Evaluation of Pathogen Risks and Testing Considerations for Chinook Salmon Egg Movements Between New Zealand and California
Links
- Document: Report (1.0 MB pdf) , HTML , XML
- Download citation as: RIS | Dublin Core
Acknowledgments
We thank the California Department of Fish and Wildlife (CDFW) for contributing funding to support this project. We thank CDFW and the Winnemem Wintu Tribe (non-federally recognized tribal group) for providing input, reviews, and comments to improve the final version of this report. We also thank the following individuals for contributing their time and expertise to this work: Ian Gardner (University of Prince Edward Island), Lori Gustafson (U.S. Department of Agriculture), Jason Roberts (CDFW), Mark Adkison (CDFW), Melanie Cheung (consultant to the Winnemem Wintu Tribe), Hamish Stevens (Fish & Game New Zealand), Steve McKnight (Fish & Game New Zealand), and Nora Hickey (Washington State University).
Executive Summary
Oncorhynchus tshawytscha (Walbaum in Artedi, 1792; Chinook salmon) were historically abundant in the McCloud River but are now extirpated from this tributary owing to dam construction and lack of passage. Planning efforts to restore populations above Shasta and Keswick Dams are currently underway, including an evaluation of potential source populations. One potential source is New Zealand Chinook salmon, which are believed to have originated from tributaries of the Sacramento River. These fish could be returned to California if reintroduction risks, including risks of pathogen introduction, could be sufficiently mitigated. The U.S. Geological Survey was contracted to provide scientific support for reintroduction efforts, including evaluating the risks of pathogen transmission via the movement of Chinook salmon eggs from New Zealand to the McCloud River. This report estimates pathogen risks associated with egg movement and considers epidemiological and biosecurity measures to minimize these risks.
Pathogen risks associated with the movement of Chinook salmon eggs from New Zealand were evaluated based on pathogen virulence, transmission route, and geographic distribution. These criteria identified 14 moderate- and high-risk pathogens out of the 30 pathogens evaluated. Pathogen species and strains were considered high risk if they have the potential for vertical transmission (that is, transmission from parent to offspring), are moderately or highly virulent, and are exotic to the Sacramento River Basin. According to these criteria, we identified the following pathogens as high risk:
• New Zealand rickettsia-like organisms 1 and 2.—These bacterial pathogens have been associated with mortality events in farmed Chinook salmon from the South Island of New Zealand but have not been detected in other regions.
• Pilchard orthomyxovirus (POMV).—POMV has been detected in Sardina pilchardus (Walbaum, 1792; pilchards) and Salmo salar (Linnaeus, 1758; Atlantic salmon) from the coasts of southern Australia and Tasmania. POMV can cause relatively high mortality rates and may be indirectly transmitted via contaminated water sources.
• Infectious pancreatic necrosis virus (IPNV).—IPNV has a wide geographic distribution and is present in the Sacramento River Basin, but the IPNV-like viruses detected in Australia and New Zealand are unique from those found in the United States.
• Yersinia ruckeri.—This bacterial pathogen is the causative agent of enteric redmouth disease and has a widespread geographic distribution. However, the strains that are present in Australia and New Zealand are unique from those found in North America.
Strategic use of testing and biosecurity measures can minimize pathogen risks associated with the movement of eggs. The most effective measures include iodophor treatment of eggs to remove external pathogens, testing of all the adult fish from which gametes are obtained, and a quarantine period after transport to confirm pathogen testing results. Additional measures to enhance biosecurity could include testing the quarantined fish following emergence and (or) developing a fish health history of the source population through pathogen monitoring.
1. Introduction
At the time of this writing, Federal, State, and Tribal co-managers of salmon are working to re-establish Oncorhynchus tshawytscha (Walbaum in Artedi, 1792; Chinook salmon) above Shasta Dam in the Sacramento River Basin in California (National Oceanic and Atmospheric Administration, 2014; Winnemem Wintu Tribe and others, 2023). Consulting firms (Anchor QEA and HDR) and the U.S. Geological Survey were contracted to provide scientific support for this effort. Several source populations are under consideration for reintroduction, including Chinook salmon from the South Island of New Zealand. These populations originated from eyed eggs that were exported from the Sacramento River Basin in the late 1800s and early 1900s (McDowall, 1994; Yoshiyama and Fisher, 2001; Winnemem Wintu Tribe and others, 2023). Several early introductions to the South Island of New Zealand were successful and resulted in small runs of Chinook salmon that persist to this day. As with any fish stocking or reintroduction effort, however, there is potential for inadvertent movement of pathogens if Chinook salmon from New Zealand were to be reintroduced into the Sacramento River Basin.
The introduction of a lethal communicable disease above the Shasta Dam could have significant negative impacts on Chinook salmon and O. mykiss (Walbaum, 1792; rainbow trout) populations in the Sacramento River Basin that are listed under the U.S. Endangered Species Act (16 U.S.C. 1531–1544), as well as other native fishes throughout the region. Because Chinook salmon are anadromous, there is potential for pathogens to be transmitted in both freshwater and marine environments. Therefore, evaluating and minimizing the risk of pathogen introduction is a critical step in reintroduction planning (Anderson and others, 2014). By considering existing aquatic pathogen surveillance data from both California and New Zealand, in addition to implementing strategic testing and biosecurity measures for transported eggs, salmon co-managers can minimize the risks associated with pathogen introduction during reintroduction.
Pathogen surveillance is currently being conducted above Shasta Dam, including in the McCloud River (U.S. Fish and Wildlife Service, 2001), which will aid in understanding current pathogen distributions in the system. However, the sample size and spatiotemporal scale of these surveillance efforts are not yet sufficient to determine the pathogen risks above Shasta Dam with confidence. Therefore, for the purposes of this report, we do not distinguish between pathogens detected above Shasta Dam from those detected below it in the Sacramento River Basin. Presently, tissue testing of Chinook salmon in New Zealand is ongoing to establish a fish health history and to improve confidence that these populations are free from pathogens of potential concern. This includes sampling from post-spawned Chinook salmon in tributaries of the Rangitata River (Deep Creek and Deep Stream) and the Rakaia River (Mellish Stream). Samples are also collected from juvenile Chinook salmon from Lake Heron, which are from the Rakaia River population that spawns in Mellish Stream. Both Federal and State fish importation requirements (50 CFR §16.13; Fish and Game Commission, 2025) guided the selection of analytical tests, which are currently conducted by the Washington Animal Disease Diagnostic Laboratory in Pullman, Washington.
The following are important considerations for evaluating the pathogen risks associated with the reintroduction of Chinook salmon from New Zealand:
-
• For pathogens of potential concern, what is the risk of their introduction and establishment via fertilized egg shipments from New Zealand Chinook salmon? This consideration is addressed in sections 2 and 3 of this report.
-
• How can pathogen surveillance of source populations, egg disinfection, and the quarantine, monitoring, and testing of fish after hatching reduce the risk of introducing infectious pathogens of potential concern from New Zealand Chinook salmon to California? This consideration is discussed in sections 4 and 5 of this report.
2. Risk Assessment Criteria for Fish Pathogens
We evaluated 30 pathogens that have been associated with disease in fish, particularly in salmonids, and are of potential concern for the reintroduction of Chinook salmon from New Zealand to the McCloud River (table 1). These pathogens of potential concern included 12 viruses, 12 bacteria, and 6 parasites. Several criteria were used to evaluate the relative risk associated with the introduction of each pathogen, including the degree of virulence, potential for vertical transmissibility, and whether the pathogen is endemic or exotic to the Sacramento River Basin. These criteria were used to classify pathogens according to the relative risk they pose to fish in the receiving watershed, which may inform the design of testing and biosecurity measures for egg movement.
Table 1.
