The purpose of these interpretive discussions is to provide a perspective on regional- and national-scale variations in element and mineral distributions in soils and their likely causes. The significant spatial variations shown by most elements and minerals can commonly be attributed to geologic sources in underlying parent materials, but other spatial variations seem clearly related to additional factors such as climate, the age of soils, transported source material, and anthropogenic influences. We attempt to distinguish the influence of these various factors on a regional and national scale. Numerous more local features might similarly be related to these same factors, but these features also have some probability of being an artifact of a random sampling of variable compositions, so that there is some probability of samples with similar compositions occurring in clusters of two or more adjacent sites by chance. Distinguishing such random occurrences from true variability is beyond the scope of the data from which these maps are constructed. Some caution, therefore, is advisable in interpreting the significance of these more local features unless some unique sources or processes can clearly be related to them.
Strontium (Sr) is a metallic element known for the brilliant red color that its salts give to flares and fireworks. It is also used to refine zinc (Zn) and is combined with iron (Fe) to produce ferrite magnets. Strontium–90, a radioactive isotope of Sr, is a byproduct of nuclear explosions and nuclear reactors. This isotope can substitute for calcium (Ca) in bone tissue and can lead to cancer. Elemental Sr is considered nonessential for humans and is generally nontoxic in normal exposures. Exposure to high levels of Sr can impair bone growth in children. More information about the toxicity of Sr (or other elements and substances) and its potential negative human health impacts can be found at the Agency for Toxic Substances and Disease Registry (ATSDR) website, or click to download a fact sheet about Sr.
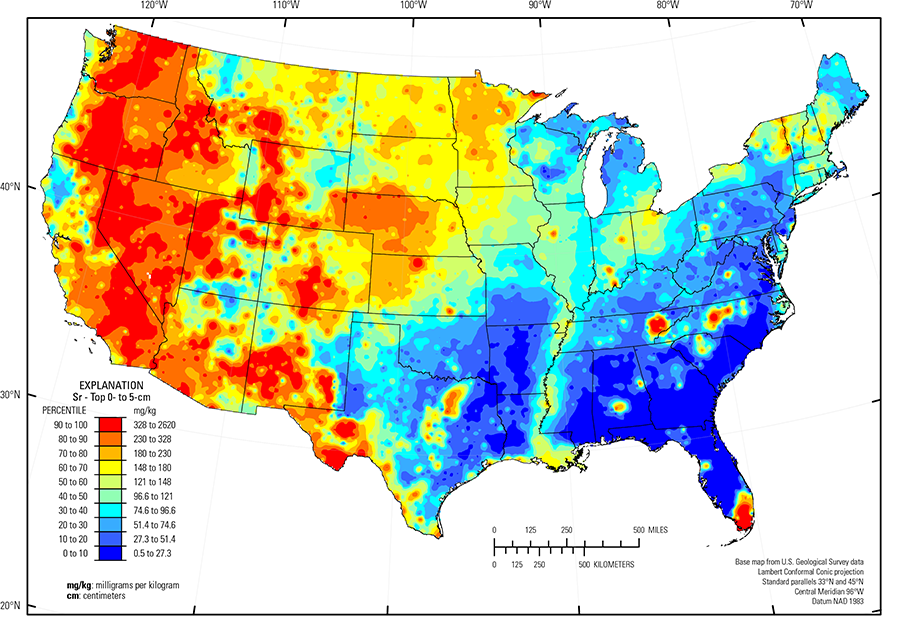
The distribution of Sr in soils of the conterminous United States is primarily controlled by the composition of the underlying soil parent material and climate, as Sr is easily mobilized by weathering. The abundance of Sr in the Earth's upper continental crust is approximately 320 milligrams per kilogram (mg/kg) (Rudnick and Gao, 2003). Because Sr is geochemically very similar to Ca, Sr substitutes for Ca in Ca–rich plagioclase and Ca carbonate minerals. Strontium also partitions strongly into potassium (K) feldspar. Thus, the distribution of Sr in soil is somewhat complex as it can be present in soils that develop on mafic rocks, such as basalt, gabbro, and diabase, and it also may be present in soil where parent materials contain Ca carbonate minerals, such as calcite in limestone, dolomite in dolostone, or gypsum in evaporite deposits. Basalt, gabbro, and diabase have an approximate Sr concentration of 400 mg/kg while limestone has an approximate concentration of 500 mg/kg. Other common rock types contain smaller amounts of Sr with granite having an average concentration of 220 mg/kg and sandstone having an average concentration of 100 mg/kg. Calcite and other carbonate minerals in soil are readily dissolved by infiltrating rainwater or groundwater, and in temperate humid climates the dissolved constituents (such as Sr) may be leached from the soil profile. Many soils in arid and semiarid environments are often calcareous as a lack of rainfall restricts leaching, creating secondary, pedogenic carbonate minerals that may contain Sr.
