for criteria derivation (U.S. Geological Survey, variously dated). These standards were adopted into Montana State law in 2020 and approved by the EPA in 2021 (EPA, 2021). To inform the development of these site-specific selenium standards, the U.S. Geological Survey (USGS) began a water-quality monitoring program in calendar year 2019 to understand spatial and temporal limnological and biological processes that control variations in selenium concentrations and loads throughout Lake Koocanusa and the upper Kootenai River. The water-quality monitoring program continued through water years 2022 and 2023 (October 1, 2021, to September 30, 2023).
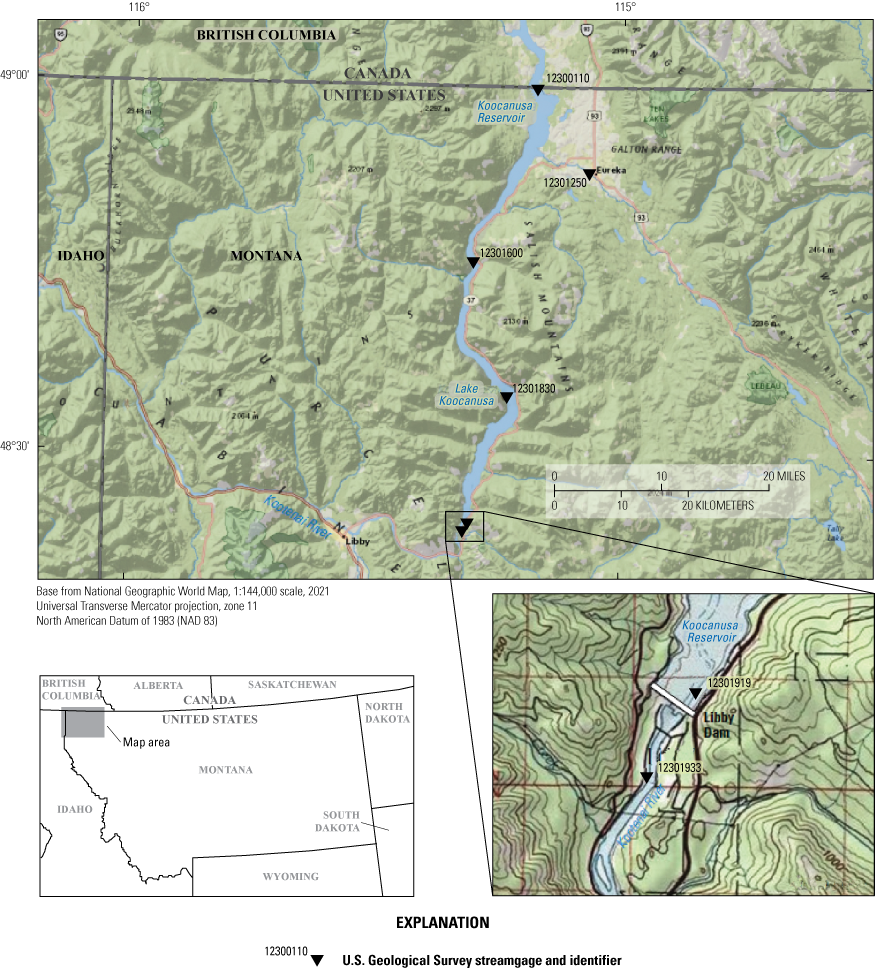
Kootenai River watershed, including Lake Koocanusa (also known as “Koocanusa Reservoir”) and major tributaries.
Purpose and Scope
The purpose of this report is to describe the sampling and analysis plan (SAP) of the USGS Koocanusa/Kootenai long-term water-quality monitoring program. This SAP follows the policies and procedures of the USGS; specifically, the USGS “National Field Manual for the Collection of Water-Quality Data” (NFM) (U.S. Geological Survey, variously dated) and “Guidelines and Standard Procedures for Continuous Water-Quality Monitors—Station Operation, Record Computation, and Data Reporting” (Wagner and others, 2006). Deviations from standard USGS procedures or procedures used where no standard procedures were available are detailed in this SAP. This report includes the logistics, sampling data-collection scheme and design, pre- and post-collection processes, and quality-assurance and quality-control procedures. The requirements for data management and review, as well as procedures to ensure health and safety, are also provided.
Study Area
Lake Koocanusa was created by the construction of Libby Dam in 1972, 26 km upstream from the town of Libby, Mont., and impounds water from about 23,271 square kilometers (km2), or 47 percent, of the Kootenai River drainage basin (Woods, 1982). Three Canadian rivers (Kootenay [Canadian spelling of Kootenai], Elk, and Bull Rivers (not shown)) supply 87 percent of the inflow to Lake Koocanusa (Bonde and Bush, 1982). The lake has a volume of 7.16 cubic kilometers (km3), and it reaches 77 km from Libby Dam in Montana to the Canada–U.S. border and 68 km farther into British Columbia (U.S. Army Corps of Engineers, 2020). Lake Koocanusa has a mean depth of 38 meters (m), a maximum depth of 106 m at the Libby Dam forebay, and a mean water residence time of about 9 months (Easthouse, 2013). Normal full pool and minimum regulated lake elevations are 750 and 697 m, respectively (Easthouse, 2013). Downstream from the lake, below Libby Dam, the flow of Kootenai River is reduced for most of the year, with peak outflows of as much as 750 cubic meters per second from late May through June and in December (Easthouse, 2013).
Background
Selenium in Lake Koocanusa has been detected at concentrations above State and Federal water-quality and aquatic life standards (Montana Department of Environmental Quality, 2020b). Selenium is a required micronutrient for most organisms, but elevated concentrations in water bioaccumulate in egg-laying fish and birds, causing various sublethal effects and death (Hamilton, 2004). Nitrate and sulfate loads to the reservoir also are of concern (Storb and others, 2023). Nitrate loading has potentially negative effects on phytoplankton and zooplankton community composition, and high nitrogen to phosphorus ratios can cause alteration of the lake’s trophic status (Elser and others, 2000). One source of selenium and nitrate in the Kootenai River Basin is the excavation of bedrock in the Elk Valley to access coal seams for metallurgical steelmaking and coal production (Mahmood and others, 2017; Wellen and others, 2018). There are currently (2026) five open-pit coal mines in this region of southeastern British Columbia, all operated by Elk Valley Resources (formerly Teck Coal, a subsidiary of Teck Resources Limited), with an annual production volume in 2020 of approximately 21 million tons of metallurgical coal (Teck Resources Limited, 2020).
Since 1984, total selenium concentrations in the Elk River have been measured at a British Columbia environmental monitoring station (site 0200016; not shown) (British Columbia Ministry of Forests, Lands, Natural Resource Operations, and Rural Development, 2022) 3.5 km above its discharge into Lake Koocanusa. Detected concentrations of total selenium ranged from 0.70 microgram per liter (µg/L; 1984) to 9.65 µg/L (2021). Since 1998, dissolved selenium concentrations have been reported in the Elk River at the same monitoring location and ranged from 1.10 µg/L (1999) to 9.46 µg/L (2021). In 2012, the Montana Department of Environmental Quality (MTDEQ) listed the lake as threatened by selenium (reporting as required by section 305(b) of the United States Federal Clean Water Act [33 U.S.C. 1251 et seq.]). In April 2013, the British Columbia Minister of Environment issued a Ministerial Order (No. M113) under the Environmental Management Act to reduce water-quality effects of mining activities to the Elk River and Koocanusa Reservoir (Province of British Columbia, 2013). Site-specific selenium standards of 0.8 µg/L for dissolved selenium in the water column and 15.1 mg/kg dry weight for fish egg (ovary) tissue (in addition to the muscle and whole-body standards) were established for the lake in 2020 following collaborative work by the MTDEQ, the British Columbia Ministry of Environment and Climate Change Strategy, the Lake Koocanusa Monitoring and Research Working Group, and a Selenium Technical Subcommittee (Administrative Rules of Montana 2020; Montana Department of Environmental Quality, 2020b). Simultaneously, this administrative rule maintained the existing selenium standard for the Kootenai River in Montana of 3.10 µg/L for dissolved selenium in the water column and 15.1 mg/kg dry weight for fish egg (ovary) tissue (in addition to the muscle and whole-body standards). The standards were adopted into Montana State law in 2020 and approved by the EPA in 2021 (EPA, 2021).
Monitoring water quality (and fish populations) in Lake Koocanusa and the Kootenai River has been ongoing since at least 1967, when the U.S. Army Corps of Engineers (USACE) and the USGS began collecting water samples (Easthouse, 2013) to better understand the relation between Libby Dam (completed in 1972) operations on the lake and downstream effects on the Kootenai River. Monitoring and research by these and other agencies have increased recently because of concerns that discharge from coal mining operations has led to increased selenium concentrations in the lake and the Kootenai River (Montana Department of Environmental Quality, 2020a).
In calendar year 2019, the USGS began a water-quality monitoring program in Lake Koocanusa and the Kootenai River just below Libby Dam (hereafter referred to as the “Koocanusa/Kootenai water-quality monitoring program”) to provide high-frequency water-quality data. In water years 2022–23 (October 1, 2021 to September 30, 2023), the specific objectives of this study were to (1) collect weekly composite water-quality samples for selenium from multiple depths in Lake Koocanusa at the International Boundary; (2) collect nitrate and sulfate samples from the lake and the river below the dam to understand their effect on hydrologic processes in the lake; (3) collect high-frequency continuous vertical profile data to understand limnological processes contributing to variations in selenium concentrations 4 to 12 profiles per day; and (4) establish a baseline water-quality dataset based on selenium concentrations measured in samples collected at lake and river sites.
Approach
Water-quality samples in the Koocanusa/Kootenai monitoring program were collected to measure concentrations of selenium, nutrients, anions, organic carbon, and other constituents. The overall approach for sample and data collection included the following components.
-
• A pontoon platform (fig. 2) on Lake Koocanusa at the international boundary (Lake Koocanusa at International Boundary; USGS site identifier 12300110; hereafter referred to as “site LIBB”; fig. 1; tables 1 and 2).
-
o The pontoon platform at site LIBB automatically collected vertical water-quality profile data, turbidity, dissolved oxygen concentration, pH, specific conductance, and water temperature using a vertical profile monitor 12 times per day (every other hour).
-
o The pontoon platform at site LIBB automatically collected composite water-quality samples during the summer (April–November) using a ServoSipper sampling system (refer to “High-Frequency Sample Collection from ServoSipper During Nonwinter—Site LIBB” section). Samples were collected automatically each day from multiple depths (table 2). Weekly samples were composited from the daily samples for each depth and submitted for laboratory analysis.
-
-
• A submersible ServoSipper sampler deployed using ice buoys to automatically collect composite water-quality samples from two depths at site LIBB during the winter of water year 2023 (November 2022 to April 2023; fig. 1; tables 1 and 2). Samples were collected automatically from those two depths. Weekly samples were composited from the daily samples and submitted for laboratory analysis.
-
• Discrete water-quality samples were collected manually every 4–6 weeks during summer (April–November) from varying depths during water years 2022–23 at site LIBB, on Lake Koocanusa at the Libby Dam forebay (Lake Koocanusa at Forebay, near Libby, Mont.; USGS site identifier 12301919; hereafter referred to as “site LIBF”), at Tenmile Creek (Lake Koocanusa at Tenmile Creek near Libby, Mont.; USGS site identifier 12301830; hereafter referred to as “site LIBT”; sampled 3 times in water year 2022 before discontinued in June 2022) (fig. 1; tables 1 and 2).
-
• Manual collection of discrete water-quality samples every 4–6 weeks year round from varying depths on Lake Koocanusa at Lake Koocanusa bridge (Lake Koocanusa below Pinkham Creek near Rexford, Mont.; USGS site identifier 12301600; hereafter referred to as “site LIBP”; fig. 1; tables 1 and 2) beginning August 2022.
-
• Manual collection of discrete water-quality samples every 4–6 weeks year round from the Tobacco River at Eureka, Mont. (USGS site identifier 12301250; hereafter referred to as “site TOBR”) beginning November 2022 (fig. 1; tables 1 and 2).
-
• Manual collection of depth-integrated water-quality samples every 4–6 weeks year round from the thalweg of the Kootenai River below Libby Dam near Libby, Mont. (USGS site identifier 12301933; hereafter referred to as “site LIBW”; fig. 1; tables 1 and 2).
-
• Technical review of the analytical results, quality-control data evaluation, and data management plan implementation (refer to “Data Management and Reporting” section).
-
• Distribution of approved data to the public using the USGS National Water Information System (NWIS) database (U.S. Geological Survey, 2023a).
Table 1.