Pathogens of potential concern that may be introduced via the movement of Chinook salmon eggs from New Zealand to California and their relative risk designation as determined by their potential for vertical transmission, virulence, and presence in the Sacramento River Basin and surrounding area.[NZ, New Zealand]
| Pathogens of potential concern |
Pathogen of salmonids |
Vertically transmitted |
Virulence to fish hosts |
Detected in NZ region |
Present in or around Sacramento River Basin |
Relative risk designation |
References |
|---|---|---|---|---|---|---|---|
| Pilchard orthomyxovirus (POMV) | Yes | Unknown | High | Yes‡ | No | High | Godwin and others, 2020; Mohr and others, 2020; Samsing and others, 2021; Samsing and others, 2022 |
| Infectious pancreatic necrosis virus (IPNV) | Yes | Yes | High | Yes | No | High | Tisdall and Phipps, 1987; Bootland and others, 1991; Dopazo, 2020 |
| Oncorhynchus masou virus (OMV) | Yes | Probable | Moderate | No | No | Moderate | Kimura and others, 1981; Anders and Yoshimizu, 1994; Yoshimizu and others, 1995; Yoshimizu, 2016 |
| Infectious hematopoietic necrosis virus (IHNV) | Yes | Probable | High | No | Yes | Moderate | Dixon and others, 2016; Bendorf and others, 2022 |
| Infectious salmon anemia virus (ISA) | Yes | Yes | High | No | No | Moderate | Mardones and others, 2014; Nylund and others, 2019 |
| Herpesvirus salmonis (HPV-1) | Yes | Yes | Moderate | No | No | Moderate | Hanson and others, 2011; Eaton and Hendrick, 1994 |
| Iridoviruses | Yes | Probable | Low | Yes | Yes | Moderate | Georgiadis and others, 2001; Whittington and others, 2010; Holopainen and others, 2012 |
| Salmonid alphavirus | Yes | Yes | Low | No | No | Moderate | Boscher and others, 2006; McLoughlin and Graham, 2007; Bratland and Nylund, 2009 |
| Viral nervous necrosis virus (VNNV) | No | Yes | High | Yes | Yes† | Moderate | Azad and others, 2006; Doan and others, 2017 |
| Piscine orthoreovirus (PRV) | Yes | No | Moderate | No | No | Negligible | Palacios and others, 2010; Wessel and others, 2020 |
| Viral erythrocytic necrosis virus (VENV) | Yes | No | High | No | No | Negligible | Hershberger and others, 2009; Winton and Hershberger, 2016; Pagowski and others, 2019 |
| Viral hemorrhagic septicemia virus (VHSV) | Yes | No | High | No | Yes† | Negligible | Batts and others, 2014; Mohammadisefat and others, 2023 |
| New Zealand Rickettsia-like organisms (NZ-RLO) | Yes | Unknown | Moderate | Yes | No | High | Brosnahan and others, 2017; Brosnahan and others, 2019; Jaramillo and others, 2024 |
| Yersinia ruckeri (enteric redmouth disease) | Yes | Unknown | High | Yes | Yes | High | Glenn and others, 2015; Barnes and others, 2016 |
| Piscirickettsia salmonis (rickettsiosis of finfish) | Yes | Yes | High | No | Yes | Moderate | Fryer and Hedrick, 2003; Larenas and others, 2003; Corbeil and Crane, 2019 |
| Renibacterium salmoninarum (bacterial kidney disease) | Yes | Yes | High | No | Yes | Moderate | Wiens, 2011; Delghandi and others, 2020 |
| Aeromonas salmonicida (furunculosis) | Yes | No | Moderate | No | Yes | Negligible | Davis, 1947; Hirvelä-Koski, 2005; Menanteau-Ledouble and others, 2016 |
| Edwardsiella ictaluri (enteric septicemia of catfish) | No | No | High | No | No | Negligible | Baxa and others, 1990; Machimbirike and others, 2022 |
| Francisella sp. | Yes | No | High | No | No | Negligible | Birkbeck and others, 2011; Colquhoun and Duodu, 2011 |
| Lactococcus garvieae | Yes | No | Moderate | Yes | Yes‡ | Negligible | Eldar and others, 1999; Vendrell and others, 2006; Environmental Protection Authority, 2020 |
| Lactococcus petauri | Yes | No | Moderate | No | Yes‡ | Negligible | Saticioglu and others, 2023; Heckman and others, 2024 |
| Tenacibaculum maritimum (saltwater mouthrot) | Yes | No | Moderate | Yes | Yes† | Negligible | Nowlan and others, 2020; Mabrok and others, 2023 |
| Vibrio (Listonella) anguillarum | Yes | No | High | Yes | Yes | Negligible | Eguchi and others, 2000; Hickey and Lee, 2018 |
| Weissella ceti | Yes | No | Moderate | No | No | Negligible | Good and others, 2016; Duman and others, 2023 |
| Nucleospora salmonis (microsporidia) | Yes | Yes | Moderate | No | Yes | Moderate | Hendrick and others, 2012; Kent and others, 2014 |
| Neoparamoeba perurans (amoebic gill disease) | Yes | No | Moderate | Yes | Yes† | Negligible | Young and others, 2008; Oldham and others, 2016; Johnson-Mackinnon, 2019 |
| Ichthyophonus hoferi | Yes | No | High | No | Yes | Negligible | Patterson, 1996; Kocan and others, 2004 |
| Sphaerothecum destruens (Rosette Agent) | Yes | No | High | No | No | Negligible | Andreou and others, 2012; Gozlan and Combe, 2023 |
| Tetracapsuloides bryosalmonae (myxozoa) | Yes | No | High | No | Yes | Negligible | Sterud and others, 2007; Abd-Elfattah and others, 2014 |
| Myxobolus cerebralis (whirling disease) | Yes | No | High | Yes | Yes | Negligible | Hewitt and Little, 1972; Bartholomew and Reno, 2002; Gilbert and Granath, 2003 |
2.1. Vertical Transmission
Vertical transmission is the infection of the next generation of fish via the movement of pathogens into fertilized eggs from milt or ovarian fluids. Pathogens that enter eggs via vertical transmission are able to avoid standard iodophor inactivation procedures. The potential for a pathogen to be vertically transmitted increases its risk of spreading via egg shipments. Laboratory experiments have demonstrated that vertical transmission can occur with certain viral, bacterial, and eukaryotic pathogens. In this assessment, vertical transmission is designated as “Yes” for pathogens with published evidence to support vertical transmission, “No” for pathogens with published evidence that they cannot be vertically transmitted, “Probable” for pathogens with suggestive evidence for vertical transmission (for example, unofficial reports or studies from closely related taxa), and “Unknown” when a determination cannot be made owing to a lack of information, such as for newly described pathogens (table 1). For the purposes of this assessment, Yes, Probable, and Unknown are treated equally for risk ranking (fig. 1).
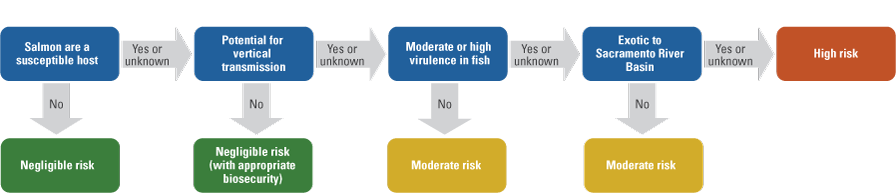
Flow chart for assigning levels of relative risk to pathogens of potential concern that may be imported from New Zealand to the Sacramento River Basin by movement of fertilized Chinook salmon eggs.
2.2. Virulence Criteria
Virulence of a pathogen is highly dependent on environmental conditions, species, and other factors, and thus can be challenging to categorize. To broadly evaluate the virulence of pathogens of potential concern (table 1), we considered (1) whether it is a primary pathogen that causes disease in fish with or without the presence of significant stressors, (2) whether it causes significant mortality in aquaculture settings, and (3) whether there is evidence that it causes mortality or morbidity in wild fish. Pathogens were given a “low virulence” designation if they are unlikely to cause mortality in the absence of significant stress (for example, thermal or crowding stress), a “moderate virulence” designation if they cause notable mortality in aquaculture settings but not in wild fish populations, and a “high virulence” designation if they cause high mortality in aquaculture settings or are known to cause disease, morbidity, or mortality in wild populations.
2.3. Pathogen Testing in New Zealand
New Zealand’s aquaculture industry, in addition to environmental changes such as climate change, invasive species, and pollution that may alter disease dynamics, highlights the growing need to understand aquatic disease ecology in this region (Lane and others, 2020). Limited availability of pathogen testing data from Chinook salmon in New Zealand, combined with the historical introduction of various salmonid species from a wide geographic range (Closs, 2024), supports the need for pathogen monitoring to better understand fish health risks. New Zealand is the world’s largest producer of farmed Chinook salmon, yet publicly available reports of pathogen testing for this species are limited for both aquaculture and naturally produced populations. As a result, it is unclear whether certain pathogens are truly absent from New Zealand or have simply not been reported. For example, there have not been any published reports of Chinook salmon in New Zealand being infected with Renibacterium salmoninarum, the causative agent of bacterial kidney disease, even though this pathogen has an otherwise worldwide distribution (Brynildsrud and others, 2014). This risk assessment includes whether a pathogen has previously been detected in New Zealand or Australia based on the available public information (table 1), but because of the limited availability of pathogen testing information in New Zealand, reported presence did not influence a pathogen’s risk designation (table 1; fig. 1).
3. Relative Risk Categories for Fish Pathogens
Each pathogen species or strain of potential concern was assigned to one of three categories of relative risk (table 1; fig. 1). For this assessment, we assume that eyed eggs are the transported life stage and that all eggs are iodophor treated as a minimum biosecurity measure prior to movement. A “negligible risk” designation represents the lowest level of risk and represents pathogens that do not infect salmon or are not transmissible via appropriately disinfected eggs. A “moderate risk” designation is applied where transfer via eggs is possible, but virulence is low, and the pathogen species or strain is already present in the Sacramento River Basin. A “high risk” designation is appropriate for pathogen species or strains of unknown, moderate, or high virulence, with confirmed, possible, or unknown vertical transmissibility, that are exotic to the receiving watershed. High-risk pathogen species and strains have the potential to negatively affect the survival of native fish if introduced to the McCloud River.
4. Profiles of High-Risk Pathogens
Based on the criteria described in section 2 and the information in table 1, we identified four pathogens that could present a high degree of risk to fish species in the Sacramento River Basin and surrounding area if they were to be imported via the movement of Chinook salmon eggs. Here, we provide additional information on the biology, epidemiology, and available diagnostic tests for each of these four pathogens.
4.1. Rickettsia-Like Organisms 1 and 2 (NZ-RLO1, NZ-RLO2)
In 2015, a rickettsia-like organism was identified in moribund farmed Chinook salmon from the South Island of New Zealand. The bacterium, designated “New Zealand rickettsia-like organism 1” (NZ-RLO1), was found to be similar to an rickettsia-like organism isolated from diseased Salmo salar (Linnaeus, 1758; Atlantic salmon) in Tasmania, Australia (Corbeil and others, 2005) based on comparison between partial 16S RNA and internal transcribed spacer (ITS) genes (Brosnahan and others, 2017; Gias and others, 2018). A second strain, designated “New Zealand rickettsia-like organism 2” (NZ-RLO2), was subsequently isolated from the same farm (Gias and others, 2018). Both strains were identified during a high mortality event in a period of stressful environmental conditions (seawater temperatures greater than 17.5 degrees Celsius [°C]). A recent vaccine development study found that juvenile Chinook salmon intraperitoneally (IP) injected with NZ-RL01 developed more severe disease and higher morbidity relative to juveniles IP injected with NZ-RLO2 (Jaramillo and others, 2024). For NZ-RLO1, morbidity differed by route of infection, with unvaccinated fish experiencing mortality rates of 79 percent (IP injection) and 12 percent (exposure via bath immersion). IP-infected fish displayed peritonitis, particularly around the adipose tissue, immersion-infected fish displayed dermal lesions, and both exhibited hepatocellular necrosis and degeneration. In a separate study, the pathogenicity of NZ-RLO2 was evaluated by IP injecting Chinook salmon smolts with high, medium, and low doses of bacteria (Brosnahan and others, 2019). Mortality was dose dependent, with 100-percent mortality observed in high and medium dose treatments and 63-percent mortality in the low dose treatment after 30 days. Mortality also increased following a temperature spike, suggesting temperature dependence. Diseased fish consistently displayed inflammation of the pancreas and surrounding adipose tissue, as well as dose-dependent pathology of the kidney, liver, spleen, and to a lesser degree, brain and heart (Brosnahan and others, 2019).
Rickettsial bacterial infections are commonly detected from axenic tissue samples with the use of antibiotic-free cell cultures derived from the host species (for example, Chinook salmon embryo [CHSE]-214). Cytopathic effects (CPE) and intracellular bacterial staining methods are used to reveal the presence of rickettsial bacteria to be later identified by genetic sequencing. NZ-RLO1 has also been cultured in CHSE cells at 15 °C (Jaramillo and others, 2024), and NZ-RLO2 has been cultured in CHSE-214, Epithelioma papulosum cyprinid (EPC), and several non-fish cell lines, with optimal growth in fish cell lines observed at 15 °C (Gias and others, 2018). A quantitative polymerase chain reaction (qPCR) test for Piscirickettsia salmonis (Corbeil and others, 2003) has been used for NZ-RLO1 and NZ-RLO2 (Jaramillo and others, 2024). Additionally, two qPCR assays (designed to be used in conjunction) are available that are specific to NZ-RLO2 (Gias and others, 2018).