Despite the relative abundance of Sr in many rocks and minerals, celestite (SrSO4) and strontianite (SrCO3) are the only Sr minerals with sufficient Sr for economic recovery. The distribution of mineral resource deposits with Sr as a commodity (major or minor) in the United States, extracted from the U.S. Geological Survey (USGS) Mineral Resource Data System (MRDS) website, can be seen by hovering the mouse here. Statistics and information on the worldwide supply of, demand for, and flow of Sr–bearing materials are available through the USGS National Minerals Information Center (NMIC) website.
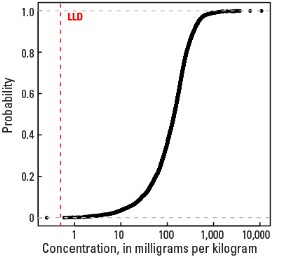
In our data, the median concentration of Sr is 142 mg/kg in the soil C horizon, 122 mg/kg in the soil A horizon, and 121 mg/kg in the top 0- to 5-cm layer. Only three samples out of the entire sample suite had Sr concentrations less than the 0.5-mg/kg lower limit of determination (LLD) (see the summary statistics [open in new window]). Spatial map patterns for Sr in the soil A horizon and the top 0- to-5 cm layer are similar to one another. Anomalously high Sr concentrations in the soil C horizon (one site) and the soil A horizon (one site) were removed prior to constructing the interpolated maps and are shown as diamond symbols on the maps. Differences in the median values between the soil C horizon and topsoils (soil A horizon and top 0- to 5-cm layer), and their respective map patterns likely reflect Sr behavior during weathering under differing climate conditions.
Many of the highest Sr concentrations in the soil C horizon occur in arid to semiarid areas where secondary carbonate or sulfate minerals, including gypsum, calcite, dolomite, or aragonite, are present. Examples of these areas include the Southern Desertic Basins, Plains, and Mountains (USDA, 2006) in southern New Mexico and western Texas; the Great Salt Lake Area (USDA, 2006), Utah, and the Mojave Desert (USDA, 2006), California and Nevada. Strontium in these secondary minerals may have an original carbonate source from fine windblown materials or a more local source from weathering silicate minerals. Soil developed on limestone of the Edwards Plateau (USDA, 2006) in west–central Texas has subdued Sr levels, whereas soil in the Texas Blackland Prairie, Northern Part (USDA, 2006) in east–central Texas that developed on marine marl and chalk has relatively high Sr concentrations. Soils developed in shallow freshwater biogenic marl formed on limestone bedrock within the Everglades (USDA, 2006) also contain significant Sr. Biogenic carbonate (possibly in shelly material) may also explain scattered Sr occurrences along the Gulf Coast of Texas and Louisiana.
Strontium in soil in Washington, Oregon, western and central Idaho, Nevada, southern California and Arizona, southwestern New Mexico, southern Colorado, and western Wyoming reflects inherited plagioclase from soil parent materials that include mafic and intermediate igneous rocks. Soil in southeastern Wyoming and much of northern and western Nebraska, including the Nebraska Sand Hills (USDA, 2006)) (an area of unconsolidated sand and sand sheets) has relatively high Sr concentrations compared to soil in surrounding areas, such as the Northern Rolling High Plains, Southern Part (USDA, 2006) Wyoming, the Central High Tableland (USDA, 2006) Colorado and Kansas, and loess sheets in Kansas and eastern Nebraska. These higher Sr concentrations are attributed to the presence of plagioclase in soil parent materials.
The distribution of Sr in the Upper Midwest is related to glacial provenance. In this region, melting of glacial ice following late Wisconsinan period advances (16,000 to 12,000 years ago) left the region north of the southern glacial limit (Soller and others, 2012) mantled with a blanket of mixed, immature sediments, from which present–day soil developed. Individual ice lobes (Grimley, 2000) created distinct patterns in soil mineralogy and geochemistry because of varying provenance and ice transport paths. Carbonate– and shale–rich 'gray' till in western Minnesota, North Dakota, South Dakota, and Iowa, was derived from Cretaceous sedimentary rocks (dolostone, limestone, and shale) that contained abundant Ca–bearing minerals, including calcite and dolomite. In contrast, 'red' quartz– and feldspar–rich till and lake sediments deposited in northeastern and central Minnesota have a combined local Precambrian basalt and gabbro provenance (Duluth Complex), and a more mixed distant Precambrian granite/basalt provenance that contribute Sr to soil throughout this region. Interestingly, soil developed on glacial deposits of the James Lobe (Grimley, 2000) in central and eastern South Dakota has higher Sr (and manganese (Mn)) concentrations than soil in other areas of 'gray' till in western Minnesota, although the two areas have similar glacial provenance.