Sample and data-collection locations and equipment, U.S. Geological Survey Koocanusa/Kootenai water-quality monitoring program, water years 2022–23.[USGS, U.S. Geological Survey; ADCP, acoustic Doppler current profiler]
| USGS site name | USGS site identifier | Abbreviated site identifier | Latitude (decimal degrees) | Longitude (decimal degrees) | Equipment and associated data-collection activity | Reference to data |
|---|---|---|---|---|---|---|
| Lake Koocanusa at International Boundary | 12300110 | LIBB | 48.99556 | −115.17861 | Pontoon platform with continuous vertical profile monitor and automated ServoSipper sampler; ADCP; real-time data transmission; discrete sampling; submersible ServoSipper sampler deployed with ice buoys tethered together; continuous water-quality monitor and ADCP (continuous water-quality monitor and ADCP discontinued in November 2022) | U.S. Geological Survey (2025a) |
| Tobacco River at Eureka, Montana | 12301250 | TOBR | 48.87795 | −115.05446 | No pontoon platform or unattended sensors; streamgage; discrete sampling beginning in November 2022 | U.S. Geological Survey (2025b) |
| Lake Koocanusa below Pinkham Creek near Rexford, Montana | 12301600 | LIBP | 48.82690 | −115.26712 | No pontoon platform or unattended sensors; discrete sampling began in August 2022 | U.S. Geological Survey (2025c) |
| Lake Koocanusa at Tenmile Creek near Libby, Montana | 12301830 | LIBT | 48.58500 | −115.23111 | No pontoon platform or unattended sensors; discrete sampling (sampling discontinued in June 2022) | U.S. Geological Survey (2025d) |
| Lake Koocanusa at Forebay near Libby, Montana | 12301919 | LIBF | 48.41194 | −115.30917 | No pontoon platform or unattended sensors; discrete sampling | U.S. Geological Survey (2025e) |
| Kootenai River below Libby Dam near Libby, Montana | 12301933 | LIBW | 48.40067 | −115.32028 | Streamgage with stationary (fixed point) multiparameter monitor and automated ServoSipper sampler; real-time data transmission; discrete sampling (stationary multiparameter water-quality monitor discontinued in February 2023) | U.S. Geological Survey (2025f) |
Table 2.
Types and frequencies of data collected in the U.S. Geological Survey Koocanusa/Kootenai water-quality monitoring program, water years 2022–23.[ADCP, acoustic Doppler current profiler; min, minute; m, meter]
| Abbreviated site identifier (table 1) |
Data-collection activity1 | Collection depth | Collection frequency | Reference to data |
|---|---|---|---|---|
| LIBB | Lake: continuous vertical water-quality profile and ADCP velocity data collection | 243-m maximum for profile; 45-m maximum for ADCP | 4 to 12 profiles per day (April–November; ADCP velocity every 15 min per 1-m depth; both data-collection activities discontinued November 2022 | U.S. Geological Survey (2025a) |
| LIBB | Lake: ServoSipper sampling during summer | 3Four depths: 3 m, 10 m, 25 m, and 35 m below the surface | Weekly composite (April–November) | U.S. Geological Survey (2025a) |
| LIBB | Lake: submersible ServoSipper sampling during winter | Two depths: 3 m and 10 m below the surface | Weekly composite (November–April) | U.S. Geological Survey (2025a) |
| LIBB | Lake: discrete sample collection | Water year 2022: 3 m below the surface and 3 m above bottom. Water year 2023: 3 m below surface, depth determined by change in specific conductance of the water column, and 3 m above bottom |
Every 4–6 weeks (April–November) | U.S. Geological Survey (2025a) |
| TOBR | River: discrete sample collection | Depth-integrated using the equal width increment sampling protocol | Every 4–6 weeks (year round) | U.S. Geological Survey (2025b) |
| LIBP | Lake: discrete sample collection | Water year 2022: 3 m below the surface and 3 m above bottom. Water year 2023: 3 m below surface, depth determined by change in specific conductance of the water column, and 3 m above bottom |
Every 4–6 weeks (year round) | U.S. Geological Survey (2025c) |
| LIBT | Lake: discrete sample collection | 3 m below surface and 3 m above bottom | Every 4–6 weeks (April–November); discontinued June 2022 | U.S. Geological Survey (2025d) |
| LIBF | Lake: continuous vertical water-quality profile | 492-m maximum for profile | 4 to 12 profiles per day; discontinued October 2022 | U.S. Geological Survey (2025e) |
| LIBF | Lake: continuous ServoSipper sampling | Two depths: 3 m and 20 m below surface | Weekly composite (November–April); discontinued October 2022 | U.S. Geological Survey (2025e) |
| LIBF | Lake: discrete sample collection | Water year 2022: 3 m below the surface and 3 m above bottom. Water year 2023: 3 m below surface, depth determined by change in specific conductance of the water column, and 3 m above bottom |
Every 4–6 weeks (April–November) | U.S. Geological Survey (2025e) |
| LIBW | River: continuous vertical profile monitor data collection | Fixed point, varies with river stage | Every 15 minutes (April–November); discontinued February 2023 | U.S. Geological Survey (2025f) |
| LIBW | River: transect and multi-vertical profile monitor data collection | 55.18 m | Vertical profiles at 10 points across river, twice per year (year round) | U.S. Geological Survey (2025f) |
| LIBW | River: continuous ServoSipper sampling | 61.82 m | Weekly composite (year round) | U.S. Geological Survey (2025f) |
| LIBW | River: discrete sample collection | Depth-integrated at thalweg | Every 4–6 weeks (year round) | U.S. Geological Survey (2025f) |
Changes from the Water Year 2021 Sampling and Analysis Plan
This SAP for the Koocanusa/Kootenai water-quality monitoring program differed from the water year 2021 SAP (Caldwell Eldridge and others, 2023) by the following modifications:
-
1. Discontinuation of high-frequency sample collection with the ServoSipper and water-quality vertical profile measurements at site LIBB in October 2021.
-
2. Discontinuation of sample collection for selenium speciation analysis at all sites in April 2022.
-
3. Discontinuation of sample collection at site LIBT in June 2022.
-
4. Addition of discrete sampling every 4–6 weeks year round at one lake site (site LIBP) in August 2022.
-
5. Addition of discrete sampling every 4–6 weeks year round at one river site (site TOBR) in November 2022.
-
6. Discontinuation of water-quality vertical profile data collection and acoustic Doppler current profile (ADCP) data collection at one lake site (site LIBB) in November 2022.
-
7. Discontinuation of the water-quality vertical profile data collection at one river site (site LIBW) in February 2023.
-
8. Addition of submersible ServoSipper at one lake site (site LIBB) during the winter and early spring of water year 2023 (November 2022–April 2023).
Organization and Responsibilities
The USGS personnel collected water-quality samples, data, and related information for the Koocanusa/Kootenai water-quality monitoring program during water years 2022 and 2023. Specific responsibilities of key personnel are listed in table 3. The project lead and field lead are responsible for ensuring that personnel who perform sample collection have appropriate training and are cognizant of onsite safety considerations. Water-quality samples were shipped to two laboratories for chemical analyses: the USGS National Water Quality Laboratory (NWQL; Denver, Colorado) and the Brooks Applied Laboratory (Bothell, Washington). The Brooks Applied Laboratory was used primarily to analyze samples for selenium concentrations (total and dissolved) and selenium speciation (selenium speciation was discontinued in April 2022). All other samples were analyzed at the NWQL (tables 4 and 5).
The addresses and contacts for the laboratories are:
U.S. Geological Survey, National Water Quality Laboratory
P.O. Box 25585
Denver, Colorado 80225-0585
(303) 236-3500
Brooks Applied Laboratory
18804 North Creek Parkway, Suite 100
Bothell, Washington 98011
(206) 632-6206
Amanda Royal, Laboratory Project Manager
Table 3.
Personnel in the U.S. Geological Survey Koocanusa/Kootenai water-quality monitoring program, water years 2022–23.Sampling and Analysis Plan
Sampling procedures in the Koocanusa/Kootenai water-quality monitoring programs were based on methods and requirements described in the NFM (U.S. Geological Survey, variously dated). Continuous environmental data will be recorded, processed, and reported as described in Wagner and others (2006). Deviations from standard USGS procedures or procedures used where no standard procedures are available are described in the following report sections. Water-quality sampling procedures were regularly overseen by the WY–MT WSC Water-Quality Specialist to ensure field protocols were followed. Field methods were reviewed annually within the WY–MT WSC in accordance with WY–MT WSC internal technical review procedures. The methods were independently audited once every 3 years during a review coordinated by the USGS Office of Quality Assurance.
Sampling Locations and Frequencies
Samples and data were collected at six sites during water years 2022–23 in the Koocanusa/Kootenai water-quality monitoring program. The sites, frequencies, data types, and equipment used for data and sample collection are described in tables 1 and 2. The abbreviated identifiers for USGS stations were based on but different from the abbreviated site identifiers used by the USACE (Easthouse, 2013).
Site LIBB had a pontoon platform with an automated ServoSipper sampler, a novel automated water sampler that uses a peristaltic pump and a series of valves to collect water samples, and a continuous vertical profile monitor (fig. 1; table 1; the vertical profile monitor was discontinued in November 2022). During the winter (November–April), a submersible ServoSipper sampler was deployed at this site with two ice buoys tethered together to collect samples under ice. Site LIBB was also equipped with an ADCP (until November 2022) to measure water current velocities using the Doppler effect of sound waves scattered back from particles within the water column (Côté and others, 2000; tables 1 and 2). Vertical profile and ADCP data were transmitted remotely in near real-time during operation. In addition to automated sampling, discrete samples were collected at site LIBB every 4–6 weeks between April and November.
Site TOBR in the Tobacco River included a streamgage and was sampled every 4–6 weeks, year round, beginning in November 2022 (fig. 1; tables 1 and 2). Site LIBP was sampled from a boat during the summer and from a bridge during winter, every 4–6 weeks year round beginning in August 2022. Site LIBT was sampled every 4–6 weeks beginning in April 2022 and ending in June 2022 (3 sampling events), when sampling was discontinued at this site. Site LIBF was sampled every 4–6 weeks between April and November (tables 1 and 2). Site LIBW on the Kootenai River included a streamgage, a stationary water-quality monitor, and a ServoSipper housed in the gage house. This site was sampled every 4–6 weeks, year round.
Sample Collection, Preservation, and Processing
This section describes the procedures for discrete and high-frequency water-quality sample collection, including sample trip and site visit preparation, documentation, sample tracking, and shipping to laboratories for analyses. Sampling equipment was cleaned at the WY–MT WSC water-quality laboratory in Helena, Mont., before taking the equipment into the field and after the first use. The Kemmerer sampler was also field-cleaned with deionized water and 5-percent hydrochloric acid (HCl) between sampling sites. All samples were collected following strict adherence to USGS guidelines for obtaining representative samples from the environment. This required field personnel to (1) understand the study objectives, including data-quality requirements, in the context of the water system being sampled; (2) minimize contamination and error during the sampling process; and (3) be aware of and document conditions that could compromise the quality of a sample (U.S. Geological Survey, variously dated). In addition, field personnel were expected to perform the following throughout the sampling process:
-
• Use prescribed “clean hands/dirty hands” techniques for parts-per-billion trace metal and other low-level sampling (U.S. Geological Survey, variously dated).
-
• Wear appropriate disposable, powderless gloves.
-
• Change gloves before each new step and as needed if gloves become contaminated during sample collection and processing, and avoid hand contact with contaminating surfaces.
-
• Never contact water samples using gloved or ungloved hands.
-
• Use equipment constructed of inert materials with respect to the analytes of interest.
-
• Use only equipment that has been cleaned according to prescribed procedures.
-
• Field rinse equipment as directed.
-
• Follow a prescribed order for collecting samples.
Sampling and Data Collection in Lake Koocanusa
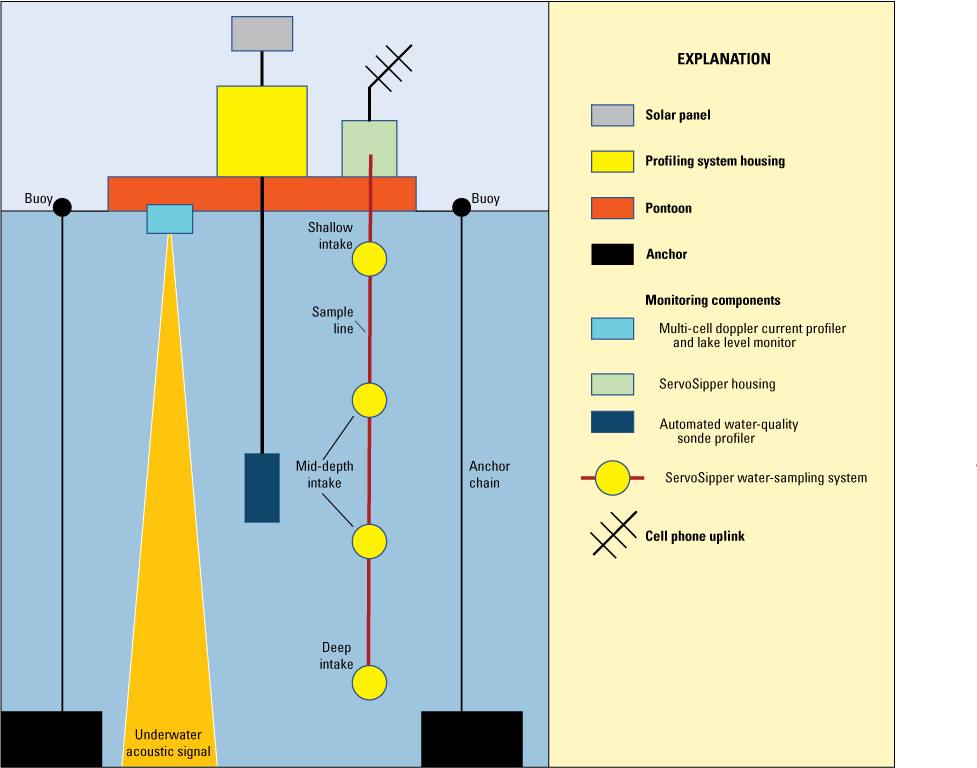
A pontoon platform (figs. 2 and 3) was deployed at site LIBB (fig. 1) to provide daily monitoring of total and dissolved selenium concentrations (table 4), vertical profile data, and ADCP velocity data. The vertical profile and ADCP velocity data collection were discontinued in November 2022 (tables 1 and 2). The platform at site LIBB was moored 1.21 km south of the Canadian border and accessed by boat. ServoSipper sampling systems developed by Thomas Chapin (USGS, Denver, Colo.) were mounted to the pontoon platform. These systems were set up differently for deployment during the winter to early spring so that samples could be stored longer; the field crew was unable to visit the site during icy conditions. Operation and use of the vertical profile and water-sampling equipment and discrete sample collection procedures at the lake sites are described in the following sections.