4.2. Pilchard Orthomyxovirus (POMV)
Pilchard orthomyxovirus (POMV) is a virus that was first isolated from apparently healthy Sardina pilchardus (Walbaum, 1792; pilchards) off the South Australian coast in 1998. The virus was subsequently found in marine farmed Atlantic salmon in Tasmania during routine surveillance in 2006, where it has been linked with disease outbreaks since 2012 (Godwin and others, 2020; Mohr and others, 2020). POMV is related to infectious salmon anemia virus but has relatively low homology with this virus and other orthomyxoviruses, suggesting POMV may represent a new genus (Mohr and others, 2020). POMV can affect all life stages of Atlantic salmon and causes pathological effects to major organs, including the liver, kidney, spleen, heart, and eyes (Godwin and others, 2020). Experimental infections of juvenile Chinook salmon have resulted in relatively high morbidity rates (IP injection: 12–74 percent; immersion: 63 percent; cohabitation: 12.5–46 percent). Laboratory research has shown that transmission can occur indirectly via contaminated water sources, and direct contact between fish is not needed (Samsing and others, 2021). It is not known whether Chinook salmon from the South Island region of New Zealand migrate past affected areas in Tasmania or the South Australian Coast. However, pilchards are highly migratory, and previous epizootics of other pathogens in this species have spanned both Australia and New Zealand (Whittington and others, 1997); thus, it is possible that Chinook salmon in New Zealand could be exposed via the natural migration of pilchards in this region.
POMV causes CPE in the CHSE-214 cell line and can also replicate in Atlantic salmon kidney cells. Virus replication is temperature dependent, with progressively higher in vitro replication between 10 and 20 °C (Godwin and others, 2020). A highly sensitive real-time quantitative polymerase chain reaction (RT-qPCR) test that targets two viral genes has recently been validated and is available for suspected CPE and (or) testing directly from tissues (Samsing and others, 2022). This test can detect concentrations as low as one plasmid copy per microliter and is significantly more sensitive than previously developed tests for detecting infection in field-collected samples.
4.3. Infectious Pancreatic Necrosis Virus (IPNV)
IPNV is an aquatic birnavirus that has a wide geographical distribution. The disease was first reported in Salvelinus fontinalis (Mitchill, 1814; brook trout) from Canada (M’Gonigle, 1941) and has since been detected on every continent except Antarctica (Dopazo, 2020). IPNV causes necrosis of the pancreatic tissue and may be isolated from the spleen and kidney of affected fish. IPNV tolerates a wide range of environmental conditions and infects multiple host species and life stages, but juvenile salmonids tend to be the most susceptible (Dopazo, 2020). Infected fish can become carriers, thus increasing the risk of vertical transmission (Bootland and others, 1991). The virulence of IPNV also varies significantly among strains and is difficult to predict. Moreover, salmonid hosts may be resistant to one strain of IPNV but susceptible to another (Hillestad and others, 2021). A type of IPNV was isolated from Chinook salmon returning to the Rakaia River in the South Island of New Zealand in the 1980s and has never been detected again (Tisdall and Phipps, 1987), but an IPNV-like virus has since been detected in multiple species in Australia (Crane and others, 2000). It is likely that the birnaviruses detected in New Zealand and Australia are unique from IPNV strains found in the United States and thus could be treated as exotic pathogens to California.
IPNV causes CPE in CHSE-214 and other fish cell lines (Kelly and others, 1978). Multiple molecular diagnostics are available, including RT-qPCR and nested polymerase chain reaction (Eissler and others, 2025). Virus serotypes can also be identified via a neutralization test following isolation from cell culture (Dopazo and Barja, 2002).
4.4. Enteric Redmouth Disease (Yersinia ruckeri)
Yersinia ruckeri is a widely distributed bacterial pathogen that was originally isolated from rainbow trout in the United States (Ross and others, 1966) and has been detected in trout above Shasta Dam (U.S. Fish and Wildlife Service, 2001). Virulence mechanisms differ among strains of Y. ruckeri, with differences often corresponding to geographic origin (Kumar and others, 2015). The Y. ruckeri lineages isolated from Australia and New Zealand are distinct from North American strains, and it is unclear how these strains would affect salmonid species in the Sacramento River Basin if they were to be introduced (Barnes and others, 2016). Y. ruckeri has consistently caused mortality in Atlantic salmon aquaculture in Tasmania since 1987, but an effective vaccine is now available for aquacultural use (Barnes and others, 2016). In New Zealand, Y. ruckeri has occurred sporadically in Chinook salmon since 1989 (Anderson and others, 1994), but this strain appears to have low virulence and has not been a major cause of mortality; therefore, vaccination has not been considered necessary in New Zealand (Barnes and others, 2016).
In general, diseased fish may become anorexic and lethargic, with hemorrhage around the mouth (leading to the common name, enteric redmouth disease) and develop inflammation and septicemia in most organs (Kumar and others, 2015). Transmission rates for Y. ruckeri are highly dependent on environmental conditions, as the bacteria are typically released from the lower intestine when carrier fish become stressed (for example, by elevated temperatures or crowding; Kumar and others, 2015). The bacteria can survive in fresh or brackish water for more than 4 months (Thorsen and others, 1992), and some strains are able to form biofilms that may cause recurrent outbreaks. Although early vaccination treatments in aquaculture systems are generally very protective, such preventative measures are impractical for the protection of wild fish populations. In addition to horizontal and environmentally mediated transmission, there is a report that Y. ruckeri has the potential to be vertically transmitted (Glenn and others, 2015). Glenn and others (2015) detected Y. ruckeri DNA in unfertilized eggs and ovarian fluid; however, they did not detect viable bacteria from eggs, and therefore, vertical transmission remains unconfirmed.
Multiple methods and diagnostic tests are available for Y. ruckeri. The bacterium can be isolated using several different growth media and at a wide range of temperatures, with optimal growth occurring between 20 and 28 °C (Kumar and others, 2015). Multiple serology and molecular detection methods exist, including PCR-based tests that can detect low levels of infection that may occur in asymptomatic carriers (Gibello and others, 1999; Bastardo and others, 2012).
5. Risk Reduction Approaches
To mitigate the effects of pathogens of potential concern, managers may choose to employ several epidemiology and biosecurity measures. These measures include pathogen surveillance of the source population (New Zealand Chinook salmon), disinfection of transported eggs, and the quarantine, monitoring, and testing of juveniles produced from transported eggs.
5.1. Epidemiology and Testing Approaches
Designing an efficient sampling scheme to provide confidence in the disease-free status of transported eggs is critical because of the limited number of New Zealand Chinook salmon potentially available for disease testing. One approach that may reduce the need for large-scale population-level surveillance when eggs are the transported life stage is testing all parents for vertically transmitted pathogens, combined with egg disinfection to reduce the possibility of external contamination from water sources. In this manner, a high degree of confidence (constrained only by testing sensitivity) in the disease-free status of the transported gametes can be achieved. To overcome issues with unknown or inadequate testing sensitivity, multiple tissue samples from each individual can be collected and tested to reduce the possibility of generating a false negative result. In some scenarios, 100-percent testing of broodstock combined with appropriate disinfection protocols may provide sufficient confidence in the disease freedom of transported fish or gametes. An important consideration for this approach is the need to contain the eggs and hatched fish in quarantine until the testing has been completed. This provides an additional opportunity for sampling and testing this population directly (more information on this in section 5.3).
If managers require information on disease prevalence in the source population in addition to the transported individuals or gametes, surveillance strategies should be carefully considered. Sample size needs can be calculated given the size of the population of interest, expected disease prevalence (percentage of infected individuals), test sensitivity (percentage of infected individuals that will test positive), and desired level of confidence (Cannon, 2001). In scenarios where there is insufficient baseline data to define expected disease prevalence, the maximum acceptable level of prevalence (and associated level of confidence) may be determined based on the risk tolerance of the decisionmakers. The percentage of the population that needs to be sampled increases as population size, test sensitivity, or expected prevalence decrease or as the desired confidence level increases. The American Fisheries Society–Fish Health Section Blue Book recommends sampling sufficient numbers of fish to provide 95-percent confidence of disease freedom. For large population sizes (like those greater than 100,000) and an assumed pathogen prevalence of 5 percent, this generally translates to 60 fish (American Fisheries Society–Fish Health Section, 2014). If pathogen prevalence is assumed to be 2 percent, then a sample size of 150 is required to achieve the same confidence level. For sampling free-ranging populations, additional factors such as geography, life history, and age structure of the population may impact sample size considerations.
For population-level pathogen surveillance, sample size needs may be reduced through several considerations. The first of these is risk-based sampling, where individuals from a high-risk group (for example, post-spawned adults) are sampled preferentially (Stärk and others, 2006). This can result in higher detection rates, and thus a smaller number of fish need to be sampled to determine whether a disease is present in the population. For example, Gustafson and others (2005) found that targeting moribund fish for infectious salmon anemia virus surveillance resulted in significantly higher detection probability than sampling randomly from the population. In Chinook salmon, adult fish become immunosuppressed and highly susceptible to infections prior to spawning (Dolan and others, 2016); therefore, risk-based sampling targeted at sexually mature or post-spawned moribund adults could reduce the number of samples needed to detect disease in a population. The strategy of sampling moribund fish to reduce overall sample size needs has been recommended for research facilities (Kent and others, 2011) and aquaculture facilities (Gustafson and others, 2015). Because sample size calculations depend on expected prevalence, sampling moribund fish or an age class that is more likely to be infected can significantly decrease sample size needs. Sample size can be adjusted if information is available on the expected prevalence of each pathogen within the population as a whole versus the subpopulation targeted for sampling. Gustafson and others (2015) provided a hypothetical scenario in which a moribund group was expected to have a 5-percent prevalence of a disease, relative to the expected 2 percent in the population at large. In this scenario, the same confidence level could be achieved by sampling 69 moribund fish versus a random sample of 175 fish from the wider population. On the other hand, if a pathogen is presumed to be equally distributed across host life stages, then sampling could likewise include both juveniles and adults to achieve confidence in disease-free status. The second consideration is sampling over time, or the use of existing surveillance data, as long-term data can enhance confidence in the disease-free status of a population (Gustafson and others, 2024). Although the frequency and sample size required for testing depend on the distribution of the target pathogen, repeated sampling that is carried out regularly over time with negative results tends to decrease the intensity of sampling that must be conducted to maintain confidence in disease freedom.