Soil throughout the Piedmont (Fenneman and Johnson, 1946) is old and has undergone long–term weathering in a humid to semi–humid environment. Many soils are clay–rich and highly leached, but in a few areas, such as central North Carolina, soils underlain by metamorphosed mafic rocks still contain plagioclase and thus, Sr.
The Gulf and Atlantic Coastal Plain (Fenneman and Johnson, 1946) is bisected by the Southern Mississippi River Alluvium and the Southern Mississippi Valley Loess (USDA, 2006). Alluvial sediments have deposited in the Mississippi River valley as the river flooded in recent geologic time. When these sediments dried, winds picked up the fine material and deposited it in thick loess sheets, mainly along the east side of the river valley. The youngest loess sheets are about 10,000 years old. A pattern of higher Sr concentrations in soils developed on these young sediments reflects long–range transport of Sr–bearing material from areas containing plagioclase from the upper part of the Mississippi River drainage basin.
The high Sr concentrations in the soil A horizon and top 0- to 5-cm layer in Tennessee have no apparent link to a geologic source.
Statistics - 0 TO 5 CM
| Number of samples | 4,841 |
| LLD | 0.5 mg/kg |
| Number below LLD | 0 |
| Minimum | 0.5 mg/kg |
| 5 percentile | 12.7 mg/kg |
| 25 percentile | 63.4 mg/kg |
| 50 percentile | 121 mg/kg |
| 75 percentile | 203 mg/kg |
| 95 percentile | 426 mg/kg |
| Maximum | 2,620 mg/kg |
| MAD | 96.5 mg/kg |
| Robust CV | 79.8% |
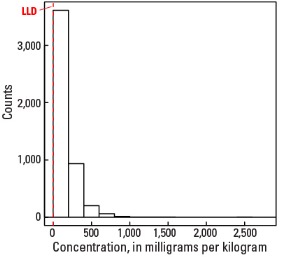
Histogram
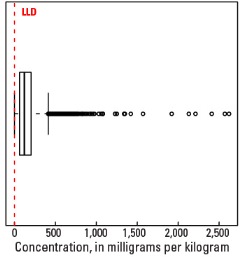
Boxplot
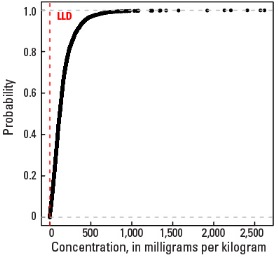
Empirical cumulative distribution function
Statistics - A Horizon
| Number of samples | 4,813 |
| LLD | 0.5 mg/kg |
| Number below LLD | 2 |
| Minimum | <0.5 mg/kg |
| 5 percentile | 13 mg/kg |
| 25 percentile | 64 mg/kg |
| 50 percentile | 122 mg/kg |
| 75 percentile | 204 mg/kg |
| 95 percentile | 432 mg/kg |
| Maximum | 7,080 mg/kg |
| MAD | 97.9 mg/kg |
| Robust CV | 80.2 % |
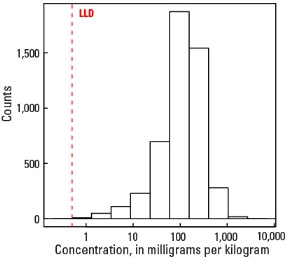
Histogram
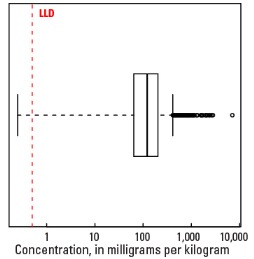
Boxplot
Empirical cumulative distribution function
Statistics - C Horizon
| Number of samples | 4,780 |
| LLD | 0.5 mg/kg |
| Number below LLD | 1 |
| Minimum | <0.5 mg/kg |
| 5 percentile | 14.4 mg/kg |
| 25 percentile | 71.2 mg/kg |
| 50 percentile | 142 mg/kg |
| 75 percentile | 232 mg/kg |
| 95 percentile | 479 mg/kg |
| Maximum | 10,900 mg/kg |
| MAD | 115 mg/kg |
| Robust CV | 81.2 % |
Histogram

Boxplot