Pontoon platform used to collect water samples in Lake Koocanusa (also referred to as “Koocanusa Reservoir”). Photograph by Thomas Chapin, U.S. Geological Survey.
Submersible ServoSipper sampler deployed at Lake Koocanusa (also referred to as “Koocanusa Reservoir”) at the International Boundary, Montana (U.S. Geological Survey station identification number 12300110) during winter (November through April), water years 2022–23.
Table 4.
List of analytes, collection and preservation methods, and hold times for high-frequency ServoSipper samples collected in the U.S. Geological Survey Koocanusa/Kootenai water-quality monitoring program, water years 2022–23.[USGS, U.S. Geological Survey; NWQL, National Water Quality Laboratory; mL, milliliter; µm, micrometer; <, less than]
Continuous Vertical Water-Quality Profiles at the Pontoon Platform in Lake Koocanusa—Site LIBB
Continuous (subdaily) vertical profiles of turbidity, dissolved oxygen concentration and percent saturation, pH, specific conductance, and water temperature were recorded using a multiparameter sonde mounted to the pontoon platform at site LIBB. The sonde was connected to a data logger/control system connected to an electric winch that lowered the sonde to the appropriate depth (fig. 3; table 2). Four to 12 vertical profiles were completed per day, and data were recorded at every meter. The profiling equipment was serviced during discrete sampling visits every 4–6 weeks, and continuous records were processed according to established USGS protocols (Wagner and others, 2006). The sondes were deployed in April (water year 2022) and retrieved in November (water year 2023). Site LIBB was also equipped with an ADCP that collected velocity profiles every 15 minutes at 1-m depth intervals (table 2).
High-Frequency Sample Collection from ServoSipper During Nonwinter—Site LIBB
The nonwinter ServoSipper on the pontoon platform at site LIBB was deployed in April (water year 2022) and retrieved in November (water year 2023). ServoSipper samples were collected at the depths and frequencies in table 2 and analyzed for unfiltered and filtered (total and dissolved) selenium (table 4). Each ServoSipper sample was a weekly composite of 5- to 7-milliliter (mL) samples collected into one centrifuge tube daily. The ServoSipper pump and valve system automatically collected eight composite samples per depth—4 filtered and 4 unfiltered—in 50-mL plastic centrifuge tubes that were pre-loaded with 100 microliters (µL) of 7.5 Normal Ultrex nitric acid and 100 µL of deionized water to preserve the samples. The samples were checked for underfilling or overfilling during sample retrieval. If field personnel identified an error with sample collection, that information was documented, and the samples were discarded.
The ServoSipper system and power package were secured to the pontoon platform and placed inside an insulated cooler for protection against wave splash (fig. 2). A solar panel was mounted on the top of the cooler to recharge the battery power source. Semi-rigid silicone pump tubing was deployed through the bottom of the platform. The silicone tubing size for the pinch valves was 1/16-inch internal diameter and 1/8-inch outer diameter. Some of the transfer lines had 1/8-inch internal diameters and 1/4-inch outer diameters. The long intake line to the different sample depths consisted of Kynar tubing with a 1/8-inch internal diameter and a 3/16-inch outer diameter. The pump tubing was inside of a weighted, 2.5-centimeter diameter polyvinyl chloride (PVC) conduit attached to the side of the monitoring platform. Multiple sample access ports were pre-drilled into the conduit and screened with nylon mesh to allow sampling at different depths. The ServoSipper at site LIBB was visited for sample retrieval and equipment maintenance every 4–6 weeks, which was the same frequency as for collecting the discrete samples. Samples were retrieved, checked, bottled, and labeled by two people using the following process:
-
1. Disconnect the intake and waste line and unplug the battery lead before removing the tray.
-
2. Download the sample log using the PICAXE software (Revolution Education, Ltd., United Kingdom) and convert it to an electronic spreadsheet. The log contains the time stamp, fill volume, and condition comments for each sample and whether the sample was retained. Discard samples that overfill, partially fill, or are compromised.
-
3. Retrieve samples from one tray at a time.
-
4. Following the clean hands/dirty hands approach (U.S. Geological Survey, variously dated), disconnect the sample vials, one at a time, from the ServoSipper tray and record the fill volume.
-
5. Pour the sample from the 50-mL sample vial into a 125-mL, acid-rinsed bottle and label the bottle with the USGS site identifier, date range, sample time, depth, and if the sample was filtered or unfiltered.
-
6. Once labeled, place these bottles in a clean bag and label the bag with the USGS site identifier, sample depth, and sample collection date. Place all samples on ice.
-
7. Prepare a tray for the next sampling trip. Check the battery voltage and replace the battery if necessary. Inspect all lines for leaks or crimps and replace the old desiccant packets, if needed.
-
8. Remove used filters and flush the manifold tubing (called the “racetrack”) with deionized water for at least 30 seconds.
-
9. Add new filters, flush the line, and filter with about 8–10 mL of deionized water.
-
10. Load new, cleaned (within the past 1 week) 50-mL vials containing preservative into the cleaned tray. Vials are cleaned with phosphate-free detergent, a dilute acid (5-percent HCl) solution, and deionized water.
-
11. Create at least one blank sample for quality control using analyte-free (deionized) water for each tray. Use the tray to pump the blank water directly into the vial. Transfer the blank water into a bottle and label it with the USGS site identifier, collection date, sample time, sample depth, and whether the sample was filtered or unfiltered.
-
12. Start a new program on the tray and replace it in the cooler. Reconnect all lines.
-
13. Before leaving the site, check the ServoSipper to verify that the first-day samples have been filled and that everything is working properly.
High-Frequency Sample Collection from ServoSipper During Winter—Site LIBB
Two ServoSipper sampling systems were deployed at site LIBB from November to April (fig. 4; table 2). Each ServoSipper sampling system was deployed using two ice mooring buoys tethered together (two buoys for a single ServoSipper). ServoSipper samples were collected at the depths and frequencies in table 2 and analyzed for total and dissolved selenium (table 4). Each ServoSipper pump and valve system automatically collected 24 weekly composite samples (5- to 7-mL samples every day, 7 days per week), 12 filtered and 12 unfiltered, in 50-mL syringes pre-loaded with 7.5 Normal Ultrex nitric acid to preserve the samples until retrieval (fig. 4). The standard syringe plungers were replaced with O-rings to prevent sample contamination as the syringe filled. A syringe containing inorganic blank water was added to each ServoSipper sampler for quality control because the lake site could not be accessed during winter. Each ServoSipper was moored approximately 1.5 m below the water surface to prevent intake lines from freezing during deployment. Each ServoSipper sampling canister was deployed using counterweights to achieve the ideal depth of approximately 1.5 m. Intake lines extended below the sampling canisters along each mooring line to sample at 3 m and 10 m below the water surface. Each mooring line extended to the surface and was attached to a 60-inch ice buoy, which was designed to slip under moving ice and allow the ServoSipper samplers to remain stationary.
A single sampling canister is a self-contained unit that uses a 10-inch piece of plexiglass tube that is sealed at both ends with a double O-ring aluminum plug. Inlet and outlet access points on the ServoSipper are located on the top end (fig. 4). Communication access is available while the unit is sealed. Each ServoSipper sampler contained its own power supply, circuitry, pump, and manifold inside the sampling cannister (fig. 4).

Image of submersible ServoSipper samplers that were deployed at site LIBB in Lake Koocanusa (also known as “Koocanusa Reservoir”) during the winter to early spring (November through April; Lake Koocanusa at the International Boundary, Montana, U.S. Geological Survey site identifier 12300110), water years 2022–23. Photograph by Chad Reese, U.S. Geological Survey.
Discrete Sample Collection in Lake Koocanusa
Discrete water-quality samples were collected every 4–6 weeks at sites LIBB and LIBT from April through November (table 2); sampling at site LIBT was discontinued in June 2022. At site LIBP, discrete water-quality samples were collected every 4–6 weeks year round. Samples from LIBP were collected from a boat during the summer and from a bridge during the winter, but the sampling processes were the same. Samples were collected, filtered, and preserved according to procedures described by Ward and Harr (1990), Horowitz and others (1994), and the NFM (U.S. Geological Survey, variously dated) and analyzed for the constituents listed in table 5 and table 1.1. All constituents were measured in samples from all discrete sampling events at all sites. Water samples were collected with a Kemmerer sampler, which is a weighted, acrylic, transparent cylinder with end caps that close when triggered by a messenger (metal weight manually dropped down the line to the Kemmerer sampler) to entrap the water sample inside. The Kemmerer sampler has a lock to keep the stoppers open as the sampler is lowered through the water column. When the sampler is closed, the entire weight of the sampler and contents is carried upon the lower valve, which forces the sampler to sit securely on the lower valve and prevents water leakage. Samples were collected from two depths: 3 m below the water surface and 3 m above the lake bottom. Whole-water samples were collected directly from the Kemmerer sampler. The water samples were not mixed in a churn splitter because the lake had few suspended particles or sediment, and the water column was stationary. When severe weather occurred, samples were processed immediately upon returning to the shore (laboratory vehicle). Otherwise, samples were processed onsite. Filtered samples were collected by transferring water from the Kemmerer sampler into a 2-liter acid-washed bottle and filtering with a peristaltic pump that passed the water through silicone tubing and a capsule or disk filter.
Table 5.
List of analytes, collection and preservation methods, laboratory information, and hold times for discrete samples collected in the U.S. Geological Survey Koocanusa/Kootenai water-quality monitoring program, water years 2022–23.[CASRN, Chemical Abstracts Services Registry Numbers (a registered trademark of the American Chemical Society); USGS, U.S. Geological Survey; CaCO3, calcium carbonate; NWQL, National Water Quality Laboratory; mL, milliliter; µm, micrometer; <, less than; N, normal; mm, millimeter; GF/F, glass fiber filter; BAL, Brooks Applied Laboratory]
Preparing for Sampling
When sampling from a boat, sampling team navigated to within 10 m of sites without a pontoon platform and secured the boat to prevent drifting. This distance may have increased on windy days if the anchor dragged. Upon arrival at each site, the following steps were performed:
-
1. Conduct a safety inspection of the site, including pontoon platforms, and vicinity, as outlined in the Job Hazard Analysis (app. 2).
-
2. Before disturbing the site, describe and record the site and environmental conditions in field notes or field forms. This information must include the USGS site identifier, sampling date and time, personnel at the site, weather conditions, field observations, and an inventory of samples to be collected. Note any unusual conditions.
-
3. Measure and record the site depth using a depth sounder. Use this depth to determine where to place the Kemmerer sampler during sample collection.
-
4. Use a recently calibrated (within 1 week of data-collection activities) water-quality sonde to record a vertical profile of turbidity, dissolved oxygen concentration, pH, specific conductance, and water temperature throughout the entire water column, from the lake surface to the lake bottom.
-
5. Apply clean nitrile gloves. Replace gloves as needed during sample collection.
-
6. Create a clean environment for sample collection, identify the “clean hands” and “dirty hands” personnel (U.S. Geological Survey, variously dated), and unpack the sampling equipment while minimizing contamination. Record the time sampling begins.
-
7. Collect blank samples for quality assurance before collecting environmental samples. Do not field-rinse blank samples.
-
8. Add preservative (acid) as needed (table 5), and immediately place on ice. Record the lot numbers of used blank water and preservatives (acids) in the field notes.
Collecting Water-Quality Samples using a Kemmerer Sampler
Sampling equipment and bottles were field rinsed to condition (equilibrate) the equipment to the sample environment and to help ensure removal of all cleaning-solution residues or blank water. Discrete water-quality samples at the lake sites were collected 3 m below the water surface and 3 m above the lake bottom. Additional samples were collected to capture conditions at the observed range of specific conductance based on the vertical profile data (refer to “Preparing for Sampling” section). The maximum specific conductance is often at the thermocline when the lake is stratified. Samples were collected with a Kemmerer sampler using the following procedure:
-
1. Rinse the Kemmerer sampler with native site water three times.
-
2. Using the clean hands/dirty hands approach described in chapter A4 of the NFM (U.S. Geological Survey, variously dated), lower the Kemmerer sampler to the desired depth and send the messenger down the line to trigger the sampler to close at both ends. This collected water is the sample.
-
3. Collect whole-water (unfiltered) water samples directly from the Kemmerer sampler using the containers, volumes, and preservation methods listed in table 5. Collect chlorophyll-a samples from the lake as with other whole-water samples and filter these samples following the procedure outlined under “Filtration of Chlorophyll-a Samples” section. Rinse the collection containers twice with deionized water and once with unfiltered native water, fill the bottles to the shoulder, add preservatives (acids) if needed, and immediately place them on ice.
-
4. Record the lot numbers of preservatives used (acids) in the field notes.
-
5. Collect filtered (dissolved) subsamples after collecting the whole-water samples. To minimize contamination, filter the samples directly from the Kemmerer sampler through silicone tubing using a peristaltic pump that passes the water through a conditioned, disposable capsule or disk filter directly into the collection container.
