ORGANIC COMPOUNDS
The organochlorine insecticides and metabolites, and total PCBs analyzed in the fish-tissue and bed-sediment samples are manmade organic compounds. Although those compounds are known to be lipophilic (Hebert and Keenleyside, 1995), analysis of the YELL fish data indicated virtually no correlation between lipid content and contaminant concentration. Therefore, the fish-tissue contaminant concentrations were not adjusted for lipid content. There were too few detections in the bed-sediment data to determine if contaminant concentrations in bed sediment were correlated with the organic carbon concentration in the sediment.
|
Of the 12 organic compounds detected in the fish-tissue samples, the most compounds per site were detected at integrator sites
(fig. 4,
table 4) which reflect multiple environmental settings. Five or six compounds, including
dieldrin, chlordane and metabolites, and metabolites of DDT were detected in common carp from site 10 on the Bighorn River, site 11 on Bighorn Lake, and site 14 on the Shoshone River. Four compounds were detected in the carp from site 18 on Goose Creek and in the cutthroat trout from site 1 on the Yellowstone River at the outlet of Yellowstone Lake. The Goose Creek site reflects mixed environmental settings, including forest, rangeland, agriculture, and urban development. The Yellowstone River at site 1 drains an area of primarily undeveloped land in Yellowstone National Park and surrounding wilderness areas
(fig. 1). At the other integrator sites on the main stem of the Yellowstone River (sites 3, 6, 16, and 24),
p,p´-DDE was the only compound detected in the fish samples. |
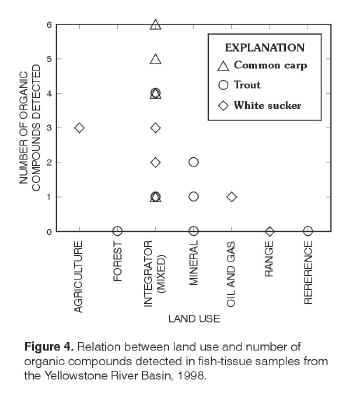
Figure 4. Relation between lad use and
number of organic compounds detected in fish-tissue samples from the
Yellowstone River Basin, 1998.
(Click on image for a larger version, 74kb). |
The fish sample from the agricultural indicator site on Arrow Creek (site 7) contained dieldrin and two metabolites of DDT at relatively low concentrations. No organic compounds were detected in the fish samples from rangeland indicator sites 21 and 22, from forest indicator sites 15 and 17, mineralized indicator site 9, or from reference site 12.
The organic compound most frequently detected in the fish-tissue samples was p,p´-DDE, which is a metabolite of the organochlorine insecticide DDT. The compound
p,p´-DDE was detected at 15 of 21, or 71 percent, of the sampling sites (fig.
5). The largest concentrations of
p,p´-DDE were 58 µg/kg wet weight in common carp from Goose Creek (site 18) and 51.3 µg/kg in cutthroat trout from the Yellowstone River at the outlet of Yellowstone Lake (site 1). The parent compound,
p,p´-DDT, was detected in cutthroat trout from the Yellowstone River (site 1) and West Fork Mill Creek (site 4). |
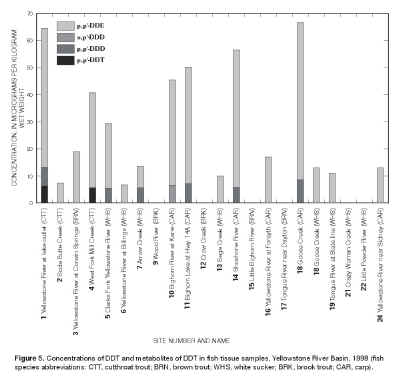
Figure 5. Concentration of DDT and
metabolites of DDT in fish tissue samples, Yellowstone River Basin, 1998
(fish species abbreviations: CTT, cutthroat trout; BRN, brown trout; WHS,
white sucker; BRK, brook trout; CAR, carp).
(Click on image for a larger version, 121kb). |
The presence of DDT, DDD, and DDE in fish-tissue samples from sites in the area of Yellowstone National Park probably reflects the historical spraying of DDT to control spruce budworm. For example, DDT was sprayed on 71,678 acres in the northern part of Yellowstone National Park during 1957 (Cope, 1961). Samples collected in conjunction with the 1957 spraying indicated the presence of DDT in fish tissue from within the sprayed area, downstream of the sprayed area, and in a control sample from Pelican Creek in the Park but well outside of the sprayed area (Cope, 1961). The presence of DDT or its metabolites in fish tissue from sampling sites 1, 2, 3, and 4 and Pelican Creek, which are well outside of the documented spray area, might result from either a historically more extensive application program for DDT than that documented by Cope (1961) or from environmental transport in the more than 40 years since the documented spraying. DDT application was banned in the United States in 1972 due to its persistence in the environment, but is still used in other parts of the world.
Organic compounds including DDT are transported atmospherically on a global scale (Nowell and others, 1999, p. 199), and deposition can increase with altitude (Blais and others, 1998). Global atmospheric deposition probably is not the primary source of DDT in fish-tissue samples from sites in the area of Yellowstone National Park, however, considering the poor correlation of the fish-tissue concentrations of DDT and metabolites with either altitude or precipitation.