5.2. Egg Disinfection
Disinfection treatment is an important biosecurity measure that is critical for reducing the risk of transporting infectious bacteria, viruses, and parasitic organisms in egg shipments. The efficacy of these treatments depends on whether the pathogen is vertically transmitted. For example, infectious pancreatic necrosis virus (IPNV; Bootland and others, 1991), infectious hematopoietic necrosis virus (Mulcahy and Pascho, 1985), and salmonid alphaviruses (McLoughlin and Graham, 2007; Bratland and Nylund, 2009) all have shown the potential for vertical transmission, indicating that external disinfection is not considered sufficient to prevent transmission of the pathogen via infected eggs. For pathogens that are not vertically transmitted, contamination of eggs can often be eliminated via external disinfection. Chinook salmon eggs can be disinfected by povidone iodine immediately post-fertilization during water hardening prior to transport and again at the eyed egg stage upon receipt at the target facility to significantly reduce potential biological contaminants on the surface of the egg (Huysman and others, 2018). The U.S. Fish and Wildlife Service (2022)’s recommended treatment protocol is to use a dual disinfection of fish eggs in 50-milligram-per-liter (mg/L) iodine solution for 30 to 60 minutes during water hardening, followed by a secondary disinfection in 100 mg/L iodine for 10 to 30 minutes upon arrival. External horizontally transmitted pathogens, such as Aeromonas salmonicida, on the surface of eggs from infected broodstock or water sources can be consistently inactivated if the recommended disinfection protocol is followed without reusing iodine solutions for each batch of eggs (Cipriano and others, 2001). Although vertically transmitted pathogens may be resistant to iodine egg treatments, following established protocols for disinfection is still critical for minimizing the risk of other pathogens. Care must be taken to follow protocols and achieve the proper concentration of iodine solution to ensure effective treatment of the egg surface. Following treatment, any water used with eggs prior to transport should be sterilized to avoid contamination of eggs with the untreated source water.
5.3. Quarantine, Monitoring, and Testing
Holding transported eggs and juvenile fish in quarantine for a period after transport can enhance confidence in pathogen freedom and reduce the risk of pathogen introduction via movement of eyed eggs. This may be particularly important when adequate testing of the source fish population and (or) broodstock is not available. Appropriate quarantine periods may vary among pathogens depending on the relative risk and incubation period of each pathogen within the host species of interest (Humphrey, 1994). It is also important to note that holding temperature can affect pathogen incubation time; therefore, longer quarantines may be necessary for fish held at low temperatures. For example, standard holding periods of 30 days are often used for zoo and aquarium fish, but Boerman and others (2006) recommend doubling this holding period when fish are held at 12–15 °C. Pathogens that have a long incubation period, such as R. salmoninarum, may extend the quarantine period several more weeks if there is an indication that the broodstock were infected (Munson and others, 2010). During quarantine, risk-based sampling can be applied by testing any fish that develop signs of disease. If no signs of disease emerge in the transported population, random sampling of fish using the epidemiological principles discussed in section 5.1 may be used to achieve the desired level of confidence in pathogen freedom. Exposing a subpopulation of the quarantined fish to a stressor before sacrificing them for fish health testing could improve the chances of isolating a pathogen, if one is present at levels lower than the detection threshold. Temperature stress or treating fish with dexamethasone, known to lower the innate immune system of fish (Lovy and others, 2008), may increase the replication or pathological effects of low-level pathogens, thus improving their detection capability. The impacts of temperature and dexamethasone may vary based on the biology of each pathogen.
5.4. Approaches for Identifying Unknown Risks
Because of the possible presence of pathogens not specifically tested for and the large geographic distance of the proposed egg movement, managers may wish to include additional methods to detect infectious agents not considered in this pathogen risk assessment. Many pathogen testing protocols are specific to the detection of only that pathogen species, though methods that are less specific have the advantage of detecting a range of infectious agents. For example, the culture of viruses and bacteria in cell culture and (or) growth media can provide a broad assessment of potential pathogens and could be conducted alongside other diagnostics for specific pathogens. Considering that unknown pathogens may be a risk associated with the international movement of eggs, managers may require the characterization of unknown agents that cause CPE in viral cell culture assays and the identification of any bacteria isolated. These organisms could then be characterized to better understand if they are considered pathogens of fish and thus if they pose a risk for egg movements. Care should be taken while interpreting bacterial isolates that come from spawning adult fish, as it is possible to isolate a variety of opportunistic and inconsequential microbes. If a pathogen is detected, characterization may include identification of the strain or genetic type to determine whether it differs from strains found in the receiving watershed.
Methods that are not biased towards detecting certain pathogens and instead focus on recognizing a disease condition in the fish can alert investigators to a disease and potential unknown infectious agent. This may be conducted by careful examination of fish for gross external and internal lesions during necropsy. Disease-causing agents may be associated with certain gross lesions, like enlarged organs, hemorrhage, ulcerations, cysts, granulomas, and other inflammatory processes, which may be identified through careful observation during a necropsy. If gross lesions are observed, histology is useful to further determine the etiology of these lesions. Histological examination of organs may be used to evaluate for microscopic anomalies or pathology, which may be otherwise missed in gross observation. This enhanced evaluation of samples can complement fish health testing to improve confidence that unknown disease agents are not present in the source fish population.
6. Combined Measures to Minimize Risk
The risk of importing pathogens via egg movements can be reduced through a combination of the biosecurity and testing measures outlined in section 5. Though it is not possible to eliminate risk completely, risk can be minimized by selecting a combination of these measures. Disinfecting eggs immediately post-fertilization prior to transport and again post-transport (eyed eggs) is a low-cost and effective method to eliminate pathogens that occur on the egg surface; therefore, managers may choose to use this method for all transported eggs. However, owing to the potential for vertical transmission of certain pathogens, the overall risk of introducing pathogens to California from New Zealand via iodine-treated eggs is still high. If one additional measure were implemented (100-percent broodstock testing and quarantine until testing is completed; or quarantine, monitoring, and testing of juveniles; or 3–5 years of population surveillance), the cumulative risk of pathogen introduction via egg transport could be downgraded to moderate. To achieve a low cumulative risk of importing any pathogens via disinfected eggs, managers could implement 100-percent broodstock testing combined with quarantine, monitoring, and testing of juveniles. This approach best ensures that adequate testing has been completed directly on the population in question. If monitoring is not achievable at this level, then conducting testing of the source population to develop a 3–5-year fish health history can increase confidence that certain pathogens are not present within the source population.
7. Conclusions
In this report, we evaluated the pathogen-associated risks of moving Chinook salmon eggs from New Zealand to the Sacramento River Basin. Potential pathogens of concern were ranked based on pathogen virulence, transmission route, and geographic distribution. Although several pathogens were identified that pose a high risk to the recipient ecosystem, a number of epidemiology and biosecurity methods are available to reduce the risk to a level that is acceptable to salmon co-managers. Measures to reduce the risk of pathogen importation include pathogen surveillance of the source population, disinfection of transported eggs, and the quarantine, monitoring, and testing of juveniles prior to release.
References Cited
Abd-Elfattah, A., Fontes, I., Kumar, G., Soliman, H., Hartikainen, H., Okamura, B., and El-Matbouli, M., 2014, Vertical transmission of Tetracapsuloides bryosalmonae (Myxozoa), the causative agent of salmonid proliferative kidney disease: Parasitology, v. 141, no. 4, p. 482–490, accessed September 1, 2025, at https://doi.org/10.1017/S0031182013001650.
American Fisheries Society–Fish Health Section, 2014, Fish health section blue book—Suggested procedures for the detection and identification of certain finfish and shellfish pathogens (2020 ed.): American Fisheries Society, accessed September 8, 2025, at https://units.fisheries.org/fhs/fish-health-section-blue-book-2020/.
Anders, K., and Yoshimizu, M., 1994, Role of viruses in the induction of skin tumours and tumour-like proliferations of fish: Diseases of Aquatic Organisms, v. 19, p. 215–232, accessed September 1, 2025, at https://doi.org/10.3354/dao019215.
Anderson, J.H., Pess, G.R., Carmichael, R.W., Ford, M.J., Cooney, T.D., Baldwin, C.M., and McClure, M.M., 2014, Planning Pacific salmon and steelhead reintroductions aimed at long-term viability and recovery: North American Journal of Fisheries Management, v. 34, no. 1, p. 72–93, accessed September 1, 2025, at https://doi.org/10.1080/02755947.2013.847875.
Andreou, D., Arkush, K.D., Guégan, J.-F., and Gozlan, R.E., 2012, Introduced pathogens and native freshwater biodiversity—A case study of Sphaerothecum destruens: PLoS One, v. 7, no. 5, accessed September 1, 2025, at https://doi.org/10.1371/journal.pone.0036998.
Azad, I., Jithendran, K., Shekhar, M., Thirunavukkarasu, A., and De la Pena, L., 2006, Immunolocalisation of nervous necrosis virus indicates vertical transmission in hatchery produced Asian sea bass (Lates calcarifer Bloch)—A case study: Aquaculture (Amsterdam, Netherlands), v. 255, no. 1–4, p. 39–47, accessed September 1, 2025, at https://doi.org/10.1016/j.aquaculture.2005.04.076.
Barnes, A.C., Delamare-Deboutteville, J., Gudkovs, N., Brosnahan, C., Morrison, R., and Carson, J., 2016, Whole genome analysis of Yersinia ruckeri isolated over 27 years in Australia and New Zealand reveals geographical endemism over multiple lineages and recent evolution under host selection: Microbial Genomics, v. 2, no. 11, accessed September 1, 2025, at https://doi.org/10.1099/mgen.0.000095.
Bartholomew, J.L., and Reno, P.W., 2002, The history and dissemination of whirling disease: American Fisheries Society Symposium, v. 26, p. 1–22. [Also available at https://www.researchgate.net/publication/285691216_The_history_and_dissemination_of_whirling_disease.]
Bastardo, A., Ravelo, C., and Romalde, J.L., 2012, Multilocus sequence typing reveals high genetic diversity and epidemic population structure for the fish pathogen Yersinia ruckeri: Environmental Microbiology, v. 14, no. 8, p. 1888–1897, accessed September 1, 2025, at https://doi.org/10.1111/j.1462-2920.2012.02735.x.