-
6. Condition a new disposable 0.45-micrometer (µm) pore-size capsule or disk filter. Choose either a high-capacity filter (700 square centimeters [cm2]) for high turbidity conditions or a low-capacity filter (19.6 cm2) for low turbidity conditions. Attach a new disposable filter to the outflow end of the silicone tubing, add the intake end of the tubing to deionized blank water, and flush the filter with 2 liters of water for capsule filters or 50 mL for disk filters. Record the lot number of the disposable filter, remove the tubing from the deionized blank water, and purge the filter until it is clear of water.
-
7. Attach the intake end of the tubing to the Kemmerer sampler and remove the capsule or disk filter from the threaded connection at the outflow end. Turn on the pump and rinse the tubing with lake water. Turn off the pump, return the capsule or disk filter to the outflow end, and restart the pump. Rinse the polyethylene collection containers twice with deionized water and once with filtered lake water. Do not rinse the amber glass containers for organic carbon analysis.
-
8. Collect filtered water samples directly from the Kemmerer sampler using the containers, volumes, and preservation methods listed in table 5. After field rinsing the containers, fill them to the shoulder, add preservatives (acids) if needed, and immediately place them on ice. Record the lot numbers of the preservatives (acids) in the field notes.
-
9. For each primary sample, use a new, conditioned filter. After sample collection, purge the tubing of all water and discard the filter.
-
10. Clean the Kemmerer sampler between sites. Rinse it once with 5-percent HCl and three times with deionized water.
After returning to the office, ship all water-quality samples via overnight delivery to the analyzing laboratories and within the hold times shown in table 5.
Filtration of Chlorophyll-a Samples
Chlorophyll-a samples were filtered using a different procedure than for dissolved nutrients and trace elements. These samples were filtered the same day they were collected and typically processed together after all other samples were collected using the following procedure:
-
1. Avoid exposing the samples to direct light or sunlight.
-
2. Create a clean work surface and apply clean nitrile gloves. Clean plastic bags work well to cover surfaces.
-
3. Assemble the filtration equipment: a vacuum or hand pump with a pressure gage and tubing (the tubing will not be in contact with the sample and does not require cleaning), a side arm filter flask (catchment flask), a 47-millimeter fritted disk base (with a funnel or cup and a means for attaching it to the base, such as a magnet or clamp), a graduated cylinder, a rubber stopper, and clean, blunt-tip forceps. With the clean forceps, position a new glass fiber filter (GF/F, 0.3-µm pore size) grid-side down onto the center of the disk base.
-
4. Mix the sample by gently inverting the container several times. Use the graduated cylinder to measure a volume proportional to the relative biomass in the sample or water clarity to minimize filter clogging, typically 750–1,000 mL. Record the total volume filtered.
-
5. Apply vacuum to pass the sample through the filter. Do not exceed a vacuum pressure of 20 centimeters mercury.
-
6. When filtering is complete, rinse the filter, funnel or cup sides, and graduated cylinder with deionized water and pass this water through the filter. This will ensure no particles are lost to the sides of these containers.
-
7. Carefully release the vacuum and remove all equipment surrounding the filter.
-
8. Using clean forceps, carefully fold one side of the filter over the other with the sample contents to the inside. Do not touch the filtered material.
-
9. Place the filter in a small petri dish labeled with the USGS site identifier, sample collection date, sample collection time, and filtered volume. Wrap the petri dish in labeled foil and immediately freeze.
-
10. Clean the filtration apparatus and equipment with deionized water before filtering subsequent samples.
-
11. Store the filtered chlorophyll-a samples on dry ice or in a freezer until they are shipped to the laboratory (table 5).
-
12. Never allow the frozen filters to thaw before the laboratory receives them.
Sample and Data Collection at the Kootenai and Tobacco River Sites
This section describes the sampling and data-collection procedures at site LIBW in the Kootenai River and site TOBR in the Tobacco River. Site LIBW was located at a USGS streamgage and was equipped with a stationary water-quality monitor (until February 2023) to collect data continuously at a fixed point and an automated ServoSipper to collect high-frequency water-quality samples (tables 1 and 2). Discrete water-quality samples also were collected at the thalweg from a cableway at this site. Site TOBR did not have a ServoSipper or unattended sensors, and only discrete samples were collected here from a pedestrian bridge or by wading.
Continuous Water-Quality Data Collection in the Kootenai River—Site LIBW
A YSI model EXO2 water-quality sonde (YSI, Inc., Yellow Springs, Ohio) was located at the streamgage house on the riverbank of the Kootenai River at site LIBW to measure turbidity, dissolved oxygen concentration and percent saturation, pH, specific conductance, and water temperature. The sonde was housed in a 30-foot-long section of 4-inch diameter PVC pipe with a lockable cap. In the winter, heat tape was applied down the pipe to free the sonde from ice at the water’s surface. A cable housed in 3/4-inch diameter PVC conduit ran from the sonde to the data-collection platform. The sonde recorded data every 15 minutes and transmitted data through the data-collection platform hourly. The sonde was deployed all year and serviced every 4 to 6 weeks, depending on the weather, and the continuous records were processed according to established USGS protocols (Wagner and others, 2006). Multidepth profile data were collected twice per year from a cableway, according to Wagner and others (2006). High-frequency ServoSipper sampling at this site was as described for site LIBB (refer to “High-Frequency Sample Collection from ServoSipper During Nonwinter—Site LIBB” section). The ServoSipper was deployed all year, and this site was visited every 4 to 6 weeks for sample retrieval and equipment maintenance, depending on the weather.
Discrete Sample Collection from the Cableway—Site LIBW
Discrete water-quality samples were collected every 4–6 weeks year round, depending on the weather, from site LIBW (table 2). This site is near the ServoSipper intake, and the location was determined by reviewing ADCP measurements from the cableway at various flows to determine the thalweg of the cross section, which is where the highest velocities occur. The site is well-mixed during dam releases, because it is approximately 1 km downstream from the dam. There are no tributary inflows between Libby Dam and the sample site. Samples were collected from a cableway at a pre-determined, single horizontal point (the cable was marked with red paint at about 44.2 m from the right bank) using a US D–96 depth-integrating sampler (Edwards and Glysson, 1999; Federal Interagency Sedimentation Project, 2015). This device uses a plastic or Teflon nozzle and a plastic or Teflon bag as a sample container to collect flow-weighted samples in streams. Samples were collected, processed, filtered, and preserved according to procedures described by Ward and Harr (1990), Horowitz and others (1994), and the NFM (U.S. Geological Survey, variously dated) and analyzed for the constituents listed in table 5.
Sampling equipment and bottles were field rinsed to condition or equilibrate the equipment to the sample environment and to help ensure that all cleaning-solution residues or blank water were removed. Discrete water-quality samples were collected using the following procedure:
-
1. Rinse the sample collection nozzle, nozzle holder, bag, and churn splitter with native site water three times.
-
2. Using the clean hands/dirty hands approach, lower the US D–96 sampler from the cableway to collect a flow-weighted, depth-integrated sample.
-
3. Pour the sample from the US D–96 sampler into a churn splitter.
-
4. Repeat steps 2 and 3 until there is enough water to rinse and fill all the sample containers.
-
5. Collect whole-water (unfiltered) samples from the churn splitter during continuous mixing using the containers, volumes, and preservatives listed in table 5. Collect chlorophyll-a samples as with other whole-water samples and filter these samples the same day as collection following the procedure outlined in the “Filtration of Chlorophyll-a Samples” section. Rinse the collection containers twice with deionized water and once with unfiltered native water, fill the bottles to the shoulder, add preservatives (acids) if needed, and immediately place them on ice.
-
6. Record the lot numbers of preservatives used (acids) in the field notes.
-
7. Collect filtered (dissolved) subsamples after collecting the whole-water samples. To minimize contamination, filter the samples directly from the churn splitter through silicone tubing using a peristaltic pump that passes the water through a conditioned, disposable capsule or disk filter directly into the collection container.
-
8. Condition a new disposable 0.45-µm pore-size capsule or disk filter. Choose either a high-capacity filter (700 cm2) for high turbidity conditions or a low-capacity filter (19.6 cm2) for low turbidity conditions. Attach a new disposable filter to the outflow end of the silicone tubing, add the intake end of the tubing to deionized blank water, and flush the filter with 2 liters of water for capsule filters or 50 mL for disk filters. Record the lot number of the disposable filter, remove the tubing from the deionized blank water, and purge the filter until it is clear of water.
-
9. Attach the intake end of the tubing to the spigot of the churn splitter and remove the capsule or disk filter from the threaded connection at the outflow end. Turn on the pump and rinse the tubing with water. Turn off the pump, return the capsule or disk filter to the outflow end, and restart the pump. Rinse the polyethylene collection containers twice with deionized water and once with filtered site water. Do not rinse the amber glass containers for organic carbon analysis.
-
10. Collect filtered water directly from the churn splitter sampler using the containers, volumes, and preservation methods listed in table 5. After field rinsing the containers, fill them to the shoulder, add preservatives (acids) if needed, and immediately place them on ice. Record the lot numbers of the preservatives (acids) in the field notes.
-
11. For each primary sample, use a new, conditioned disposable filter. After sample collection, purge the tubing of all water and discard the filter.
Sample Documentation and Chain of Custody
Data collected in the Koocanusa/Kootenai water-quality monitoring program must be legally defensible, so the transfer of samples and sampling data was strictly controlled. Standard chain-of-custody (CoC) procedures, when required, were followed for field documentation, sample labeling, packaging, and shipping of all samples. This section summarizes the procedures used in this study, which closely followed those outlined in the CoC and documentation section in U.S. Geological Survey (variously dated).
Site Files and Field Notes
Site files containing descriptive information on location, conditions, purpose, and ancillary information were kept for each data-collection site (Schroder and Shampine, 1992). Much of this information was stored electronically. A site description was prepared for each water-quality site where continuous data was recorded. The mandatory site information included the following: agency code, project number, USGS site identifier, latitude, longitude, latitude-longitude accuracy, horizontal datum, Water Science Center code, State code, county code, land net location, location map, map scale, altitude, method of altitude measurement, accuracy of altitude, vertical datum, hydrologic unit code, topographic setting, agency use of site, date inventoried, site type, data type, data reliability code, and use of site.
Field notes must contain sample information, including the USGS site identifier, agency code, sample date and time, sample conditions, sample purpose, quality-assurance activities, weather conditions, field parameters, and lot numbers of chemicals and equipment. These notes were maintained using a USGS-developed Microsoft Excel workbook called Superfly.
Sample Labeling
All samples were collected and stored in the required container type (tables 4 and 5) and clearly labeled. Labels were preprinted or hand-written in the field with a permanent waterproof marking pen using a waterproof label (for example, Avery Waterproof Laser Address Labels, product 5520) securely attached to the sample container. When preprinted labels were used, field personnel ensured the label and the information on it remained intact and legible throughout the processing and shipping process.
Further guidance on sample processing and container types is available in chapter 5 of the NFM (U.S. Geological Survey, variously dated). Each sample label and the analytical service request (ASR) forms that accompany the samples showed the USGS site identifier, project identification number (on ASR only), sample type, sample depth, date of collection (month/day/year), time of collection (24-hour format) and bottle type.
Sample Tracking
A sample tracking spreadsheet was created and maintained to record all samples collected and shipped to analyzing laboratories to ensure the complete and timely receipt of analytical results and accurate transfer of data to NWIS (U.S. Geological Survey, 2025n). Technical personnel were responsible for recording the required information. The project lead or designated technical personnel reviewed the sample tracking spreadsheet to determine if analyses were missing or if corrective action(s) were taken.
Sample Custody
All samples collected in the Koocanusa/Kootenai water-quality monitoring program were documented according to the procedures outlined in book 9, chapter A4 of the NFM (U.S. Geological Survey, variously dated). To maintain a clear record of sample custody from collection through sample analysis and to inform the laboratory of the analysis being requested for each sample, samples shipped to Brooks Applied Laboratory included CoC and ASR forms. Samples shipped to NWQL included ASR forms, but they did not include CoC forms because they were not required. The NWQL is a research laboratory that does not provide methods for regulatory purposes and, therefore, does not require the CoC process. Samples received at the NWQL were promptly logged, checked for proper preservation (if required), and placed in storage areas for each sample type to await analysis. No further security procedures were involved. Samples were disposed of after normal holding times using routine disposal procedures. CoC (if required) and ASR forms were sealed in plastic bags inside the shipping containers.
Samples were packaged and sent for analysis as a batch shipment, when feasible. Three copies of the CoC form and two copies of the ASR form were completed for each sample. Two copies of the CoC and one copy of the ASR accompanied each shipment, and one copy of each was saved for project records. CoC forms, when required, were signed and dated by a member of the field team with the date and time of sample transfer. Upon receipt of the samples, the laboratory receiver completed the transfer by signing and dating the CoC form. One copy of the CoC was left at the laboratory, and another copy was returned to USGS project personnel.
Sample Packaging
All samples were collected in the containers specified in tables 4 and 5. Following collection, sample bottle caps were checked to ensure tight, secure seals. Glass containers were wrapped in foam sleeves or bubble packs designed for shipping glass containers to prevent breakage during shipping. Chilled samples were packed with sufficient ice to keep the temperature at or below 4 degrees Celsius. The volume of ice should be equal to or greater than the volume occupied by the samples. Twice the volume of ice was used during warm months. During winter months, samples were packed to prevent freezing during shipment. “Blue ice” or other types of commercially available re-freezable ices were not used because samples could become contaminated or may not maintain an adequate temperature. Chlorophyll-a and selenium speciation samples were shipped using dry ice to keep the contents of the shipping containers frozen. These samples were placed in separate shipping containers. Due to potential overpressure from carbon dioxide from the dry ice, these shipping containers were ventilated. When shipping dry ice, guidelines provided by the shipping service were strictly followed.