Comparison of data from this study to historical data indicates concentrations of DDT in fish tissue have declined with time in the study area. Both this study and Cope (1961) analyzed whole-body brown trout from the Yellowstone River at Corwin Springs (site 3). Cope reported concentrations of 1,100 µg/kg DDT and 500 µg/kg DDE in brown trout collected on August 3, 1957, and 1,200 µg/kg DDT and 900 µg/kg DDE in brown trout collected on August 11, 1957. The brown trout from the same site on August 27, 1998 contained 19 µg/kg
p,p´-DDE; the DDT concentration was less than the reporting limit of 5 µg/kg. The concentrations of total DDT (sum of DDT and metabolites of DDT) in brown trout from site 3 were about two orders of magnitude less in 1998 than in 1957.
The concentration of total DDT in fish also appears to have decreased over time in the Yellowstone River near Sidney (site 24,
table 4). Two samples of common carp collected there during 1984, as part of the U.S. Fish and Wildlife Service National Contaminant Biomonitoring Program (NCBP) nationwide sampling, contained 40 µg/kg and 50 µg/kg total DDT, respectively (Schmitt and others, 1990). The common carp collected on July 28, 1998, from the Yellowstone River near Sidney contained 13 µg/kg total DDT, all in the form of the metabolite
p,p´-DDE. The predominance of DDE and DDD relative to DDT (fig.
5) in the 1998 samples is consistent with the environmental degradation of the parent DDT to its metabolites, as described by Nowell and others (1999).
Concentrations of total DDT in fish from the YELL also were low compared to the geometric mean concentration of 260 µg/kg total DDT in NCBP samples from across the United States in 1984 (Schmitt and others, 1990). The median concentration of total DDT in the YELL samples was 13 µg/kg; the maximum was 66.6 µg/kg. Median concentrations of total DDT in fish-tissue samples from other NAWQA studies in the Rocky Mountain region were 92 µg/kg in the South Platte River Basin (Tate and Heiny, 1996), 69 µg/kg in the Upper Snake River Basin (Maret and Ott, 1997), and 9.6 µg/kg in the upper Colorado River Basin (Stephens and Deacon, 1998). In an investigation of an irrigation drainage project near Riverton, Wyoming
(fig. 1), Peterson and others (1991) reported concentrations of 10 to 20 µg/kg total DDT in rainbow trout and 100 to
200 µg/kg total DDT in common carp.
Fish samples from Goose Creek (site 18) and the Tongue River at State line (site 19) contained PCBs. The concentrations of total PCBs were 190 µg/kg in carp from Goose Creek, 50 µg/kg in white sucker from Goose Creek, and 56 µg/kg in white sucker from the Tongue River at site 19. The source of the PCBs in the Tongue River might be the tributary inflow from Goose Creek. PCBs are synthetic compounds that have been banned in the United States since 1979 because of high bioconcentration factors, but they were widely used as plasticizers, lubricants, and in heat-transfer systems such as capacitors (Smith and others, 1988, p. 26-35). As noted earlier, Goose Creek receives drainage from multiple land uses, including urban development.
Data from other NAWQA study units in the northern and middle Rocky Mountain region indicate the occurrence and distribution of PCBs is related to land use. In the upper Snake River basin, PCBs were detected in 39 percent of the fish-tissue samples, but at only mixed and agricultural land use sites, at concentrations ranging from 50 to 1,900 µg/kg (Maret and Ott, 1997, p. 9, 15). In the South Platte River basin, PCBs were detected in 67 percent of the fish-tissue samples at concentrations ranging from 56 to 1,000 µg/kg and were largest in urban and mixed land-use settings (Tate and Heiny, 1996). In contrast, PCBs were detected at only one site in the upper Colorado River Basin (Stephens and Deacon, 1998). The nationwide average concentration of PCBs in fish tissue from the NCBP was 390 µg/kg in 1984 (Schmitt and others, 1990). The PCB concentrations from an irrigation drainage investigation near Riverton, Wyoming were less than the reporting limit for rainbow trout samples and ranged from 100 to 150 µg/kg wet weight in common carp samples (Peterson and others, 1991). Concentrations of total PCBs, total DDT, and other organochlorine insecticides in fish tissue from the YELL were low compared to concentrations from 20 NAWQA study units across the United States as reported in Wong and others (2000).
Concentrations of organochlorine compounds in fish samples from the YELL were less than the concentrations recommended for the protection of wildlife that eat fish: total chlordane and dieldrin, 100 µg/kg; total DDT, 1,000 µg/kg; and PCBs, 500 µg/kg (National Academy of Sciences and National Academy of Engineering, 1973). Samples of fish fillets from Bighorn Lake, Cooney Reservoir, and Tongue River Reservoir in the Montana portion of the YELL were analyzed for PCBs and no fish consumption advisories were issued (Montana Department of Public Health and Human Services, 1995).