Batts, W.N., Lovy, J., Getchell, R., Faisal, M., Standish, I., Warg, J.V., Phelps, N.B., Glenney, G., and Winton, J.R., 2014, Viral hemorrhagic septicemia (updated June 2020), chap. 2.2.7 of Section I—Diagnostic, in Fish health section blue book—Suggested procedures for the detection and identification of certain finfish and shellfish pathogens (2020 ed.): American Fisheries Society, accessed September 8, 2025, accessed September 1, 2025, at https://units.fisheries.org/fhs/fish-health-sectionblue-book-2020/.
Baxa, D., Groff, J., Wishkovsky, A., and Hedrick, R., 1990, Susceptibility of nonictalurid fishes to experimental infection with Edwardsiella ictaluri: Diseases of Aquatic Organisms, v. 8, no. 2, p. 113–117, accessed September 1, 2025, at https://doi.org/10.3354/dao008113.
Bendorf, C.M., Yun, S.C., Kurath, G., and Hedrick, R.P., 2022, Comparative susceptibilities of selected California Chinook salmon and steelhead populations to isolates of L genogroup infectious hematopoietic necrosis virus (IHNV): Animals (Basel), v. 12, no. 13, accessed September 1, 2025, at https://doi.org/10.3390/ani12131733.
Birkbeck, T., Feist, S., and Verner–Jeffreys, D., 2011, Francisella infections in fish and shellfish: Journal of Fish Diseases, v. 34, no. 3, p. 173–187, accessed September 1, 2025, at https://doi.org/10.1111/j.1365-2761.2010.01226.x.
Boerman, I., Jansen, C., and Suselbeek, L., 2006, Quarantine protocols for fishes—Research on the use and extensiveness of quarantine protocols among EUAC and EAZA members: Leeuwarden, Netherlands, Van Hall Larenstein Hogeschool, master's thesis, 61 p., accessed September 8, 2025, accessed September 1, 2025, at https://www.academia.edu/1928590/Quarantine_protocols_for_fishes.
Bootland, L.M., Dobos, P., and Stevenson, R.M., 1991, The IPNV carrier state and demonstration of vertical transmission in experimentally infected brook trout: Diseases of Aquatic Organisms, v. 10, no. 1, p. 13–21, accessed September 1, 2025, at https://doi.org/10.3354/dao010013.
Boscher, S.K., McLoughlin, M., Le Ven, A., Cabon, J., Baud, M., and Castric, J., 2006, Experimental transmission of sleeping disease in one-year-old rainbow trout, Oncorhynchus mykiss (Walbaum), induced by sleeping disease virus: Journal of Fish Diseases, v. 29, no. 5, p. 263–273, accessed September 1, 2025, at https://doi.org/10.1111/j.1365-2761.2006.00716.x.
Bratland, A., and Nylund, A., 2009, Studies on the possibility of vertical transmission of Norwegian salmonid alphavirus in production of Atlantic salmon in Norway: Journal of Aquatic Animal Health, v. 21, no. 3, p. 173–178, accessed September 1, 2025, at https://doi.org/10.1577/H08-038.1.
Brosnahan, C., Ha, H., Booth, K., McFadden, A., and Jones, J., 2017, First report of a rickettsia-like organism from farmed Chinook salmon, Oncorhynchus tshawytscha (Walbaum), in New Zealand: New Zealand Journal of Marine and Freshwater Research, v. 51, no. 3, p. 356–369, accessed September 1, 2025, at https://doi.org/10.1080/00288330.2016.1242081.
Brosnahan, C., Munday, J., Davie, P., Kennedy, L., Preece, M., Barnes, S., Jones, J., and McDonald, W., 2019, Pathogenicity of the bacterium New Zealand rickettsia-like organism (NZ-RLO2) in Chinook salmon Oncorhynchus tshawytscha smolt: Diseases of Aquatic Organisms, v. 134, no. 3, p. 175–187, accessed September 1, 2025, at https://doi.org/10.3354/dao03364.
Brynildsrud, O., Feil, E.J., Bohlin, J., Castillo-Ramirez, S., Colquhoun, D., McCarthy, U., Matejusova, I.M., Rhodes, L.D., Wiens, G.D., and Verner-Jefferys, D.W., 2014, Microevolution of Renibacterium salmoninarum—Evidence for intercontinental dissemination associated with fish movements: The ISME Journal, v. 8, no. 4, p. 746–756, accessed September 1, 2025, at https://doi.org/10.1038/ismej.2013.186.
Cannon, R., 2001, Sense and sensitivity—Designing surveys based on an imperfect test: Preventive Veterinary Medicine, v. 49, no. 3–4, p. 141–163, accessed September 1, 2025, at https://doi.org/10.1016/S0167-5877(01)00184-2.
Cipriano, R.C., Novak, B.M., Flint, D.E., and Cutting, D.C., 2001, Reappraisal of the federal fish health recommendation for disinfecting eggs of Atlantic salmon in iodophor: Journal of Aquatic Animal Health, v. 13, no. 4, p. 320–327, accessed September 1, 2025, at https://doi.org/10.1577/1548-8667(2001)013<0320:ROTFFH>2.0.CO;2.
Closs, G.P., 2024, Salmonids in New Zealand—Old ways in new lands, in Lobon-Cervia, J., Budy, P., and Gresswell, R., eds., Advances in the ecology of stream-dwelling salmonids: Cham, Switzerland, Springer Nature Switzerland AG, Fish & Fisheries, v. 44, p. 441–459, accessed Month Day, Year, accessed September 1, 2025, at https://doi.org/10.1007/978-3-031-44389-3_19.
Colquhoun, D.J., and Duodu, S., 2011, Francisella infections in farmed and wild aquatic organisms: Veterinary Research (Faisalabad), v. 42, 15 p., accessed September 1, 2025, at https://doi.org/10.1186/1297-9716-42-47.
Corbeil, S., and Crane, M., 2019, Piscirickettsia salmonis—Aquatic and terrestrial Australian and New Zealand standard diagnostic procedures: Australian Government Department of Agriculture, Fisheries and Forestry, accessed September 8, 2025, at https://www.agriculture.gov.au/agriculture-land/animal/health/laboratories/procedures/anzsdp/piscirickettsia-salmonis.
Corbeil, S., Hyatt, A.D., and Crane, M.S.J., 2005, Characterisation of an emerging rickettsia-like organism in Tasmanian farmed Atlantic salmon Salmo salar: Diseases of Aquatic Organisms, v. 64, p. 37–44, accessed September 1, 2025, at https://doi.org/10.3354/dao064037.
Corbeil, S., McColl, K.A., and Crane, M.S.J., 2003, Development of a TaqMan quantitative PCR assay for the identification of Piscirickettsia salmonis: Bulletin of the European Associations of Fish Pathologists, v. 23, no. 3, p. 95–101, accessed Month Day, Year, accessed September 1, 2025, at https://eafp.org/download/2003-Volume23/Issue%203/23_095.pdf.
Crane, M.S.J., Hardy-Smith, P., Williams, L.M., Hyatt, A.D., Eaton, L.M., Gould, A., Handlinger, J., Kattenbelt, J., and Gudkovs, N., 2000, First isolation of an aquatic birnavirus from farmed and wild fish species in Australia: Diseases of Aquatic Organisms, v. 43, no. 1, p. 1–14, accessed September 1, 2025, at https://doi.org/10.3354/dao043001.
Davis, H., 1947, Infectious diseases among wild fishes: Transactions of the American Fisheries Society, v. 74, no. 1, p. 21–25, accessed September 1, 2025, at https://doi.org/10.1577/1548-8659(1944)74[21:IDAWF]2.0.CO;2.
Delghandi, M.R., El-Matbouli, M., and Menanteau-Ledouble, S., 2020, Renibacterium salmoninarum—The causative agent of bacterial kidney disease in salmonid fish: Pathogens (Basel, Switzerland), v. 9, no. 10, accessed September 1, 2025, at https://doi.org/10.3390/pathogens9100845.
Dixon, P., Paley, R., Alegria-Moran, R., and Oidtmann, B., 2016, Epidemiological characteristics of infectious hematopoietic necrosis virus (IHNV)—A review: Veterinary Research (Faisalabad), v. 47, 26 p., accessed September 1, 2025, at https://doi.org/10.1186/s13567-016-0341-1.
Doan, Q., Vandeputte, M., Chatain, B., Morin, T., and Allal, F., 2017, Viral encephalopathy and retinopathy in aquaculture—A review: Journal of Fish Diseases, v. 40, no. 5, p. 717–742, accessed September 1, 2025, at https://doi.org/10.1111/jfd.12541.
Dolan, B.P., Fisher, K.M., Colvin, M.E., Benda, S.E., Peterson, J.T., Kent, M.L., and Schreck, C.B., 2016, Innate and adaptive immune responses in migrating spring-run adult Chinook salmon, Oncorhynchus tshawytscha: Fish & Shellfish Immunology, v. 48, p. 136–144, accessed September 1, 2025, at https://doi.org/10.1016/j.fsi.2015.11.015.
Dopazo, C.P., 2020, The infectious pancreatic necrosis virus (IPNV) and its virulence determinants—What is known and what should be known: Pathogens (Basel, Switzerland), v. 9, no. 2, accessed September 1, 2025, at https://doi.org/10.3390/pathogens9020094.
Dopazo, C.P., and Barja, J.L., 2002, Diagnosis and identification of IPNV in salmonids by molecular methods, in Cunningham, C.O., ed., Molecular Diagnosis of Salmonid Diseases—Reviews—Methods and Technologies in Fish Biology and Fisheries v. 3: Dordrecht, Springer, p. 23–48, accessed September 1, 2025, at https://doi.org/10.1007/978-94-017-2315-2_2.
Duman, M., Altun, S., Saticioglu, I.B., and Romalde, J.L., 2023, A review of bacterial disease outbreaks in rainbow trout (Oncorhynchus mykiss) reported from 2010 to 2022: Journal of Fish Diseases, v. 48, no. 9, accessed September 1, 2025, at https://doi.org/10.1111/jfd.13886.
Eaton, W.D., and Hendrick, R.P., 1994, Herpesvirus diseases of salmonids, chap. 2.2.3 of Section I—Diagnostic, in Fish health section blue book—Suggested procedures for the detection and identification of certain finfish and shellfish pathogens (2020 ed.): American Fisheries Society, accessed August 31, 2025, accessed September 1, 2025, at https://units.fisheries.org/fhs/wp-content/uploads/sites/30/2017/08/2.2.3-Herpesvirus-salmonids-2014.pdfhttps://units.fisheries.org/fhs/fish-health-section-blue-book-2020/.