Sample containers were sealed in two plastic, sealable bags (double bagged). Prior to shipping, each sample was compared to entries on the CoC and ASR forms to ensure bottles and CoC/ASR entries matched. If ice was required for sample preservation, samples were packaged in thermally insulated, rigid coolers; samples shipped with dry ice were placed in small, rigid coolers that could be ventilated (for example, Styrofoam). Each package of samples was sealed with shipping tape and shipped by the most expedient means possible. The temperature of each cooler shipped to NWQL was measured during sample login and documented in the Laboratory Information Management System. Samples were also checked for proper preservation upon receipt. Samples or coolers that arrived at NWQL above the minimum temperature requirement were processed and flagged accordingly. The resulting laboratory analytical data were reviewed and either maintained (with the appropriate remark code in NWIS) or rejected and not reported.
Coolers were inspected before packing samples to ensure that they were in acceptable condition; coolers with missing handles, loose fitting lids, and cracks were not used. Shipping containers were lined with two (one bag inside of the other) heavyweight plastic bags. To isolate the samples from the ice melt, samples were placed in heavy, sealable bags. Bagged sample containers and ice were placed inside the plastic bags, and the bags were sealed with a knot, filament tape, or twist ties. Shipping containers were securely taped around the outside to minimize leaking and to maintain sample integrity. Coolers with spigots were sealed around the spigot opening with silicone or epoxy to prevent leakage. Excess packaging, such as bubble wrap, was not used.
Quality Assurance and Quality Control
The quality-assurance (QA) and quality-control (QC) procedures used in the USGS Koocanusa/Kootenai water-quality monitoring program ensure the usability and reliability of all data. Key components of the QA/QC plan for this work include, but are not limited to, following standard procedures for decontaminating field equipment; maintaining, calibrating, and operating field equipment and instrumentation in accordance with manufacturer’s instructions; following standard operating procedures outlined in the NFM (U.S. Geological Survey, variously dated); using standard field forms; using skilled personnel for sampling; and adhering to this SAP. The QC samples described in this section ensure that data collected in the field, analyzed by the laboratory, and entered into NWIS will be of known and appropriate quality to meet the study objectives.
U.S. Geological Survey Quality-Assurance Program
To address QC issues related to water-quality activities, the USGS Water Mission Area has implemented policies and procedures designed to ensure that all scientific work by or for the Water Mission Area is consistent and of documented quality. One tool used to ensure compliance is the technical review of water activities in USGS Water Science Centers. Each USGS Water Science Center has developed Internal Technical Review Procedures describing the Center’s plan to ensure quality assure their programs. These Internal Technical Review Procedures are submitted to the USGS Office of Quality Assurance and are conducted by multidiscipline teams that visit and review each Center regularly. These reviews include, but are not limited to, the following activities:
-
• Observing field methods, equipment and field installations, vehicles, laboratories, and warehouses for adequacy, safety, and compliance with policies and guidelines.
-
• Reviewing data records and analytes, technical files, and documentation such as flood plans and model archives.
-
• Reviewing QA plans and procedures that cover program planning, field measurements, sample collection, laboratory analyses, data interpretation, and report preparation and publication.
-
• Reporting the review findings to the Center at the conclusion of each review, including major comments and recommendations.
In addition, the USGS Quality Systems Branch (U.S. Geological Survey, 2025g) produces water and sediment performance-assessment samples and data to provide the USGS laboratory users, USGS laboratories, and field personnel with quantitative performance information to assess data quality. Specific QA programs within the Quality Systems Branch include the Inorganic Blind Sample Project (U.S. Geological Survey, 2025h), the Organic Blind Sample Project (U.S. Geological Survey, 2025i), the Laboratory Evaluation Project (U.S. Geological Survey, 2025j), the National Field Quality Assurance (NFQA) project (U.S. Geological Survey, 2025k), and the Standard Reference Sample Project (U.S. Geological Survey, 2025l), among others.
The USGS NWQL is accredited by the National Environmental Laboratory Accreditation Conference (NELAC) Institute (National Environmental Laboratory Accreditation Conference Institute, 2025), actively participates in the NELAC Institute National Environmental Laboratory Accreditation Program and participates in the proficiency testing program specified by the NELAC Institute. The NWQL also operates under the Quality Management System (QMS) required of all USGS laboratories. The QMS defines the policies, procedures, and documentation that ensure the laboratory continually meets a defined standard of quality that is designed to provide customers with legally defensible data of known and documented quality and, where applicable, demonstrates regulatory compliance. The QMS establishes the standard under which all laboratory operations are performed, including the laboratory’s organization, objectives, and operating philosophy. The QMS has been prepared to ensure that the laboratory operates a quality system in compliance with the 2003 NELAC standard.
Additionally, all field personnel in the USGS Koocanusa/Kootenai water-quality monitoring program participate in the USGS NFQA program. For QA and consistency among instruments and users, the NFQA program was created in 1979. The program monitors the proficiency of alkalinity, pH, and specific conductance measurements determined by the USGS water-quality field analysts. Field personnel and water-quality analysts are sent proficiency samples of pH, specific conductance, and alkalinity (added in 1984) annually. Participants who receive an unsatisfactory performance rating are given a second round of samples (Stanley and others, 1992). Results of the NFQA program are reviewed by a Water Science Center Water-Quality Specialist. If unsatisfactory performance persists, additional training and testing are provided to achieve satisfactory performance or staff are reassigned.
Field Quality Control
Quality-control samples were collected to determine the acceptability of performance in the data-collection process and to provide a basis for evaluating the adequacy of procedures that were used to obtain the data. The QA plan for this project followed the QA Plan for Water-Quality Activities for the WY–MT WSC (unpublished, internal document that follows the recommended USGS practices described at U.S. Geological Survey, 2025m). The QC samples were collected along with all primary environmental samples following USGS protocols described in chapter A4 of the NFM (U.S. Geological Survey, variously dated). Between 10 and 20 percent (or more) of all samples analyzed consisted of QC samples (table 3.1), and these QC sample types are described in table 6. Field blank samples consisted of American Chemical Society reagent-grade inorganic blank water or pesticide-grade organic blank water (for organic carbon analysis) processed through clean sampling equipment prior to collection of environmental samples.
Table 6.
Types of quality-control samples collected in the U.S. Geological Survey Koocanusa/Kootenai water-quality monitoring program, water years 2022–23.[IBW, inorganic-grade blank water]
Equipment Calibration, Maintenance, and Documentation
Equipment was calibrated and maintained as described in the USGS NFM (U.S. Geological Survey, variously dated), Wagner and others (2006), and in the manufacturer’s instructions (YSI, undated). Instruments that were not left in the field for unattended deployments were calibrated and checked the first day of the sampling trip. Instruments left in the field for unattended operation were calibrated just prior to deployment and checked against a recently calibrated field monitor following the criteria in table 7. Sensors on the field sonde, including pH, specific conductance, and dissolved oxygen were recalibrated in the field if they did not meet these criteria. Service visits (every 4–6 weeks) for unattended instruments were also routinely conducted. During these service visits, a calibrated “reference” sonde equipped with the same sensors as the site monitor was used to check the reasonableness of the deployed site sonde and to independently check for environmental changes that occurred during instrument servicing. Side-by-side comparison measurements were recorded, ensuring that the reference and site sonde readings were closely aligned in time. The site sonde was then removed, cleaned, and redeployed. After cleaning, readings from the site and reference sondes were recorded again. Environmental changes observed by the reference sonde during cleaning were considered when calculating fouling errors (Wagner and others, 2006).
Calibration checks were then performed on the site sonde, with careful documentation of calibrations and checks to ensure data accuracy. Calibration drift corrections were calculated following the procedures outlined in Wagner and others (2006). Sensors were cleaned every 4–6 weeks but were not recalibrated unless they were outside the criteria specified in table 7. In addition to calibration information, the date, personnel initials, and maintenance activities (for example, battery or sensor replacement, cleaning, or wiper repair/replacement) were documented. Maintenance records of field equipment, including the manufacturer, make, model, and serial number were maintained.
Table 7.
Water-quality property stabilization criteria and calibration guidelines used for sensor data collection in the U.S. Geological Survey Koocanusa/Kootenai water-quality monitoring program water years 2022–23.[Criteria are based on the recommendations provided in Wagner and others (2006). ±, plus or minus; µS/cm at 25 °C, microsiemen per centimeter at 25 degrees Celsius; mg/L, milligram per liter; FNU, formazin nephelometric unit]
Documentation of sonde calibrations and calibration checks are critical for ensuring accuracy of the data. All sensor values were recorded before and after each cleaning and calibration so that the data could be adjusted proportionately to the amount of drift (Wagner and others, 2006).
Decontamination
All water-sampling equipment used in the USGS Koocanusa/Kootenai water-quality monitoring program was cleaned (decontaminated) to the extent required for low-level chemical analyses before sample collection (U.S. Geological Survey, variously dated). Equipment was decontaminated to ensure the equipment was not a source of contamination that could affect the ambient concentrations of target analytes. The proper removal of contaminants from equipment minimizes the likelihood of sample cross-contamination and reduces or eliminates the transfer of contaminants to the sites that were sampled. A description of samples collected as part of the quality control of cleaning procedures for this study is included in the “Field Quality Control” section of this report.
Equipment cleaning procedures vary depending on the types of water samples collected and the sensitivities of the analyses. Collection equipment and re-used sample bottles were thoroughly decontaminated with phosphate-free detergent, a dilute acid (5-percent HCl) solution, and deionized water before taking them into the field. Between site visits, equipment was field cleaned by first rinsing with deionized water, then a 5-percent HCl solution, and then rinsed again with deionized water. Prior to collecting the sample, the sampling equipment was rinsed three times with native water. The sequence for equipment cleaning prior to sample collection for inorganic and (or) organic analytes was as follows:
-
1. Inspect equipment for stains, cuts, or abrasions. Replace parts as needed.
-
2. Wear appropriate disposable, powderless gloves.
-
3. Rinse equipment with deionized water directly after use while equipment is still wet, then use cleaning procedures. Do not allow collection and processing equipment to sit uncleaned in a field vehicle or elsewhere between field trips.
-
4. For equipment used to sample for inorganics and organics, soak in tap water containing 0.1-percent Liquinox detergent and scrub surfaces with a soft bristle brush.
-
5. For equipment used to sample for inorganics and organics, rinse thoroughly with tap water.
-
6. For equipment used to sample only for inorganics and organics, check equipment for metal parts. Remove and clean the metal parts separately, if possible.
-
7. For equipment used to sample only for organics: rinse with pesticide-grade blank water only.
-
8. Allow equipment to dry completely on a clean surface.
-
9. Place dried equipment in doubled storage bags. Do not bag wet equipment.
Laboratory Analysis
The following sections describe how samples collected in the Koocanusa/Kootenai water-quality monitoring program were analyzed in water years 2022–23. This description includes the laboratories used, types of analyses performed, and an overview of how data quality was determined.
Laboratories
This section describes the different laboratories, types of analyses performed, and an overview of how data quality was determined in the Koocanusa/Kootenai water-quality monitoring program in water years 2022–23.
U.S. Geological Survey National Water Quality Laboratory
The NWQL uses approved methods for determination of organic, inorganic, and radioactive substances in water, sediments, and biological tissues. The methods used by the laboratory include methods approved by the USGS, EPA, the American Public Health Association, the American Water Works Association, the Water Environmental Federation, and the American Society for Testing and Materials.
The NWQL has an extensive quality-assurance program that includes multiple levels of data review and quality-control sampling to support their analytical methods and method validation (Pritt and Raese, 1995). Quality control at NWQL is monitored by three programs: (1) the internal blind sample program, (2) the external blind sample program, and (3) bench-level QC samples. Information about the external blind sample program is available in Ludtke and Woodworth (1997). NWQL participates in performance evaluation studies and laboratory certification programs. External agencies and customer organizations audit NWQL to assess analytical methods and QA/QC programs.
Brooks Applied Laboratory
The Brooks Applied Laboratory data review process is comprehensive and includes, at minimum, a review by an analyst, QA specialist, and two project managers. More information is available in Brooks Applied Labs (2022). In a typical analytical batch, the laboratory performs QC analyses, including laboratory duplicate samples, standard reference samples, laboratory blank spike samples, and method blank samples. The laboratory also submits biannual proficiency testing results of water and soil samples to another outside laboratory for evaluation (an interlaboratory split analysis).
Sample Analyses
Laboratory analytical procedures followed approved and standard (when possible) methods, which provided reporting levels (table 1.1) that were below regulatory criteria or guidelines (where applicable) and enabled direct comparison of analytical results with these criteria. Sample and QC data for this study are reported in standard format, such as USGS open-file reports or scientific investigations reports, or as USGS data releases. The reports include a case narrative summarizing any problems encountered in the analyses.
Data-Quality Objectives
The data-quality objectives for this study followed the guidelines outlined in U.S. Department of the Interior (2008) to ensure that data of known and acceptable quality were obtained. All measurements were performed, and samples were collected, to yield consistent results that were representative of the media and conditions measured. In the USGS Koocanusa/Kootenai water-quality monitoring project, water-quality data were documented to ensure they were meaningful, representative, complete, precise, accurate, comparable, and admissible as legal evidence to meet the scientific, engineering, and regulatory needs of our cooperators. Documentation of basic information, such as compatible monitoring objectives and program design features; metadata (when, where, and how the data were collected and who collected and analyzed the data); ancillary information (explanatory variables and study-site characteristics); and legal requirements were used to evaluate data. Documentation of sufficient QA and QC information to establish the quality and uncertainty in the data and interpretations were also maintained to determine the comparability and utility of datasets for intended users.