A species comparison conducted at one site indicated higher concentrations of some organic compounds in common carp than in white sucker.
Concentrations of total PCBs and p,p´-DDE were about 4 times as large in common carp as in white sucker collected from Goose Creek
(table 4). Tate and Heiny (1996, p. 70) also found that PCB concentrations were higher in common carp than white sucker among fish collected at the same sites in the South Platte River basin.
Two of the 27 organochlorine insecticides and metabolites and total PCBs that were analyzed in bed-sediment samples were detected. Comparison of organic compounds analyzed in both fish-tissue and bed-sediment samples indicates that of the 12 pesticides detected in fish tissue, only trans-chlordane and
p,p´-DDT were detected in bed-sediment samples. The only sample that had detectable concentrations was from Goose Creek (site 18), which had 1.6 µg/kg trans-chlordane and 2.2 µg/kg
p,p´-DDT
(table 5). The more extensive occurrence of organochlorine insecticides and PCBs in the fish samples probably results from the lipophilic nature of the compounds, chemical partitioning, and bioaccumulation. The data suggest that fish are more sensitive indicators of insecticide and PCB contamination than bed sediment, when the method reporting limits are similar to each other
(table 3).
The other organic compounds detected in bed sediment are semivolatile organic compounds (SVOCs) that can be loosely grouped into two categories based on occurrence and distribution. The first category contains a broad array of SVOCs detected primarily in two specific areas of the study unit. The second category contains a few SVOCs, such as p-cresol, detected in many areas.
|
About 20 polynuclear aromatic hydrocarbon (PAH) compounds occurred in bed-sediment samples from the Yellowstone River at Billings (site 6,
table 5) and Goose Creek (site 18). The maximum concentrations of PAHs also occurred at these two sites, which are integrator sites (fig. 6) located near urban areas. Major sources of PAHs to aquatic systems nationwide include atmospheric deposition, urban sources, industrial discharges, and municipal discharges (U.S. Environmental Protection Agency, 1997). Any or all of the major sources identified by the EPA could be contributing PAHs to the bed sediment at sites 6 and 18. Some of the same PAHs detected at sites 6 and 18 also were detected, generally at lower concentrations, in the samples from site 7 on Arrow Creek and site 19 on the Tongue River. The occurrence of PAHs at Arrow Creek could reflect either an atmospheric source common to both sites 6 and 7, or contamination of water and sediment diverted from the Yellowstone River and used to irrigate lands in the Arrow Creek area. The occurrence of PAHs at the Tongue River site might be a reflection of inflow from Goose Creek. |
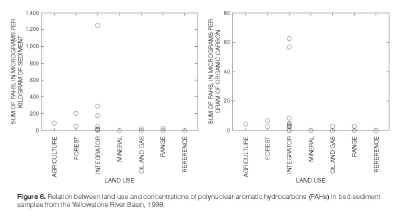
Figure 6. Relation between land use and
concentrations of polynuclear aromatic hydrocarbons (PAHs) in
bed-sediment samples from the Yellowstone River Basin, 1998.
(Click on image for a larger version, 58kb) |
Concentrations of PAHs in the YELL bed-sediment samples were less than the criteria for protection of aquatic life. After adjusting for organic carbon content of the sediment, the concentrations of individual PAHs in the YELL samples, such as benzo (a)
pyrene, benz (a) anthracene, chrysene, fluoranthene, and phenanthrene, were less than the criteria summarized by Gilliom and others (1998, p. 25). The sum of the PAHs in each of the YELL samples also was less than the threshold effect concentration of 290 µg
PAH/g organic carbon calculated by Swartz (1999, p. 783) (fig. 6).
P-cresol, phthalates, and phenol were detected in many of the samples, often at estimated concentrations below the normal method reporting limit
(table 5). One or more PAHs were detected in bed-sediment samples from 14 of 23 sites. Sources of cresols include auto and diesel exhaust, petroleum refining, and other manufacturing; small quantities originate from natural sources (Howard,1989, p. 189-216). P-cresol was detected at nine sites, reaching a maximum concentration of 7,400 µg/kg, which occurred in the bed-sediment sample from the Little Bighorn River (site 15). The maximum concentration of phenol, 140 µg/kg, also was from the Little Bighorn River. Many of the phenol concentrations and nearly all of the phthalate concentrations reported from the YELL bed-sediment samples were less than the 95th percentile concentration in laboratory blanks (Gilliom and others, 1998). For comparing SVOC concentrations in bed sediment from NAWQA study units across the United States, Gilliom and others (1998, p. 13) corrected for laboratory contamination by subtracting the following concentrations from the measured concentration in each environmental sample:
-
Bis(2-Ethylhexyl) phthalate 100 µg/kg
-
Di-n-butyl phthalate 54 µg/kg
-
Butylbenzyl phthalate 64 µg/kg
-
Phenol 27 µg/kg
-
Diethyl phthalate 25 µg/kg
Correction factors are not available specifically for the YELL data, but the reader should be aware that the data for phthalates and phenol likely are influenced by laboratory contamination.