Eguchi, M., Fujiwara, E., and Miyamoto, N., 2000, Survival of Vibrio anguillarum in freshwater environments—Adaptation or debilitation?: Journal of Infection and Chemotherapy—Official Journal of the Japan Society of Chemotherapy, v. 6, no. 2, p. 126–129, accessed September 1, 2025, at https://doi.org/10.1007/PL00012152.
Eissler, Y., Tapia, D., Canales-Aguirre, C.B., and Yáñez, J.M., 2025, Epidemiological insights into infectious pancreatic necrosis virus—Detection, distribution and persistence in Chilean salmon farms: Journal of Fish Diseases, accessed September 1, 2025, at https://doi.org/10.1111/jfd.70047.
Eldar, A., Goria, M., Ghittino, C., Zlotkin, A., and Bercovier, H., 1999, Biodiversity of Lactococcus garvieae strains isolated from fish in Europe, Asia, and Australia: Applied and Environmental Microbiology, v. 65, no. 3, p. 1005–1008, accessed September 1, 2025, at https://doi.org/10.1128/AEM.65.3.1005-1008.1999.
Environmental Protection Authority, 2020, Decision APP203966: New Zealand Government, 4 p., accessed September 1, 2025, at https://www.epa.govt.nz/assets/FileAPI/hsno-ar/APP203966/APP203966-Decision.pdf.
[California] Fish and Game Commission, 2025, Aquaculture disease control regulations, section 245 of California Code of Regulations, title 14, division 1, subdivision 1, chapter 9: Thomson Reuters Westlaw, accessed September 1, 2025, at https://govt.westlaw.com/calregs/Document/I0DFA00545B4D11EC976B000D3A7C4BC3.
Fryer, J., and Hedrick, R., 2003, Piscirickettsia salmonis—A Gram-negative intracellular bacterial pathogen of fish: Journal of Fish Diseases, v. 26, no. 5, p. 251–262, accessed September 1, 2025, at https://doi.org/10.1046/j.1365-2761.2003.00460.x.
Georgiadis, M., Hedrick, R., Carpenter, T., and Gardner, I., 2001, Factors influencing transmission, onset and severity of outbreaks due to white sturgeon iridovirus in a commercial hatchery: Aquaculture (Amsterdam, Netherlands), v. 194, no. 1–2, p. 21–35, accessed September 1, 2025, at https://doi.org/10.1016/S0044-8486(00)00503-2.
Gias, E., Brosnahan, C., Orr, D., Binney, B., Ha, H., Preece, M., and Jones, B., 2018, In vivo growth and genomic characterization of rickettsia-like organisms isolated from farmed Chinook salmon (Oncorhynchus tshawytscha) in New Zealand: Journal of Fish Diseases, v. 41, no. 8, p. 1235–1245, accessed September 1, 2025, at https://doi.org/10.1111/jfd.12817.
Gibello, A., Blanco, M., Moreno, M., Cutuli, M., Domenech, A., Domínguez, L., and Fernández-Garayzábal, J., 1999, Development of a PCR assay for detection of Yersinia ruckeri in tissues of inoculated and naturally infected trout: Applied and Environmental Microbiology, v. 65, no. 1, p. 346–350, accessed September 1, 2025, at https://doi.org/10.1128/AEM.65.1.346-350.1999.
Gilbert, M.A., and Granath, W.O., Jr., 2003, Whirling disease of salmonid fish—Life cycle, biology, and disease: The Journal of Parasitology, v. 89, no. 4, p. 658–667, accessed September 1, 2025, at https://doi.org/10.1645/GE-82R.
Glenn, R.A., Taylor, P.W., Pelton, E.H., Gutenberger, S.K., Ahrens, M.A., Marchant, L.M., and Hanson, K.C., 2015, Genetic evidence of vertical transmission and cycling of Yersinia ruckeri in hatchery-origin fall Chinook salmon Oncorhynchus tshawytscha: Journal of Fish and Wildlife Management, v. 6, no. 1, p. 44–54, accessed September 1, 2025, at https://doi.org/10.3996/012014-JFWM-010.
Godwin, S.E., Morrison, R.N., Knowles, G., Cornish, M.C., Hayes, D., and Carson, J., 2020, Pilchard orthomyxovirus (POMV). II. Causative agent of salmon orthomyxoviral necrosis, a new disease of farmed Atlantic salmon Salmo salar: Diseases of Aquatic Organisms, v. 139, p. 51–68, accessed September 1, 2025, at https://doi.org/10.3354/dao03469.
Good, C., May, T., Crouse, C., Summerfelt, S., and Welch, T., 2016, Assessing the impact of swimming exercise and the relative susceptibility of rainbow trout Oncorhynchus mykiss (Walbaum) and Atlantic salmon Salmo salar L. following injection challenge with Weissella ceti: Journal of Fish Diseases, v. 39, no. 11, p. 1387–1391, accessed September 1, 2025, at https://doi.org/10.1111/jfd.12468.
Gozlan, R.E., and Combe, M., 2023, Emergence of the fungal rosette agent in the world—Current risk to fish biodiversity and aquaculture: Journal of Fungi (Basel, Switzerland), v. 9, no. 4, accessed September 1, 2025, at https://doi.org/10.3390/jof9040426.
Gustafson, L., Gardner, I., and Remmenga, M., 2015, Surveillance for pathogens absent for two years, five years or ten years—When can we stop?: Proceedings of the Third OIE Global Conference on Aquatic Animal Health, Ho Chi Minh City, Vietnam, accessed September 8, 2025, at https://www.researchgate.net/profile/Lori-Gustafson/publication/308764255_Surveillance_for_pathogens_absent_for_two_years_five_years_or_ten_years_when _can_we_stop/links/5a303371458515a13d853df9/Surveillance-for-pathogens-absent-for-two-years-five-years-or-ten-years-when-can-we-stop.pdf.
Gustafson, L., Remmenga, M., Duncan, C., Bliss, C., Bushek, D., Carnegie, R.B., Giray, C., Meyers, T., Davis, K., and Hartman, K., 2024, Risk-based valuation of surveillance data in open environments—Methods application to a key shellfish aquaculture production region: Journal of the World Aquaculture Society, v. 56, no. 1, at https://doi.org/10.1111/jwas.13109.
Hanson, L., Dishon, A., and Kotler, M., 2011, Herpesviruses that infect fish: Viruses, v. 3, no. 11, p. 2160–2191, accessed September 1, 2025, at https://doi.org/10.3390/v3112160.
Heckman, T.I., Yazdi, Z., Older, C.E., Griffin, M.J., Waldbieser, G.C., Chow, A.M., Medina Silva, I., Anenson, K.M., García, J.C., LaFrentz, B.R., Slavic, D., Toohey-Kurth, K.L., Yant, P., Fritz, H.M., Henderson, E.E., McDowall, R., Cai, H., Adkison, M., and Soto, E., 2024, Redefining piscine lactococcosis: Applied and Environmental Microbiology, v. 90, no. 5, accessed September 1, 2025, at https://doi.org/10.1128/aem.02349-23.
Hendrick, R.P., Purcell, M.K., and Kurobe, T., 2012, Salmonid intranuclear microsporidosis, chap. 3.2.17 of Section I—Diagnostic, in Fish health section blue book—Suggested procedures for the detection and identification of certain finfish and shellfish pathogens (2020 ed.): American Fisheries Society, accessed September 1, 2025, at https://units.fisheries.org/fhs/wp-content/uploads/sites/30/2017/08/3.2.17-Nucleospora-2014.pdf.
Hershberger, P., Elder, N., Grady, C., Gregg, J., Pacheco, C., Greene, C., Rice, C., and Meyers, T., 2009, Prevalence of viral erythrocytic necrosis in Pacific herring and epizootics in Skagit Bay, Puget Sound, Washington: Journal of Aquatic Animal Health, v. 21, no. 1, p. 1–7, accessed September 1, 2025, at https://doi.org/10.1577/H08-035.1.
Hewitt, G.C., and Little, R.W., 1972, Whirling disease in New Zealand trout caused by myxosoma cerebralis (Hofer, 1903) (Protozoa: Myxosporida): New Zealand Journal of Marine and Freshwater Research, v. 6, no. 1–2, p. 1–10, accessed September 1, 2025, at https://doi.org/10.1080/00288330.1977.9515406.
Hickey, M.E., and Lee, J.L., 2018, A comprehensive review of Vibrio (Listonella) anguillarum—Ecology, pathology and prevention: Reviews in Aquaculture, v. 10, no. 3, p. 585–610, accessed September 1, 2025, at https://doi.org/10.1111/raq.12188.
Hillestad, B., Johannessen, S., Melingen, G.O., and Moghadam, H.K., 2021, Identification of a new infectious pancreatic necrosis virus (IPNV) variant in Atlantic salmon (Salmo salar L.) that can cause high mortality even in genetically resistant fish: Frontiers in Genetics, v. 12, accessed September 1, 2025, at https://doi.org/10.3389/fgene.2021.635185.
Hirvelä-Koski, V., 2005, Fish pathogens Aeromonas salmonicida and Renibacterium salmoninarum—Diagnostic and epidemiological aspects: University of Helsinki, Thesis, accessed September 8, 2025, at https://helda.helsinki.fi/bitstream/10138/18980/2/fishpath.pdf.
Holopainen, R., Tapiovaara, H., and Honkanen, J., 2012, Expression analysis of immune response genes in fish epithelial cells following ranavirus infection: Fish & Shellfish Immunology, v. 32, no. 6, p. 1095–1105, accessed September 1, 2025, at https://doi.org/10.1016/j.fsi.2012.03.011.
Humphrey, J., 1994, Introductions of aquatic animals to the Pacific Islands—Disease threats and guidelines for quarantine, v. 2 of Perspectives in aquatic exotic species management in the Pacific Islands: Noumea, New Caledonia, South Pacific Commission, 59 p., accessed September 8, 2025, at https://www.vliz.be/imisdocs/publications/314827.pdf.
Huysman, N., Bergmann, D., Nero, P., Larson, J., Sabrowski, V., and Barnes, M.E., 2018, Increasing iodine concentrations during landlocked fall Chinook salmon egg disinfection decrease bacterial numbers with no impact on egg survival: North American Journal of Aquaculture, v. 80, no. 4, p. 363–368, accessed September 1, 2025, at https://doi.org/10.1002/naaq.10042.
Jaramillo, D., Busby, B., Bestbier, M., Bennett, P., and Waddington, Z., 2024, New Zealand rickettsia-like organism and Tenacibaculum maritimum vaccine efficacy study: Journal of Fish Diseases, v. 47, no. 2, accessed September 1, 2025, at https://doi.org/10.1111/jfd.13883.