Data-Quality Assessment
The QC samples were analyzed along with the corresponding constituents, and the results were compared with those of the co-collected primary samples. A thorough discussion of the data quality is included in all written products. These sample comparisons were based on the relative percent difference calculated for each type of analysis as the absolute value of the difference between the primary and QC sample results divided by the average of the primary and QC sample results (multiplied by 100 to convert to percentage). Other factors considered in each data review included compliance with sample hold times and receipt conditions (samples arrived at the laboratory at the appropriate temperatures), results of sample blank analyses, detection limits, and adherence to laboratory control standards. Estimated values were used for evaluation purposes, whereas rejected values were not used.
Data Management and Reporting
Water-quality data were recorded electronically and stored in electronic project files to maximize their preservation. Recorded data included chemical, physical, biological, and ancillary data measured in the field. Analytical results and continuous monitoring data transmitted over the computer network or stored by an electronic data logger were also stored and securely maintained. Data and metadata that were reviewed and approved were stored in NWIS (Dupré and others, 2013; U.S. Geological Survey, 2023a).
Data Processing
Sampling information, metadata, and ancillary information were recorded electronically as field notes that were considered original record. These data were combined with analytical data from the laboratory in computer data files.
Continuous Monitoring (Sensor) Data
Continuous monitoring (sensor) data are water-quality records collected onsite by electronic sensors (mounted to sondes) and data loggers. The standard USGS methods for electronically recording data are by (1) transmitting data from a remote location by radio telemetry to a central location, and (2) field personnel recording data at the remote location. A copy of the original data was retained and archived in the office. Data were not manipulated by the field instrument or a computer except to convert record signals into data in commonly used units or to display data in a convenient format. The transfer of data from the electronic storage medium to NWIS requires thorough checking to ensure that the data have transferred successfully or that as much data as possible are recovered and errors removed. Data were adjusted (corrected) to account for calibration drift, fouling of sensors, interfering factors (from debris or animals, for example), or other errors. These adjustments (corrections) were prorated over time during the record period (Wagner and others, 2006).
Laboratory Analytical Data
Analytical data are results of field and laboratory chemical, physical, or biological determinations. Each sampling location was assigned a USGS site identifier for entry into the NWIS database. Field measurements were entered into NWIS by technical personnel as soon as possible after returning from the sampling field trip. A record number was assigned by the system and recorded, and samples were logged before data were transferred to the database. All data available from NWQL were electronically transferred to the NWIS database by the database administrator at least once per week. Hard copies of the analytical reports (known as “WATLISTs”) were forwarded to the project lead or designated personnel for initial data review and storage in project files. The NWIS Quality of Water Database (QWDATA; USGS NWIS database for discrete water-quality data) receives daily incremental backup and weekly full back up by the site administrator.
Data obtained from Brooks Applied Laboratory analyses were entered into NWIS and identified using appropriate analyzing entity codes. Data from Brooks Applied Laboratory were entered and stored in NWIS in the same manner as data from NWQL.
Data were received electronically, reviewed by the project and data leads, and uploaded to NWIS (U.S. Geological Survey, 2025n). Quality-control data were reviewed as received. If needed, rerun or follow-up analyses were requested from the corresponding laboratory.
Data Validation
Data validation is the process whereby water-quality and associated data are checked for completeness and accuracy. After validation, data records are finalized in NWIS (U.S. Geological Survey, 2023a).
Continuous Monitoring (Sensor) Data
Following the entry of continuous monitoring data into NWIS, raw data, and (or) graphs of raw data were reviewed for anomalous values, dates and times, and preliminary updating were done. Once the data were edited, the records were submitted to a designated approver.
Laboratory Analytical Data
All field notes and field measurements were reviewed for completeness and accuracy as soon as possible after returning from the field trip by field personnel. Sample status was tracked using a sample tracking spreadsheet throughout collection through review, and samples were logged into this spreadsheet immediately after returning from the field. All chemical analyses were reviewed for completeness, and questionable values were noted by the project lead or designated personnel. Prompt review is necessary to allow analytical reanalysis to be performed before sample holding times have been exceeded. Data were entered in NWIS QWDATA results in an output (WATLIST) that includes a copy of the analysis and a report of general validation checks. If data from more than one sample was available for a site, the results were compared with previous results within a hydrologic context to identify obvious errors, such as decimal errors, and sample anomalies warranting analytical reanalysis. These reports and comparisons were reviewed and noted in the sample tracking spreadsheets. Constituents for reanalysis were indicated in QWDATA by the data quality indicator set to “I,” which stands for “in review.” Once data were reviewed, the data quality indicator was set to “R,” which stands for “reviewed and accepted” if there were no issues with the data. If there were anomalous values and a reanalysis was not possible, the data quality indicator was set to “Q,” which stands for “reviewed and rejected.”
Data Storage
All water-quality data were stored in NWIS (U.S. Geological Survey, 2023a). Supporting project information or metadata that could not be entered into NWIS were stored in electronic project files. The USGS WY–MT WSC site administrator maintains backups of all locally stored electronic information. Nonelectronic information, if any, is retained by the project lead or designated personnel. Water-quality data for this study were disseminated to the public through NWIS, USGS data releases, interpretive reports, or other published documents.
Health and Safety
The job hazard analysis prepared for this study was provided to field personnel and reviewed prior to each site visit. The job hazard analysis includes an assessment of job tasks; potential hazards, unsafe acts, or conditions; required personal protective equipment; and telephone contacts (app. 2). Additional health and safety considerations are addressed in the USGS publication, “Safety and Health for Field Operations” (U.S. Geological Survey, 2014).
Training and Certification
Training is required as described in book 9, chapter A6 of the NFM, “Field Measurements” (U.S. Geological Survey, variously dated). Additional training and refresher training (for example, safety, first aid, and motorboat operation) were scheduled regularly for field staff to meet USGS training and safety requirements. The project lead or designated personnel was responsible for ensuring that the training and certifications of the USGS field personnel were successfully completed before field work begins.
Acknowledgments
The authors sincerely thank all field personnel and science-support staff in the U.S. Geological Survey Wyoming–Montana Water Science Center who collected and quality-assured water-quality and streamflow data for this project, including Chad Reese, Jonathan O’Connell, and Seth Siefken.
References Cited
Bonde, T.J.H., and Bush, R.M., 1982, Limnological investigations—Lake Koocanusa, Montana, pt. 1 of Pre-impoundment study, 1967–1972: U.S. Army Corps of Engineers, Special Report 82–21, 203 p., accessed May 28, 2025, at https://apps.dtic.mil/sti/pdfs/ADA122882.pdf.
British Columbia Ministry of Forests, Lands, Natural Resource Operations, and Rural Development, 2022, BC Water Tool: Government of British Columbia database, accessed March 2022 at https://kwt.bcwatertool.ca/watershed.
Brooks Applied Labs, 2022, Quality assurance: Brooks Applied Labs web page, accessed December 19, 2023, at https://brooksapplied.com/about/quality-assurance/.
Caldwell Eldridge, S.L., Schaar, M.A., Reese, C.B., Bussell, A.M., and Chapin, T., 2023, Sampling and analysis plan for the Lake Koocanusa and upper Kootenai River, Montana, water-quality monitoring program, 2021: U.S. Geological Survey Open-File Report 2022–1113, 32 p., accessed December 19, 2023, at https://doi.org/10.3133/ofr20221113.
Côté, J.M., Hotchkiss, F.S., Martini, M., and Denham, C.R., 2000, Acoustic Doppler current profiler (ADCP) data processing system manual (ver. 4, September 2011): U.S. Geological Survey Open-File Report 00–458, 51 p., accessed December 19, 2023, at https://doi.org/10.3133/ofr00458.
Dupré, D.H., Scott, J.C., Clark, M.L., Canova, M.G., and Stoker, Y.E., 2013, User’s manual for the National Water Information System of the U.S. Geological Survey—Water-Quality System, version 5.0: U.S. Geological Survey Open-File Report 2013–1054, 730 p., accessed December 19, 2023, at https://doi.org/10.3133/ofr20131054.
Easthouse, K., 2013, Libby Dam—Kootenai River and Lake Koocanusa water quality sampling and analysis plan 2013: U.S. Army Corps of Engineers, Seattle District, Water Management Section, 24 p., accessed December 19, 2023, at https://usace.contentdm.oclc.org/digital/collection/p266001coll1/id/8870.
Edwards, T.K., and Glysson, G.D., 1999, Field methods for measurement of fluvial sediment: U.S. Geological Survey Techniques of Water-Resources Investigations, book 3, chap. C2, 89 p., accessed December 19, 2023, at https://pubs.usgs.gov/twri/twri3-c2/.
Elser, J.J., Fagan, W.F., Denno, R.F., Dobberfuhl, D.R., Folarin, A., Huberty, A., Interlandi, S., Kilham, S.S., McCauley, E., Schulz, K.L., Siemann, E.H., and Sterner, R.W., 2000, Nutritional constraints in terrestrial and freshwater food webs: Nature, v. 408, no. 6812, p. 578–580, accessed December 19, 2023, at https://doi.org/10.1038/35046058.
Federal Interagency Sedimentation Project, 2015, Operating instructions for the US D–96 depth-integrating collapsible bag suspended-sediment sampler: Vicksburg, Miss., Federal Interagency Sedimentation Project, 14 p., accessed December 19, 2023, at https://water.usgs.gov/fisp/docs/Instructions_US_D-96_Instructions_020709.pdf.
Hamilton, S.J., 2004, Review of selenium toxicity in the aquatic food chain: Science of the Total Environment, v. 326, no. 1–3, p. 1–31, accessed December 19, 2023, at https://doi.org/10.1016/j.scitotenv.2004.01.019.
Horowitz, A.J., Demas, C.R., Fitzgerald, K.K., Miller, T.L., and Rickert, D.A., 1994, U.S. Geological Survey protocol for the collection and processing of surface-water samples for the subsequent determination of inorganic constituents in filtered water: U.S. Geological Survey Open-File Report 94–539, 57 p., accessed December 19, 2023, at https://doi.org/10.3133/ofr94539.
Ludtke, A.S., and Woodworth, M.T., 1997, USGS Blind Sample Project—Monitoring and evaluating laboratory analytical quality: U.S. Geological Survey Fact Sheet 136–97, 2 p., accessed December 19, 2023, at https://doi.org/10.3133/fs13697.
Mahmood, F.N., Barbour, S.L., Kennedy, C., and Hendry, M.J., 2017, Nitrate release from waste rock dumps in the Elk Valley, British Columbia, Canada: Science of the Total Environment, v. 605–606, p. 915–928, accessed July 15, 2023, at https://doi.org/10.1016/j.scitotenv.2017.05.253.
Montana Department of Environmental Quality, 2020a, Derivation of a site-specific water column selenium standard for Lake Koocanusa: Helena, Mont., Montana Department of Environmental Quality, 221 p., accessed March 2022 at https://deq.mt.gov/files/Water/WQPB/Standards/Koocanusa/TSD_Lake%20Koocanusa_Sep2020_Final.pdf.
Montana Department of Environmental Quality, 2020b, Selenium standards for Lake Koocanusa and the Kootenai River, rule 17.30.632 of Surface water quality standards and procedures, subchap. 17.30.6 of Water quality, chap. 17.30 of Environmental quality: Administrative Rules of Montana, Title 17, 2 p., accessed December 19, 2023, at https://rules.mt.gov/browse/collections/aec52c46-128e-4279-9068-8af5d5432d74/policies/c305326d-3297-4e41-a472-c36b01354cb1.
National Environmental Laboratory Accreditation Conference Institute, 2025, The NELAC Institute: The NELAC Institute website, accessed August 12, 2025, at https://www.nelac-institute.org/.
Presser, T.S., and Naftz, D.L., 2020, Understanding and documenting the scientific basis of selenium ecological protection in support of site-specific guidelines development for Lake Koocanusa, Montana, U.S.A., and British Columbia, Canada: U.S. Geological Survey Open-File Report 2020–1098, 52 p., accessed March 2022 at https://doi.org/10.3133/ofr20201098.
Pritt, J.W., and Raese, J.W., 1995, Quality assurance/quality control manual of the National Water Quality Laboratory: U.S. Geological Survey Open-File Report 95–443, 35 p., accessed December 19, 2023, at https://doi.org/10.3133/ofr95443.
Province of British Columbia, 2013, Environmental management act: Province of British Columbia, Order of the Minister of Environment, section 89, Ministerial Order M113: 9 p., accessed December 19, 2023, at https://www.bclaws.gov.bc.ca/civix/document/id/mo/hmo/m0113_2013.
Schroder, L.J., and Shampine, W.J., 1992, Guidelines for preparing a quality assurance plan for district offices of the U.S. Geological Survey: U.S. Geological Survey Open-File Report 92–136, 14 p., accessed December 19, 2023, at https://doi.org/10.3133/ofr92136.
Stanley, D.L., Shampine, W.J., and Schroder, L.J., 1992, Summary of the U.S. Geological Survey National Field Quality Assurance Program from 1979 through 1989: U.S. Geological Survey Open-File Report 92–163, 18 p., accessed December 19, 2023, at https://doi.org/10.3133/ofr92163.
Storb, M.B., Bussell, A.M., Caldwell Eldridge, S.L., Hirsch, R.M., and Schmidt, T.S., 2023, Growth of coal mining operations in the Elk River Valley (Canada) linked to increasing solute transport of Se, NO3−, and SO42− into the transboundary Koocanusa Reservoir (USA–Canada): Environmental Science & Technology, v. 57, no. 45, p. 17465–17480, accessed August 15, 2024, at https://doi.org/10.1021/acs.est.3c05090.
Teck Resources Limited, 2020, 2020 annual report: British Columbia, Canada, Teck Resources Limited, 142 p., accessed December 19, 2023, at https://www.teck.com/media/2020-Annual-Report.pdf.
U.S. Army Corps of Engineers, 2020, Libby Reservoir, Lake Koocanusa, water quality report, May 2020: U.S. Army Corps of Engineers, Portland District, 104 p., accessed December 2025 at https://usace.contentdm.oclc.org/utils/getfile/collection/p16021coll7/id/15443.
U.S. Department of the Interior, 2008, Data quality management guide: U.S. Department of the Interior, Office of the Chief Information Officer, [variously paged; 70 p.], accessed December 19, 2023, at https://www.nps.gov/gis/egim/library/DataQuality_2008_0824_DOI%20Data%20Quality%20Management%20Guide.pdf.
U.S. Environmental Protection Agency [EPA], 2021, 2021 Revision to: aquatic life ambient water quality criterion for selenium—Freshwater 2016: Washington, D.C., U.S. Environmental Protection Agency, EPA 822-R21-006, accessed December 19, 2023, at https://www.epa.gov/system/files/documents/2021-08/selenium-freshwater2016-2021-revision.pdf.
U.S. Geological Survey, 2014, Safety and health for field operations: U.S. Geological Survey Handbook 45–3–H, 336 p., accessed May 28, 2025, at https://www.usgs.gov/media/files/445-3-h-safety-and-health-field-operations.
U.S. Geological Survey, 2023a, USGS water data for the Nation: U.S. Geological Survey National Water Information System database, accessed December 19, 2023, at https://doi.org/10.5066/F7P55KJN.
U.S. Geological Survey, 2025a, Monitoring location Lake Koocanusa at international boundary—USGS-12300110, in USGS water data for the Nation: U.S. Geological Survey National Water Information System database, accessed May 23, 2025, at https://doi.org/10.5066/F7P55KJN. [Site information directly accessible at https://waterdata.usgs.gov/monitoring-location/12300110/.]
U.S. Geological Survey, 2025b, Monitoring location Tobacco River at Eureka, MT—USGS-12301250, in USGS water data for the Nation: U.S. Geological Survey National Water Information System database, accessed May 23, 2025, at https://doi.org/10.5066/F7P55KJN. [Site information directly accessible at https://waterdata.usgs.gov/monitoring-location/12301250/.]
U.S. Geological Survey, 2025c, Monitoring location Lake Koocanusa bl Pinkham Cr nr Rexford MT—USGS-12301600, in USGS water data for the Nation: U.S. Geological Survey National Water Information System database, accessed May 23, 2025, at https://doi.org/10.5066/F7P55KJN. [Site information directly accessible at https://waterdata.usgs.gov/monitoring-location/12301600/.]
U.S. Geological Survey, 2025d, Monitoring location Lake Koocanusa at Tenmile Cr nr Libby MT—USGS-12301830, in USGS water data for the Nation: U.S. Geological Survey National Water Information System database, accessed May 23, 2025, at https://doi.org/10.5066/F7P55KJN. [Site information directly accessible at https://waterdata.usgs.gov/monitoring-location/12301830/.]
U.S. Geological Survey, 2025e, Monitoring location Lake Koocanusa at forebay, nr Libby, MT—USGS-12301919, in USGS water data for the Nation: U.S. Geological Survey National Water Information System database, accessed May 23, 2025, at https://doi.org/10.5066/F7P55KJN. [Site information directly accessible at https://waterdata.usgs.gov/monitoring-location/12301919/.]
U.S. Geological Survey, 2025f, Monitoring location Kootenai River bl Libby Dam nr Libby MT—USGS-12301933, in USGS water data for the Nation: U.S. Geological Survey National Water Information System database, accessed May 23, 2025, at https://doi.org/10.5066/F7P55KJN. [Site information directly accessible at https://waterdata.usgs.gov/monitoring-location/12301933/.]
U.S. Geological Survey, 2025g, Quality Systems Branch: U.S. Geological Survey, Quality Systems Branch website, accessed August 12, 2025, at https://qsb.usgs.gov/.
U.S. Geological Survey, 2025h, Inorganic Blind Sample Project (IBSP): U.S. Geological Survey, Quality Systems Branch web page, accessed August 12, 2025, at https://qsb.usgs.gov/ibsp/.
U.S. Geological Survey, 2025i, Organic Blind Sample Project (OBSP): U.S. Geological Survey, Quality Systems Branch web page, accessed August 12, 2025, at https://qsb.usgs.gov/obsp/.
U.S. Geological Survey, 2025j, Laboratory Evaluation Project: U.S. Geological Survey, Quality Systems Branch web page, accessed August 12, 2025, at https://qsb.usgs.gov/lep/.
U.S. Geological Survey, 2025k, National Field Quality Assurance Project: U.S. Geological Survey, Quality Systems Branch web page, accessed August 12, 2025, at https://qsb.usgs.gov/nfqa/.
U.S. Geological Survey, 2025l, The Standard Reference Sample Project: U.S. Geological Survey, Quality Systems Branch web page, accessed August 12, 2025, at https://qsb.usgs.gov/srs/srs.
U.S. Geological Survey, 2025m, Quality Assurance Plans—Recommended practices and examples: U.S. Geological Survey web page, accessed August 12, 2025, at https://www.usgs.gov/data-management/quality-assurance-plans-recommended-practices-and-examples.
U.S. Geological Survey, 2025n, USGS water data for the Nation: U.S. Geological Survey National Water Information System database, accessed August 12, 2025, at https://doi.org/10.5066/F7P55KJN.
U.S. Geological Survey, [variously dated], National field manual for the collection of water-quality data: U.S. Geological Survey Techniques of Water-Resources Investigations, book 9, chaps. A1–A10, accessed December 19, 2023, at https://www.usgs.gov/mission-areas/water-resources/science/national-field-manual-collection-water-quality-data-nfm.
Wagner, R.J., Boulger, R.W., Jr., Oblinger, C.J., and Smith, B.A., 2006, Guidelines and standard procedures for continuous water-quality monitors—Station operation, record computation, and data reporting: U.S. Geological Survey Techniques and Methods, book 1, chap. D3, 51 p., accessed December 19, 2023, at https://doi.org/10.3133/tm1D3. [Supersedes U.S. Geological Survey Water-Resources Investigations Report 2000–4252.]
Ward, J.R., and Harr, C.A., 1990, Methods for collection and processing of surface-water and bed-material samples for physical and chemical analyses: U.S. Geological Survey Open-File Report 90–140, 71 p., accessed December 19, 2023, at https://doi.org/10.3133/ofr90140.
Wellen, C., Shatilla, N.J., and Carey, S.K., 2018, The influence of mining on hydrology and solute transport in the Elk Valley, British Columbia, Canada: Environmental Research Letters, v. 13, no. 7, p. 074012, at https://doi.org/10.1088/1748-9326/aaca9d. [Supersedes preprint https://doi.org/10.32920/23915436.]
Woods, P.F., 1982, Annual nutrient loadings, primary productivity, and trophic state of Lake Koocanusa, Montana and British Columbia, 1972–80: U.S. Geological Survey Professional Paper 1283, 24 p., accessed December 19, 2023, at https://doi.org/10.3133/pp1283.
YSI, [undated], EXO user manual—Advanced water quality monitoring platform: Yellow Springs Instruments, Inc., revision K, 244 p., accessed January 19, 2023, at https://www.ysi.com/File%20Library/Documents/Manuals/EXO-User-Manual-Web.pdf.
Appendix 1. Analytes and Methods
Table 1.1.
Analytes, methods, and method descriptions used in the U.S. Geological Survey Koocanusa/Kootenai water-quality monitoring program, water years 2022–23.[CaCO3, calcium carbonate; NWQL, National Water Quality Laboratory; --, not available; clCP–MS, cell inductively coupled plasma-mass spectrometry; µg/L, microgram per liter; mg/L, milligram per liter; ICP–MS, inductively coupled plasma-mass spectrometry; ICP–AES, inductively coupled plasma-atomic emission spectrometry; IC, ion chromatography; ICP, inductively coupled plasma; BAL, Brooks Analytical Laboratory; IC–ICP–MS, ion chromatography-inductively coupled plasma-mass spectrometry; ICP–OES, inductively coupled plasma-optical emission spectrometry; µS/cm at 25 °C, microsiemen per centimeter at 25 degrees Celsius]
| Analyte | Analyzing laboratory | Analytical method1 | Method description | Method reporting level | Reporting level unit | Reference |
|---|---|---|---|---|---|---|
| Alkalinity as CaCO3 | NWQL | TT040 | Inflection-point titration | -- | -- | Fishman and Friedman (1989) |
| Aluminum | NWQL | PLM10 | clCP–MS | 3 | µg/L | Garbarino and others (2006) |
| Ammonia, as nitrogen | NWQL | 48 | Salicylate-hypochlorite, colorimetry, discrete analyzer | 0.026 | mg/L | Patton and Gilroy (1999) |
| Cadmium | NWQL | PLM43 | ICP–MS | 0.03 | µg/L | Garbarino and others (2006) |
| Calcium | NWQL | PLA11 | ICP–AES | 0.022 | mg/L | Fishman (1993) |
| Chloride | NWQL | IC022 | IC | 0.02 | mg/L | Fishman and Friedman (1989) |
| Chlorophyll-a | NWQL | EPA 445.0 | Chromatography-fluorometry | 0.1 | µg/L | Arar and Collins (1997) |
| Cobalt | NWQL | PLM10 | clCP–MS | 0.03 | µg/L | Garbarino and others (2006) |
| Copper | NWQL | PLM10 | clCP–MS | 0.4 | µg/L | Garbarino and others (2006) |
| Dissolved organic carbon | NWQL | CMB15 | High-temperature catalytic oxidation | 0.46 | mg/L | Clesceri and others (1989) |
| Iron | NWQL | PLA11 | ICP | 5 | µg/L | Fishman (1993) |
| Magnesium | NWQL | PLA11 | ICP–AES | 0.011 | mg/L | Fishman (1993) |
| Manganese | NWQL | PLM43 | ICP–MS | 0.4 | µg/L | Garbarino and others (2006) |
| Methylseleninic acid | BAL | BAL-4201 | IC–ICP–MS | 0.01 | µg/L | Wallschlager and Bloom (2001)2 |
| Nickel | NWQL | PLM10 | clCP–MS | 0.2 | µg/L | Garbarino and others (2006) |
| Nitrite, as nitrogen | NWQL | 49 | Diazotization, colorimetry, discrete analyzer | 0.001 | mg/L | Fishman and Friedman (1989) |
| Nitrite-plus-nitrate, as nitrogen | NWQL | RED02 | Enzyme reduction-diazotization, colorimetry | 0.02 | mg/L | Patton and Kryskalla (2011) |
| Nitrogen (alkaline persulfate) | NWQL | AKP01 | Acidified alkaline persulfate digestion, colorimetry | 0.015 | mg/L | Patton and Kryskalla (2011) |
| Orthophosphate, as phosphorus | NWQL | 48 | Phosphomolybdate, colorimetry, discrete analyzer | 0.004 | mg/L | Fishman (1993) |
| Pheophytin a | NWQL | EPA 445.0 | Chromatography-fluorometry | 0.1 | µg/L | Arar and Collins (1997) |
| Phosphorus | NWQL | CL020 | Acid persulfate digestion; colorimetry | 0.007 | mg/L | Patton and Kryskalla (2011) |
| Potassium | NWQL | PLOO3 | ICP–OES | 0.03 | mg/L | Fishman and Friedman (1989) |
| Selenate | BAL | BAL-4201 | IC–ICP–MS | 0.02 | µg/L | Wallschlager and Bloom (2001)2 |
| Selenite | BAL | BAL-4201 | IC–ICP–MS | 0.03 | µg/L | Wallschlager and Bloom (2001)2 |
| Selenium | BAL | EPA 1638 MOD | ICP–MS | 0.051 | µg/L | EPA (1996) |
| Selenium | NWQL | PLM10 | clCP–MS | 0.05 | µg/L | Garbarino and others (2006) |
| Selenocyanate | BAL | BAL-4201 | IC–ICP–MS | 0.02 | µg/L | Wallschlager and Bloom (2001)2 |
| Selenomethionine | BAL | BAL-4201 | IC–ICP–MS | 0.02 | µg/L | Wallschlager and Bloom (2001)2 |
| Sodium | NWQL | PLA11 | ICP–AES | 0.06 | mg/L | Fishman (1993) |
| Specific conductance, laboratory | NWQL | WHT03 | Wheatstone bridge, laboratory | 5 | µS/cm at 25 °C | Fishman and Friedman (1989) |
| Sulfate | NWQL | IC022 | IC | 0.02 | mg/L | Fishman and Friedman (1989) |
| Total organic carbon | NWQL | COMB9 | High-temperature catalytic oxidation | 1.4 | mg/L | Clesceri and others (1989) |
| Zinc | NWQL | PLM10 | clCP–MS | 2 | µg/L | Garbarino and others (2006) |
The analytical method code is the unique analytical method identifier, which is revised when methods are updated. Identical codes indicate the same method is used.