Johnson-Mackinnon, J.C., 2019, Geographic characterization and environmental detection of Neoparamoeba perurans, the causative agent of amoebic gill disease: University of Tasmania, PhD dissertation, 223 p., accessed September 8, 2025, at https://figshare.utas.edu.au/.
Kelly, R., Souter, B., and Miller, H., 1978, Fish cell lines—Comparisons of CHSE-214, FHM, and RTG-2 in assaying IHN and IPN viruses: Journal of the Fisheries Research Board of Canada, v. 35, no. 7, p. 1009–1011, accessed September 1, 2025, at https://doi.org/10.1139/f78-164.
Kent, M.L., Buchner, C., Watral, V.G., Sanders, J.L., LaDu, J., Peterson, T.S., and Tanguay, R.L., 2011, Development and maintenance of a specific pathogen-free (SPF) zebrafish research facility for Pseudoloma neurophilia: Diseases of Aquatic Organisms, v. 95, no. 1, p. 73–79, accessed September 1, 2025, at https://doi.org/10.3354/dao02333.
Kent, M.L., Shaw, R.W., and Sanders, J.L., 2014, Microsporidia in fish, chap. 20 of Weiss, L.M., and Becnel, J.J., eds., Microsporidia—Pathogens of opportunity (1st ed.): Wiley, p. 493–520, accessed September 1, 2025, at https://doi.org/10.1002/9781118395264.ch20.
Kimura, T., Yoshimizu, M., Tanaka, M., and Sannohe, H., 1981, Studies on a new virus (OMV) from Oncorhynchus masou—I. Characteristics and pathogenicity: Fish Pathology, v. 15, no. 3–4, p. 143–147, at https://doi.org/10.3147/jsfp.15.143.
Kocan, R., Herchberger, P., and Winton, J., 2004, Ichthyophoniasis—An emerging disease of Chinook salmon in the Yukon River: Journal of Aquatic Animal Health, v.16, no. 2, p. 58–72, accessed January 21, 2026, at https://doi.org/10.1577/H03-068.1.
Kumar, G., Menanteau-Ledouble, S., Saleh, M., and El-Matbouli, M., 2015, Yersinia ruckeri, the causative agent of enteric redmouth disease in fish: Veterinary Research, v. 46, no. 103, 10 p., accessed September 1, 2025, at https://doi.org/10.1186/s13567-015-0238-4.
Lane, H.S., Brosnahan, C.L., and Poulin, R., 2020, Aquatic disease in New Zealand—Synthesis and future directions: New Zealand Journal of Marine and Freshwater Research, 42 p., accessed September 1, 2025, at https://doi.org/10.1080/00288330.2020.1848887.
Larenas, J., Bartholomew, J., Troncoso, O., Fernández, S., Ledezma, H., Sandoval, N., Vera, P., Contreras, J., and Smith, P., 2003, Experimental vertical transmission of Piscirickettsia salmonis and in vitro study of attachment and mode of entrance into the fish ovum: Diseases of Aquatic Organisms, v. 56, no. 1, p. 25–30, accessed September 1, 2025, at https://doi.org/10.3354/dao056025.
Lovy, J., Speare, D.J., Stryhn, H., and Wright, G.M., 2008, Effects of dexamethasone on host innate and adaptive immune responses and parasite development in rainbow trout Oncorhynchus mykiss infected with Loma salmonae: Fish & Shellfish Immunology, v. 24, no. 5, p. 649–658, accessed September 1, 2025, at https://doi.org/10.1016/j.fsi.2008.02.007.
M’Gonigle, R.H., 1941, Acute catarrhal enteritis of salmonid fingerlings: Transactions of the American Fisheries Society, v. 70, no. 1, p. 297–303, accessed September 1, 2025, at https://doi.org/10.1577/1548-8659(1940)70[297:ACEOSF]2.0.CO;2.
Mabrok, M., Algammal, A.M., Sivaramasamy, E., Hetta, H.F., Atwah, B., Alghamdi, S., Fawzy, A., Avendaño-Herrera, R., and Rodkhum, C., 2023, Tenacibaculosis caused by Tenacibaculum maritimum—Updated knowledge of this marine bacterial fish pathogen: Frontiers in Cellular and Infection Microbiology, v. 12, 19 p., accessed September 1, 2025, at https://doi.org/10.3389/fcimb.2022.1068000.
Machimbirike, V.I., Crumlish, M., Dong, H.T., Santander, J., Khunrae, P., and Rattanarojpong, T., 2022, Edwardsiella ictaluri—A systemic review and future perspectives on disease management: Reviews in Aquaculture, v. 14, no. 3, p. 1613–1636, accessed September 1, 2025, at https://doi.org/10.1111/raq.12665.
Mardones, F., Martinez-Lopez, B., Valdes-Donoso, P., Carpenter, T., and Perez, A., 2014, The role of fish movements and the spread of infectious salmon anemia virus (ISAV) in Chile, 2007–2009: Preventive Veterinary Medicine, v. 114, no. 1, p. 37–46, accessed September 1, 2025, at https://doi.org/10.1016/j.prevetmed.2014.01.012.
McDowall, R.M., 1994, The origins of New Zealand’s Chinook salmon, Oncorhynchus tshawytscha: Marine Fisheries Review, v. 56, no. 1, p. 1–7. [Also available at https://spo.nmfs.noaa.gov/sites/default/files/pdf-content/mfr5611.pdf.]
McLoughlin, M., and Graham, D., 2007, Alphavirus infections in salmonids—A review: Journal of Fish Diseases, v. 30, no. 9, p. 511–531, accessed September 1, 2025, at https://doi.org/10.1111/j.1365-2761.2007.00848.x.
Menanteau-Ledouble, S., Kumar, G., Saleh, M., and El-Matbouli, M., 2016, Aeromonas salmonicida—Updates on an old acquaintance: Diseases of Aquatic Organisms, v. 120, no. 1, p. 49–68, accessed September 1, 2025, at https://doi.org/10.3354/dao03006.
Mohammadisefat, P., Zorriehzahra, M.J., Adel, M., Allahbeygi Chamjangali, Z., Jabbari, M., Eftekhari, A., Farzipour, H., and Yousefian Jazi, S., 2023, Viral hemorrhagic septicemia virus (VHSV), past, present and future—A review: International Aquatic Research., v. 15, no. 3, p. 191–203, accessed September 1, 2025, at https://doi.org/10.22034/IAR.2023.1983457.1424.
Mohr, P.G., Crane, M.S.J., Hoad, J., Williams, L.M., Cummins, D., Neave, M.J., Shiell, B., Beddome, G., Michalski, W.P., Peck, G.R., Samsing, F., Wynne, J.W., Crameri, S.G., Hyatt, A.D., and Moody, N.J.G., 2020, Pilchard orthomyxovirus (POMV). I. Characterisation of an emerging virus isolated from pilchards Sardinops sagax and Atlantic salmon Salmo salar: Diseases of Aquatic Organisms, v. 139, p. 35–50, accessed September 1, 2025, at https://doi.org/10.3354/dao03470.
Mulcahy, D., and Pascho, R., 1985, Vertical transmission of infectious haematopoietic necrosis virus in sockeye salmon, Oncorhynchus nerka (Walbaum)—Isolation of virus from dead eggs and fry: Journal of Fish Diseases, v. 8, no. 4, p. 393–396, accessed September 1, 2025, at https://doi.org/10.1111/j.1365-2761.1985.tb00962.x.
Munson, A.D., Elliott, D.G., and Johnson, K., 2010, Management of bacterial kidney disease in Chinook salmon hatcheries based on broodstock testing by enzyme-linked immunosorbent assay—A multiyear study: North American Journal of Fisheries Management, v. 30, no. 4, p. 940–955, accessed September 1, 2025, at https://doi.org/10.1577/M09-044.1.
National Oceanic and Atmospheric Administration, 2014, Recovery plan for the evolutionarily significant units of Sacramento River winter-run Chinook salmon and Central Valley spring-run Chinook salmon and the distinct population segment of California Central Valley steelhead: Sacramento, Calif., National Oceanic and Atmospheric Administration, 406 p., accessed August 25, 2025, accessed September 1, 2025, at https://cawaterlibrary.net/document/recovery-plan-for-central-valley-winter-run-chinook-salmon-spring-run-chinook-salmon-and-steelhead/.
Nowlan, J.P., Lumsden, J.S., and Russell, S., 2020, Advancements in characterizing Tenacibaculum infections in Canada: Pathogens (Basel, Switzerland), v. 9, no. 12, 35 p., accessed September 1, 2025, at https://doi.org/10.3390/pathogens9121029.
Nylund, A., Brattespe, J., Plarre, H., Kambestad, M., and Karlsen, M., 2019, Wild and farmed salmon (Salmo salar) as reservoirs for infectious salmon anaemia virus, and the importance of horizontal and vertical transmission: PLoS One, v. 14, no. 4, 24 p., accessed September 1, 2025, at https://doi.org/10.1371/journal.pone.0215478.
Oldham, T., Rodger, H., and Nowak, B.F., 2016, Incidence and distribution of amoebic gill disease (AGD)—An epidemiological review: Aquaculture (Amsterdam, Netherlands), v. 457, p. 35–42, accessed September 1, 2025, at https://doi.org/10.1016/j.aquaculture.2016.02.013.
Pagowski, V.A., Mordecai, G.J., Miller, K.M., Schulze, A.D., Kaukinen, K.H., Ming, T.J., Li, S., Teffer, A.K., Tabata, A., and Suttle, C.A., 2019, Distribution and phylogeny of erythrocytic necrosis virus (ENV) in salmon suggests marine origin: Viruses, v. 11, no. 4, 16 p., accessed September 1, 2025, at https://doi.org/10.3390/v11040358.
Palacios, G., Lovoll, M., Tengs, T., Hornig, M., Hutchison, S., Hui, J., Kongtorp, R.-T., Savji, N., Bussetti, A.V., Solovyov, A., Kristoffersen, A.B., Celone, C., Street, C., Trifonov, V., Hirschberg, D.L., Rabadan, R., Egholm, M., Rimstad, E., and Lipkin, W.I., 2010, Heart and skeletal muscle inflammation of farmed salmon is associated with infection with a novel reovirus: PLoS One, v. 5, no. 7, 7 p., accessed September 1, 2025, at https://doi.org/10.1371/journal.pone.0011487.
Patterson, K., 1996, Modelling the impact of disease-induced mortality in an exploited population—The outbreak of the fungal parasite (Ichthyophonus hoferi) in the North Sea herring (Clupea harengus): Canadian Journal of Fisheries and Aquatic Sciences, v. 53, no. 12, p. 2870–2887, accessed September 1, 2025, at https://doi.org/10.1139/f96-234.