The chromatographic separation is based on Wallschlager and Bloom (2001), but with modifications to facilitate baseline separation of the organoselenium compounds. The eluting species are then detected and quantified using collision reaction cell equipped with ICP-MS.
References Cited
Arar, E.J., and Collins, G.B., 1997, Method 445.0—In vitro determination of chlorophyll a and pheophytin a in marine and freshwater algae by fluorescence (rev. 1.2, September 1997): Cincinnati, Ohio, U.S. Environmental Protection Agency, Office of Research and Development, National Exposure Research Laboratory, 22 p., accessed December 2023 at https://cfpub.epa.gov/si/si_public_record_report.cfm?Lab=NERL&dirEntryId=309417.
Fishman, M.J., ed., 1993, Methods of analysis by the U.S. Geological Survey National Water Quality Laboratory—Determination of inorganic and organic constituents in water and fluvial sediments: U.S. Geological Survey Open-File Report 93–125, 217 p., accessed March 2022 at https://doi.org/10.3133/ofr93125.
Fishman, M.J., and Friedman, L.C., eds., 1989, Methods for determination of inorganic substances in water and fluvial sediments (3d ed.): U.S. Geological Survey Techniques of Water-Resources Investigations, book 5, chap. A1, 545 p., accessed March 2022 at https://doi.org/10.3133/twri05A1.
Garbarino, J.R., Kanagy, L.K., and Cree, M.E., 2006, Determination of elements in natural-water, biota, sediment, and soil samples using collision/reaction cell inductively coupled plasma-mass spectrometry: U.S. Geological Survey Techniques and Methods, book 5, chap. B1, 87 p., accessed March 2022 at https://doi.org/10.3133/tm5B1.
Patton, C.J., and Gilroy, E.J., 1999, U.S. Geological Survey nutrient preservation experiment—Experimental design, statistical analysis, and interpretation of analytical results: U.S. Geological Survey Water-Resources Investigations Report 98–4118, 73 p., accessed March 2022 at https://doi.org/10.3133/wri984118.
Patton, C.J., and Kryskalla, J.R., 2011, Colorimetric determination of nitrate plus nitrite in water by enzymatic reduction, automated discrete analyzer methods: U.S. Geological Survey Techniques and Methods, book 5, chap. B8, 34 p., accessed March 2022 at https://doi.org/10.3133/tm5B8.
U.S. Environmental Protection Agency [EPA], 1996, Method 1638—Determination of trace elements in ambient water by inductively coupled plasma-mass spectrometry: Washington, D.C., U.S. Environmental Protection Agency, Office of Water, Engineering and Analysis Division, 50 p., accessed December 19, 2023, at https://www.epa.gov/sites/default/files/2015-10/documents/method_1638_1996.pdf.
Wallschlager, D., and Bloom, N.S, 2001, Determination of selenite, selenate, and Selenocyanate in waters by ion chromatography-hydride generation-atomic fluorescence spectrometry (IC-HG-AFS): Journal of Analytical Atomic Spectrometry, v. 16, no. 11, 6 p., accessed March 2026 at https://doi.org/10.1039/B103108M.
Appendix 2. Job Hazard Analysis for Lake Koocanusa and Upper Kootenai River, Montana, Water-Quality Monitoring Program, Water Years 2022–23
Emergency Contacts
Dial 911 or contact the Lincoln County Sheriff, Libby, Montana, 406–293–4112. For help needed, nonemergency, call the U.S. Geological Survey (USGS) Helena office at 406–457–5900, supervisor cell phone, or attempt marine band radio or cell phone communication with Abayance Bay/Mariners Haven Marina (channel 16 is monitored by marinas; phone number is 406–297–0160). If the situation requires law enforcement, contact the Lincoln County Sheriff’s Satellite Office in Eureka, Mont. (406–297–2121). If cellular coverage is unavailable, contact supervisor via text using Garmin InReach global positioning system (GPS) tracker and satellite communicator.
Safety Equipment
Required safety equipment includes the following: Coast Guard-approved personal flotation device (PFD), cable cutters for sounding cable, throw bags, throwable PFD, very high frequency marine radio, air horn, first-aid kit, fire extinguishers, flares, and Garmin InReach GPS tracker and satellite communicator. The boat also will be equipped with overnight emergency supplies including food, water, and shelter. PFDs must always be worn while on the boat.
Recommended safety equipment includes the following: PFD, sun protection, gloves, safety glasses, back support, and if working on construction activities, a hard hat and steel-toed shoes.
Potential Hazards
Servicing Field Equipment.—Electrical and battery hazards. Rocky materials can be hazardous to navigate and extremely slippery when icy or wet. Bats and mice in the streamgage can present hantavirus hazard. Make sure you have hantavirus protection before attempting to clean up rodent droppings. Algae on rocks near the water’s edge can be slippery, even if it looks dry. River regulation by the U.S. Army Corps of Engineers (USACE) can bring water levels up quickly at the streamgage. Concrete steps down to the lower level of the streamgage can become very icy and slippery. Be extremely careful when walking on slippery surfaces. If rocks are snow covered, consider just using binoculars to read the staff from the gage house at higher stages. Notify the USACE at Libby Dam if the gage house needs maintenance.
Motorboat operation in Lake Koocanusa.—Check boat maintenance and user records. File float plan. Check weather forecast before workday begins. Boat operator must evaluate weather forecast and approve lake work. Boat operator must be a USGS Certified Motorboat Operator within the last 5 years. Use extreme caution when towing boat to and from lake. Vehicle driver must have completed USGS Defensive Driving Course within the last 3 years and have experience towing a boat. Verify boat trailer is securely connected to tow vehicle (trailer tongue locked down to ball mount on tow vehicle, trailer safety chains attached to tow vehicle, emergency trailer brake chain attached to tow vehicle; trailer winch strap attached to bow; trailer winch safety chain attached to bow), and grease trailer bearings every trip. Check spare, jack, and lug wrench. File written or verbal float plan with supervisor. If float plan changes owing to factors in the field, be sure to notify supervisor. Check boat trailer lights and stern straps. Ensure boat is in good running condition with adequate fuel and oil for the day. Boat Operator will check boat safety supplies before launching and brief crew on use and location of safety equipment. Boat Operator will verify the stern plug is installed in boat. Be aware of hand placement when lifting, use proper lifting techniques (lift with legs instead of back, and ask for assistance as necessary). Perform all equipment loading in parking area when possible, not at the ramp. A PFD is required to be worn when working in, on, or over any body of water. PFDs will be equipped with reflective tape in accordance with 46 CFR 25.25–15. Boat Operator and crew must watch for obstacles in water and indications of shallow water (watercolor change, breaking waves). Use GPS and available maps to navigate to each monitoring site. Obey boating regulations. Stay clear of sail boats because they have right of way. Use caution when approaching and attaching to monitoring platform. All crew members will watch for changing weather conditions.
Dock Use.— Lines and anchors should be safely stored and inspected prior to use. Watch fingers when tying off to the dock. Boat should be secured at the bow and stern. Motor should be raised to prevent damage to the prop.
Fueling the Boat and Loading/Unloading.— If available, use nonskid surface while loading and unloading. Enter slowly and only at operators’ command. No smoking in or around watercraft while refueling or while fuel is aboard the watercraft.
Cableway Measurement.—75–100-pound weights are required. Always make a thorough visual inspection before using the cable. Watch for fishing lures hanging from the cable when over the water. Always wear PFD on the cableway. Wear back support when moving heavy equipment to and from the cableway. Make sure doors to the shelter are propped open before moving the car so they do not blow shut and hinder re-entrance to the shelter. Always watch ahead for fishing lures hanging from the cable when moving the car. Notify the USACE at Libby Dam if the cableway needs maintenance. Additional safety recommendations specific to this site: road to the gage can be shady and very icy in winter. It is best to come to the site from the west side during the winter to avoid driving the steep hill to the east.
Winter Driving.—Roadway conditions cannot be controlled by employers, but they can promote safe-driving behaviors of employees: train drivers for winter weather conditions and recognize the hazards of winter weather driving (snow/ice covered roads). Maintenance, inspection, and winterization of vehicles should be performed before winter trips. Tires are checked for tread depth and proper inflation, and winter tires installed if needed. Brakes should have proper level of fluid and be evenly balanced. Visibility systems are inspected to ensure all exterior lights, defrosters, and wipers are working properly. Recommended emergency kit is included with each field vehicle, including ice scraper, flashlight with batteries, tow chain, road maps, jumper cables, emergency flares, traction aids (bag of sand), water, and blankets.
If you are stranded in a vehicle, stay inside the vehicle. Response times may be slow during severe weather conditions. Call to notify supervisor and emergency services (911), if needed. Display a brightly colored cloth off the antenna to make passersby aware of distress. Turn on vehicle periodically each hour and run to keep warm. Keep exhaust pipe clear of snow and beware of carbon monoxide poisoning. Do minor exercise to maintain good circulation and watch for signs of frostbite and hypothermia.
Medical Emergencies
All field crew members will be responsible for necessary medications and personal requirements. USGS personnel are required to be current in cardiopulmonary resuscitation (CPR) and first aid training. If a medical emergency happens, it will be most efficient to call 911 via cell phone and request to meet an emergency crew at the nearest marina. If cell phone service is not available, try using the marine band radio to contact the nearest marina and request to meet an emergency crew.
Extreme Emergency Procedures (Sinking Boat, Uncontrollable Boat Fire)
-
1. Press “SOS” button on the Garmin InReach GPS tracker and satellite communicator (this will broadcast rescue request and GPS location to local search and rescue team) and keep Garmin InReach dry with screen facing up.
-
2. Request help on marine band radio by calling out “Mayday” three times and providing location coordinates (use boat GPS system to obtain coordinates) or verbal description of location.
-
3. Launch flares.
-
4. Abandon boat. If cell phone is operational, call 911 (cell phone coverage is only available at the international boundary site and marina).
Appendix 3. Quality-Control Samples Collected
Table 3.1.
Summary of quality-control samples collected in the U.S. Geological Survey Koocanusa/Kootenai water-quality monitoring program, water years 2022–23.[USGS, U.S. Geological Survey; QC, quality control; NA, not applicable]
Conversion Factors
International System of Units to U.S. customary units
Temperature in degrees Celsius (°C) may be converted to degrees Fahrenheit (°F) as follows:
°F = (1.8 × °C) + 32.
Datum
Vertical coordinate information is referenced to the North American Vertical Datum of 1988 (NAVD 88).
Horizontal coordinate information is referenced to the North American Datum of 1983 (NAD 83).
Altitude, as used in this report, refers to distance above the vertical datum.
Supplemental Information
Concentrations of chemical constituents in water are given in either milligrams per liter (mg/L) or micrograms per liter (µg/L).
Water year is the 12-month period from October 1 through September 30 of the following calendar year. The water year is designated by the calendar year in which it ends. For example, water year 2023 is the period from October 1, 2022, through September 30, 2023.
Abbreviations
ADCP
acoustic Doppler current profiler
ASR
analytical service request
CoC
chain of custody
EPA
U.S. Environmental Protection Agency
HCl
hydrochloric acid
MTDEQ
Montana Department of Environmental Quality
NELAC
National Environmental Laboratory Accreditation Conference
NFM
USGS National Field Manual for the Collection of Water-Quality Data
NFQA
National Field Quality Assurance
NWIS
National Water Information System
NWQL
National Water Quality Laboratory
PVC
polyvinyl chloride
QA
quality assurance
QC
quality control
QMS
quality management system
QWDATA
Quality of Water Database
SAP
sampling and analysis plan
USACE
U.S. Army Corps of Engineers
USGS
U.S. Geological Survey
WY–MT WSC
Wyoming-Montana Water Science Center
For more information about this publication, contact:
Director, USGS Wyoming-Montana Water Science Center
3162 Bozeman Avenue
Helena, MT 59601
406–457–5900
For additional information, visit: https://www.usgs.gov/centers/wy-mt-water/
Publishing support provided by the
USGS Science Publishing Network,
Rolla Publishing Service Center
Disclaimers
Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Although this information product, for the most part, is in the public domain, it also may contain copyrighted materials as noted in the text. Permission to reproduce copyrighted items must be secured from the copyright owner.
Suggested Citation
King, L.R., Caldwell Eldridge, S.L., Schaar, M.A., Schmidt, T.S., Chapin, T., and Bussell, A.M., 2026, Sampling and analysis plan for the water-quality monitoring program in Lake Koocanusa and upper Kootenai River, Montana, water years 2022–23: U.S. Geological Survey Open-File Report 2026–1005, 39 p., https://doi.org/10.3133/ofr20261005.
ISSN: 2331-1258 (online)
Study Area
| Publication type | Report |
|---|---|
| Publication Subtype | USGS Numbered Series |
| Title | Sampling and analysis plan for the water-quality monitoring program in Lake Koocanusa and upper Kootenai River, Montana, water years 2022–23 |
| Series title | Open-File Report |
| Series number | 2026-1005 |
| DOI | 10.3133/ofr20261005 |
| Publication Date | April 02, 2026 |
| Year Published | 2026 |
| Language | English |
| Publisher | U.S. Geological Survey |
| Publisher location | Reston, VA |
| Contributing office(s) | Wyoming-Montana Water Science Center, Geology, Geophysics, and Geochemistry Science Center |
| Description | vi, 39 p. |
| Country | United States |
| State | Montana |
| Other Geospatial | Lake Koocanusa and Upper Kootenai River |
| Online Only (Y/N) | Y |
| Additional Online Files (Y/N) | N |