Ross, A., Rucker, R., and Ewing, W., 1966, Description of a bacterium associated with redmouth disease of rainbow trout (Salmo gairdneri): Canadian Journal of Microbiology, v. 12, no. 4, p. 763–770, accessed September 1, 2025, at https://doi.org/10.1139/m66-103.
Samsing, F., Rigby, M., Tengesdal, H.K., Taylor, R.S., Farias, D., Morrison, R.N., Godwin, S., Giles, C., Carson, J., English, C.J., Chong, R., and Wynne, J.W., 2021, Seawater transmission and infection dynamics of pilchard orthomyxovirus (POMV) in Atlantic salmon (Salmo salar): Journal of Fish Diseases, v. 44, no. 1, p. 73–88, accessed September 1, 2025, at https://doi.org/10.1111/jfd.13269.
Samsing, F., Tengesdal, H.K., Nylund, A., Rigby, M., Wiltshire, K.H., Morrison, R.N., Godwin, S., Giles, C., Wilson, T., Mohr, P.G., Hoad, J., and Wynne, J.W., 2022, Development of a new real-time PCR for the detection of pilchard orthomyxovirus (POMV) in apparently healthy fish: Aquaculture (Amsterdam, Netherlands), v. 547, 8 p., accessed September 1, 2025, at https://doi.org/10.1016/j.aquaculture.2021.737404.
Saticioglu, I.B., Onuk, E.E., Ay, H., Ajmi, N., Demirbas, E., and Altun, S., 2023, Phenotypic and molecular differentiation of Lactococcus garvieae and Lactococcus petauri isolated from trout: Aquaculture (Amsterdam, Netherlands), v. 577, accessed September 1, 2025, at https://doi.org/10.1016/j.aquaculture.2023.739933.
Stärk, K.D., Regula, G., Hernandez, J., Knopf, L., Fuchs, K., Morris, R.S., and Davies, P., 2006, Concepts for risk-based surveillance in the field of veterinary medicine and veterinary public health—Review of current approaches: BMC Health Services Research, v. 6, no. 20, 8 p., accessed September 1, 2025, at https://doi.org/10.1186/1472-6963-6-20.
Sterud, E., Forseth, T., Ugedal, O., Poppe, T.T., Jørgensen, A., Bruheim, T., Fjeldstad, H.-P., and Mo, T.A., 2007, Severe mortality in wild Atlantic salmon Salmo salar due to proliferative kidney disease (PKD) caused by Tetracapsuloides bryosalmonae (Myxozoa): Diseases of Aquatic Organisms, v. 77, no. 3, p. 191–198, accessed September 1, 2025, at https://doi.org/10.3354/dao01846.
Thorsen, B., Enger, Ø., Norland, S., and Hoff, K.A., 1992, Long-term starvation survival of Yersinia ruckeri at different salinities studied by microscopical and flow cytometric methods: Applied and Environmental Microbiology, v. 58, no. 5, p. 1624–1628, accessed September 1, 2025, at https://doi.org/10.1128/aem.58.5.1624-1628.1992.
Tisdall, D., and Phipps, J., 1987, Isolation and characterisation of a marine birnavirus from returning quinnat salmon (Oncorhynchus tshawtscha) in the South Island of New Zealand: New Zealand Veterinary Journal, v. 35, no. 12, p. 217–218, accessed September 1, 2025, at https://doi.org/10.1080/00480169./1987.35459.
U.S. Fish and Wildlife Service, 2001, National Wild Fish Health Survey Database, accessed May 27, 2025. accessed September 1, 2025, at https://www.fws.gov/project/national-wild-fish-health-survey-data.
U.S. Fish and Wildlife Service, 2022, Handbook of aquatic health procedures and protocols: U.S. Fish and Wildlife Service, 45 p., accessed September 1, 2025, at https://www.fws.gov/sites/default/files/policy/files/aquaticanimalhealthproceduresandprotocols.pdf.
Vendrell, D., Balcázar, J.L., Ruiz-Zarzuela, I., De Blas, I., Gironés, O., and Múzquiz, J.L., 2006, Lactococcus garvieae in fish—A review: Comparative Immunology, Microbiology and Infectious Diseases, v. 29, no. 4, p. 177–198, accessed September 1, 2025, at https://doi.org/10.1016/j.cimid.2006.06.003.
Wessel, Ø., Hansen, E.F., Dahle, M.K., Alarcon, M., Vatne, N.A., Nyman, I.B., Soleim, K.B., Dhamotharan, K., Timmerhaus, G., Markussen, T., Lund, M., Aanes, H., Devold, M., Inami, M., Løvoll, M., and Rimstad, E., 2020, Piscine orthoreovirus-1 isolates differ in their ability to induce heart and skeletal muscle inflammation in Atlantic salmon (Salmo salar): Pathogens (Basel, Switzerland), v. 9, no. 12, 22 p., accessed September 1, 2025, at https://doi.org/10.3390/pathogens9121050.
Whittington, R., Becker, J., and Dennis, M., 2010, Iridovirus infections in finfish—Critical review with emphasis on ranaviruses: Journal of Fish Diseases, v. 33, no. 2, p. 95–122, accessed September 1, 2025, at https://doi.org/10.1111/j.1365-2761.2009.01110.x.
Whittington, R., Jones, J., Hine, P., and Hyatt, A., 1997, Epizootic mortality in the pilchard Sardinops sagax neopilchardus in Australia and New Zealand in 1995. I. Pathology and epizootiology: Diseases of Aquatic Organisms, v. 28, no. 1, p. 1–15, accessed September 1, 2025, at https://doi.org/10.3354/dao028001.
Wiens, G.D., 2011, Bacterial kidney disease (Renibacterium salmoninarum), in Woo, P.T.K., and Bruno, D.W., eds., Fish diseases and disorders, volume 3—Viral, bacterial and fungal infections (2d ed.): CABI, Wallingford, United Kingdom, p. 338–374. [Also available at https://doi.org/10.1079/9781845935542.0338.]
Winnemem Wintu Tribe, California Department of Fish and Wildlife, and National Oceanic and Atmospheric Administration, 2023, Agreement and co-stewardship framework for reintroduction of anadromous salmonids in the Tribal cultural landscape of the Winnemem Wintu Tribe along the McCloud River watershed: California Department of Fish and Wildlife, [unpaged, 7 p.], accessed August 25, 2025, at https://nrm.dfg.ca.gov/FileHandler.ashx?DocumentID=212724&inline.
Winton, J.R., and Hershberger, P.K., 2016, Viral erythrocytic necrosis, chap. 2.2.6 of Section I—Diagnostic, in Fish health section blue book—Suggested procedures for the detection and identification of certain finfish and shellfish pathogens (2020 ed.): American Fisheries Society, accessed September 1, 2025, at https://units.fisheries.org/fhs/wp-content/uploads/sites/30/2017/08/2.2.6-VEN-2016.pdf.
Yoshimizu, M., 2016, Oncorhynchus masou virus disease, sec. 2.2.13 of Section I—Diagnostic, in Fish health section blue book—Suggested procedures for the detection and identification of certain finfish and shellfish pathogens (2020 ed.): American Fisheries Society, accessed August 31, 2025, accessed September 1, 2025, at https://units.fisheries.org/fhs/wp-content/uploads/sites/30/2017/08/2.2.13-OMV-Disease-2014.pdf.
Yoshiyama, R.M., and Fisher, F.W., 2001, Long time past—Baird Station and the McCloud Wintu: Fisheries (Bethesda, Md.), v. 26, no. 3, p. 6–22, accessed September 1, 2025, at https://doi.org/10.1577/1548-8446(2001)026<0006:LTPBSA>2.0.CO;2.
Young, N.D., Dyková, I., Snekvik, K., Nowak, B.F., and Morrison, R.N., 2008, Neoparamoeba perurans is a cosmopolitan aetiological agent of amoebic gill disease: Diseases of Aquatic Organisms, v. 78, no. 3, p. 217–223, accessed September 1, 2025, at https://doi.org/10.3354/dao01869.
Conversion Factors
Temperature in degrees Celsius (°C) may be converted to degrees Fahrenheit (°F) as follows:
°F = (1.8 × °C) + 32.
Supplemental Information
Concentrations of chemical constituents in water are in milligrams per liter (mg/L).
Analytical sensitivity for real-time quantitative polymerase chain reactions is given as the number of plasmid copies per microliter (copy/μL).
Abbreviations
CHSE
Chinook salmon embryo
CPE
cytopathic effects
IP
intraperitoneal
IPNV
infectious pancreatic necrosis virus
NZ-RLO1
New Zealand rickettsia-like organism 1
NZ-RLO2
New Zealand rickettsia-like organism 2
PCR
polymerase chain reaction
POMV
pilchard orthomyxovirus
qPCR
quantitative polymerase chain reaction
RT-qPCR
real-time quantitative polymerase chain reaction
For more information about the research in this report, contact
Director, Western Fisheries Research Center
U.S. Geological Survey
5501-A Cook Underwood Road,
Cook, Washington 98605-9717
Manuscript approved on February 3, 2026
Publishing support provided by the U.S. Geological Survey Science Publishing Network, Baltimore and Tacoma Publishing Service Centers
Edited by Mary E. Heinz
Design and layout by Yanis X. Castillo
Disclaimers
Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Although this information product, for the most part, is in the public domain, it also may contain copyrighted materials as noted in the text. Permission to reproduce copyrighted items must be secured from the copyright owner.
Suggested Citation
Couch, C.E., Powell, D.B., and Lovy, J., 2026, Evaluation of pathogen risks and testing considerations for Chinook salmon egg movements between New Zealand and California: U.S. Geological Survey Open-File Report 2026–1065, 18 p., https://doi.org/10.3133/ofr20261065.
ISSN: 2331-1258 (online)
Study Area
| Publication type | Report |
|---|---|
| Publication Subtype | USGS Numbered Series |
| Title | Evaluation of pathogen risks and testing considerations for Chinook salmon egg movements between New Zealand and California |
| Series title | Open-File Report |
| Series number | 2026-1065 |
| DOI | 10.3133/ofr20261065 |
| Publication Date | March 03, 2026 |
| Year Published | 2026 |
| Language | English |
| Publisher | U.S. Geological Survey |
| Publisher location | Reston, VA |
| Contributing office(s) | Western Fisheries Research Center |
| Description | vi, 18 p. |
| Country | New Zealand, United States |
| State | California |
| Online Only (Y/N) | Y |
| Additional Online Files (Y/N) | N |


